Advancing ε-Caprolactone Production: A Safer Solid Peroxyacid Oxidation Strategy for Industrial Scale
Advancing ε-Caprolactone Production: A Safer Solid Peroxyacid Oxidation Strategy for Industrial Scale
The chemical manufacturing landscape for high-value lactones is undergoing a critical transformation driven by the urgent need for safer, more efficient oxidation protocols. Patent CN102731465A introduces a groundbreaking methodology for synthesizing ε-caprolactone, a pivotal intermediate in the production of biodegradable polymers and pharmaceutical excipients. This technology departs from the hazardous conventions of liquid peracid oxidation by employing a novel solid peroxyacid intermediate, specifically peroxysuccinic acid. By shifting the reaction paradigm from a liquid-phase instability to a solid-state stability model, this innovation addresses the chronic safety vulnerabilities and yield limitations that have long plagued the industry. For global procurement leaders and R&D directors, this represents not merely a chemical adjustment but a strategic upgrade in process reliability, offering a pathway to secure supply chains for high-purity pharmaceutical intermediates while mitigating the operational risks associated with explosive peroxide handling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of ε-caprolactone has relied heavily on the Baeyer-Villiger oxidation of cyclohexanone using liquid peracetic acid catalyzed by concentrated sulfuric acid. While this classic route is chemically established, it suffers from severe inherent deficiencies that compromise both economic viability and operational safety. The primary concern lies in the thermal instability of liquid peroxides; during the subsequent distillation and purification stages, the concentration of residual peroxides can reach critical levels, leading to catastrophic explosion hazards that have been documented in pilot and production settings. Furthermore, the aqueous nature of traditional hydrogen peroxide oxidation systems introduces water into the reaction matrix, which acts as a nucleophile to hydrolyze the sensitive lactone ring, significantly depressing overall yield and selectivity. The reliance on strong mineral acid catalysts also necessitates expensive corrosion-resistant equipment and generates substantial acidic wastewater, creating a heavy burden on environmental compliance and waste treatment infrastructure.
The Novel Approach
The methodology disclosed in CN102731465A fundamentally re-engineers the oxidation step by substituting the unstable liquid oxidant with a pre-formed solid peroxysuccinic acid. This solid-state oxidant is generated in situ or separately by reacting succinic anhydride with 70% hydrogen peroxide, resulting in a stable crystalline material that can be handled and dosed with precision. Because the subsequent oxidation of cyclohexanone occurs in an essentially anhydrous environment using this solid reagent, the hydrolytic degradation of the target ε-caprolactone is effectively suppressed, leading to reported selectivity improvements exceeding 99%. Moreover, the stability of the solid peroxyacid eliminates the accumulation of dangerous liquid peroxide concentrations during the workup phase, rendering the rectification process inherently safer. This approach also cleverly integrates a circular economy element, where the succinic acid byproduct is not discarded but recovered and recycled, drastically reducing raw material consumption and waste generation.
Mechanistic Insights into Solid Peroxysuccinic Acid Oxidation
The core chemical advantage of this process lies in the unique reactivity profile of solid peroxysuccinic acid compared to its liquid counterparts. In the Baeyer-Villiger oxidation mechanism, the migration of the alkyl group is facilitated by the electrophilic attack of the peroxy oxygen on the carbonyl carbon of cyclohexanone. In traditional aqueous systems, water molecules compete for the carbonyl center, leading to hydration and subsequent ring-opening hydrolysis of the formed lactone. By utilizing a solid peroxyacid derived from succinic anhydride, the reaction medium remains virtually free of free water, ensuring that the kinetic pathway favors lactone formation over hydrolytic decomposition. This anhydrous condition is critical for maintaining the integrity of the ester linkage in ε-caprolactone, allowing the reaction to proceed with high conversion rates even at moderate temperatures ranging from 20°C to 60°C. The absence of a requirement for strong acid catalysts further simplifies the reaction coordinate, removing the protonation-deprotonation cycles that often lead to side reactions and polymeric impurities.
Beyond the primary oxidation, the patent details a sophisticated mechanism for byproduct management that enhances the overall atom economy of the synthesis. During the oxidation process, succinic acid is generated as a stoichiometric byproduct, which typically presents a disposal challenge. However, this technology leverages the thermal properties of succinic acid to dehydrate it back into succinic anhydride via reactive distillation. This process involves heating the precipitated solids to temperatures between 180°C and 210°C in the presence of a water-carrying agent like o-xylene. The water produced from dehydration is continuously removed via azeotropic distillation, driving the equilibrium towards the formation of the anhydride. This regenerated anhydride can then be recrystallized and fed back into the initial oxidant preparation step, creating a closed-loop system that minimizes fresh raw material input and maximizes resource efficiency.
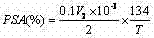
How to Synthesize ε-Caprolactone Efficiently
Implementing this synthesis route requires precise control over the preparation of the solid oxidant and the subsequent oxidation conditions to maximize yield and safety. The process begins with the careful generation of solid peroxysuccinic acid by reacting succinic anhydride with 70% hydrogen peroxide in a dichloromethane solvent system, stabilized by chelating agents to prevent metal-catalyzed decomposition. Once the solid oxidant is isolated and dried, it is slowly introduced into a solution of cyclohexanone in acetic acid, maintaining strict temperature control between 20°C and 60°C to manage the exotherm. Following the reaction, the crude mixture undergoes separation where the ε-caprolactone is distilled, and the remaining solid residues are directed to the recovery unit. For a detailed breakdown of the specific molar ratios, solvent volumes, and distillation parameters required for GMP-compliant production, please refer to the standardized protocol below.
- Prepare solid peroxysuccinic acid by oxidizing succinic anhydride with 70% hydrogen peroxide in the presence of a chelating agent, followed by filtration and drying.
- React the solid peroxysuccinic acid with cyclohexanone in acetic acid solvent at 20-60°C under atmospheric pressure to form the ε-caprolactone solution.
- Recover succinic anhydride from the precipitated solid byproducts using reactive distillation dehydration at 180-210°C, followed by recrystallization for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solid peroxyacid technology translates into tangible reductions in operational risk and long-term cost structures without compromising product quality. The elimination of unstable liquid peroxides from the distillation feed significantly lowers the insurance premiums and safety engineering costs associated with explosion-proof facilities, representing a substantial reduction in overhead capital expenditure. Furthermore, the ability to recycle the succinic anhydride byproduct creates a buffer against volatility in raw material pricing, as the effective consumption of fresh anhydride per kilogram of product is drastically reduced. This internal recycling capability enhances supply chain resilience, ensuring that production continuity is less dependent on external fluctuations in the availability of specialty oxidants or acids.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive corrosion-resistant reactors typically required for concentrated sulfuric acid catalysis, allowing for the use of standard stainless steel equipment which lowers capital investment. Additionally, the high selectivity of the reaction reduces the load on downstream purification columns, leading to significant energy savings during solvent recovery and product distillation. The qualitative reduction in waste treatment costs is also profound, as the process avoids the generation of large volumes of acidic wastewater that require neutralization and specialized disposal.
- Enhanced Supply Chain Reliability: By integrating a raw material recovery loop, the manufacturing process becomes less sensitive to supply disruptions of succinic anhydride, as a significant portion of the demand is met internally through regeneration. The stability of the solid intermediate also allows for safer storage and transportation of reagents if the synthesis is distributed across multiple sites, reducing the logistical complexities associated with shipping hazardous liquid peroxides. This robustness ensures consistent delivery schedules for high-purity pharmaceutical intermediates, critical for maintaining the production timelines of downstream polymer and drug manufacturers.
- Scalability and Environmental Compliance: The absence of heavy metal catalysts and strong mineral acids simplifies the regulatory approval process for new manufacturing sites, as the environmental footprint regarding toxic effluent is minimized. The reactive distillation unit for anhydride recovery is a scalable technology that can be easily expanded from pilot to commercial tonnage without fundamental changes to the process chemistry. This facilitates rapid capacity expansion to meet market demand while adhering to increasingly stringent global environmental standards regarding organic waste and acidic emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the safety, efficiency, and scalability of the solid peroxyacid oxidation method for ε-caprolactone. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D teams planning technology transfer and for procurement specialists assessing the long-term viability of this supply source.
Q: How does the solid peroxyacid method improve safety compared to traditional peracetic acid oxidation?
A: Traditional liquid peracetic acid methods carry significant risks of explosion during distillation due to peroxide concentration. The solid peroxysuccinic acid method utilizes a stable solid oxidant, eliminating the accumulation of unstable liquid peroxides and significantly reducing thermal runaway risks during the rectification process.
Q: What mechanism prevents the hydrolysis of ε-caprolactone in this new process?
A: In conventional hydrogen peroxide indirect oxidation, water presence leads to product hydrolysis. This patent employs a solid peroxyacid generated from succinic anhydride, creating an essentially anhydrous reaction environment that drastically inhibits hydrolysis, thereby boosting yield and selectivity.
Q: Is the raw material succinic anhydride recyclable in this synthesis route?
A: Yes, the process includes a dedicated recovery loop. The solid byproducts precipitated during synthesis are subjected to reactive distillation dehydration at high temperatures (180-210°C) to regenerate succinic anhydride, which is then purified via recrystallization for cyclic use.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ε-Caprolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient chemical processes is vital for the future of the fine chemical industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the solid peroxyacid oxidation are successfully translated into robust industrial realities. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the exacting standards required for pharmaceutical and polymer applications. We understand that consistency is key, and our advanced manufacturing facilities are designed to handle complex oxidation chemistries with the highest levels of safety and precision.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential benefits of switching to this safer route for your supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a sustainable and cost-effective supply of high-purity ε-caprolactone for your global operations.
