Advanced Solvent-Free Manufacturing of 4-Isobutyl Pyrrolidone for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical intermediates, particularly those linked to high-volume drugs like Pregabalin. Patent CN110903228A introduces a groundbreaking preparation method for 4-isobutyl pyrrolidone, utilizing a solvent-free approach that addresses significant limitations in traditional manufacturing. This technology leverages a novel combination of Hofmann degradation and thermal melt cyclization to produce R-type, S-type, or racemic 4-isobutyl pyrrolidone with exceptional efficiency. By eliminating the need for bulk solvents in the final cyclization step, this method not only enhances the environmental profile of the synthesis but also drastically simplifies the downstream purification process. For global supply chain managers and R&D directors, this represents a pivotal shift towards greener, more cost-effective production of high-value pharmaceutical intermediates. The ability to achieve purity levels exceeding 98.5% through simple pulping and filtration underscores the practical viability of this innovation for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-isobutyl pyrrolidone has been plagued by inefficient and hazardous methodologies that struggle to meet modern green chemistry standards. Existing literature, such as WO2016075082, describes routes relying on expensive and highly toxic azo reagents, which introduce severe safety risks and complicate waste disposal protocols due to their hazardous nature. Furthermore, other documented processes like CN107641093A utilize ester intermediates that require extensive solvent feeds and acylating agents, leading to inflated production costs and complex solvent recovery systems. These conventional pathways often suffer from low atom economy and generate substantial chemical waste, creating bottlenecks in large-scale manufacturing operations. The reliance on multiple solvent exchanges and harsh reagents also increases the risk of introducing difficult-to-remove impurities, thereby compromising the final quality of the intermediate. Consequently, manufacturers face elevated operational expenditures and regulatory hurdles when adhering to these outdated synthetic strategies.
The Novel Approach
In stark contrast, the method disclosed in CN110903228A offers a streamlined alternative that bypasses the complexities of esterification and toxic reagent usage. This innovative route initiates with the Hofmann degradation of 3-(carbamoylmethyl)-5-methylhexanoic acid, utilizing sodium hypobromite generated in situ to ensure high reactivity and stability. The subsequent transformation involves a solvent-free melt reaction where the intermediate amino acid is heated directly to induce cyclization, completely eliminating the need for reaction solvents in this critical step. This approach not only reduces the physical footprint of the reaction equipment but also minimizes the energy consumption associated with solvent heating and recovery. The resulting crude product contains minimal oxidation impurities, which can be effortlessly removed through a simple pulping procedure with common organic solvents. This drastic simplification of the workflow enables the production of high-purity 4-isobutyl pyrrolidone suitable for stringent pharmaceutical applications.
Mechanistic Insights into Hofmann Degradation and Thermal Cyclization
The core of this synthetic strategy lies in the precise execution of the Hofmann degradation reaction, which converts the amide functionality into a primary amine with the loss of a carbonyl carbon. In this specific embodiment, sodium hypobromite is generated in situ by reacting bromine with sodium hydroxide, avoiding the instability issues associated with storing pre-formed hypohalite solutions. The substrate, 3-(carbamoylmethyl)-5-methylhexanoic acid, undergoes rearrangement under mild alkaline conditions to yield 3-aminomethyl-5-methylhexanoic acid, commonly known as Pregabalin. This step is critical as it establishes the correct stereochemistry and carbon skeleton required for the final lactam structure. The use of extraction solvents like isobutanol or tert-butanol facilitates the isolation of the amino acid intermediate while maintaining high optical purity for chiral variants. Careful control of pH during the workup ensures the precipitation of the product as a solid, ready for the subsequent thermal transformation.
Following the degradation, the amino acid intermediate undergoes a fascinating solvent-free thermal cyclization to form the pyrrolidone ring. By heating the solid 3-aminomethyl-5-methylhexanoic acid to temperatures between 190°C and 210°C, the molecule enters a molten state where intramolecular nucleophilic attack occurs. The amine group attacks the carboxylic acid moiety, resulting in the elimination of water and the formation of the five-membered lactam ring characteristic of 4-isobutyl pyrrolidone. 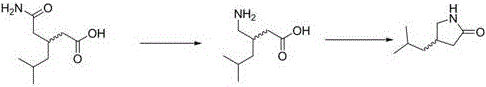 This melt reaction is advantageous because the absence of solvent prevents dilution effects, driving the equilibrium towards product formation and minimizing side reactions. Any minor oxidation impurities formed during the high-temperature process remain soluble in the pulping solvent, allowing the pure crystalline lactam to be isolated via suction filtration. This mechanism ensures a robust process capable of delivering consistent quality across different batches.
This melt reaction is advantageous because the absence of solvent prevents dilution effects, driving the equilibrium towards product formation and minimizing side reactions. Any minor oxidation impurities formed during the high-temperature process remain soluble in the pulping solvent, allowing the pure crystalline lactam to be isolated via suction filtration. This mechanism ensures a robust process capable of delivering consistent quality across different batches.
How to Synthesize 4-Isobutyl Pyrrolidone Efficiently
The synthesis of 4-isobutyl pyrrolidone via this patented method involves a logical sequence of degradation and cyclization steps designed for maximum yield and purity. Operators begin by preparing the sodium hypobromite reagent in situ, ensuring fresh and active species for the degradation of the starting amide acid. Following the isolation of the amino acid intermediate, the process shifts to a solvent-free thermal treatment where precise temperature control is maintained to facilitate ring closure.
- Perform Hofmann degradation on 3-(carbamoylmethyl)-5-methylhexanoic acid using in situ generated sodium hypobromite to yield 3-aminomethyl-5-methylhexanoic acid.
- Heat the resulting amino acid to 190-210°C under solvent-free conditions to induce melt cyclization into the lactam ring.
- Purify the crude melt by pulping in organic solvents like ethyl acetate, followed by filtration and drying to achieve >98.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free technology translates into tangible operational benefits that directly impact the bottom line. The elimination of bulk solvents in the cyclization step significantly reduces the volume of hazardous waste generated, thereby lowering disposal costs and easing regulatory compliance burdens. Additionally, the use of in situ reagent generation mitigates the risks and costs associated with transporting and storing unstable chemical intermediates. This streamlined approach enhances the overall reliability of the supply chain by reducing the number of unit operations and potential failure points in the manufacturing process. Companies adopting this method can expect a more resilient production capability that is less susceptible to fluctuations in solvent availability or pricing.
- Cost Reduction in Manufacturing: The removal of solvents from the cyclization step eliminates the capital and operational expenses related to solvent recovery systems and distillation units. By avoiding expensive azo reagents and complex esterification protocols, the raw material costs are significantly optimized, leading to a more competitive price point for the final intermediate. The high yields reported in the patent examples further contribute to cost efficiency by maximizing the output from each batch of starting material. Furthermore, the simplified purification process reduces labor hours and utility consumption, providing substantial savings in overall manufacturing overheads.
- Enhanced Supply Chain Reliability: Utilizing stable and readily available starting materials like bromine and sodium hydroxide ensures a consistent supply of reagents without dependence on specialized or scarce chemicals. The robustness of the solvent-free melt reaction means that production schedules are less likely to be disrupted by solvent quality issues or supply delays. This reliability is crucial for maintaining continuous production lines and meeting the demanding delivery timelines of downstream pharmaceutical customers. The ability to produce both chiral and racemic forms from flexible starting materials also adds versatility to the supply chain, allowing for rapid adaptation to market demands.
- Scalability and Environmental Compliance: The solvent-free nature of the key cyclization step makes this process inherently safer and easier to scale from pilot plant to commercial production volumes. Reduced solvent usage aligns perfectly with global initiatives for green chemistry and sustainability, helping manufacturers meet increasingly strict environmental regulations. The simplified waste stream, consisting mainly of aqueous layers and minimal organic pulping solvents, is easier to treat and manage compared to complex multi-solvent waste mixtures. This environmental advantage not only protects the company's reputation but also future-proofs the manufacturing site against tightening ecological legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and technical disclosures within the patent documentation. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production frameworks.
Q: How does the solvent-free method improve the purity of 4-isobutyl pyrrolidone?
A: The solvent-free melt reaction minimizes side reactions associated with solvent interactions, and subsequent pulping effectively removes oxidation impurities, yielding products with purity exceeding 98.5%.
Q: What are the safety advantages of using in situ sodium hypobromite?
A: Generating sodium hypobromite in situ from bromine and sodium hydroxide avoids the storage and handling risks of pre-formed hypohalites, which are prone to decomposition and low activity.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the elimination of bulk solvents in the cyclization step significantly reduces waste treatment loads and equipment volume requirements, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Isobutyl Pyrrolidone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering 4-isobutyl pyrrolidone with stringent purity specifications, supported by our rigorous QC labs that validate every batch against international standards. Our expertise in implementing solvent-free and green chemistry technologies allows us to offer products that are not only cost-effective but also environmentally responsible.
We invite you to collaborate with us to leverage this advanced manufacturing capability for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your operational efficiency. Let us be your partner in achieving superior quality and sustainability in pharmaceutical manufacturing.
