Advanced Synthesis of 3,5-Difluoro Biphenyl Derivatives for High-Performance Liquid Crystal Applications
Advanced Synthesis of 3,5-Difluoro Biphenyl Derivatives for High-Performance Liquid Crystal Applications
The rapid evolution of the display industry demands increasingly sophisticated liquid crystal materials that offer superior viscosity coefficients, dielectric constants, and thermal stability. At the heart of this technological advancement lies the critical need for efficient and scalable synthesis routes for key intermediates, specifically the 3,5-difluoro biphenyl derivative class. Patent CN100509722C introduces a groundbreaking preparation method that addresses longstanding economic and technical bottlenecks in the production of these vital electronic chemicals. By leveraging a modified Suzuki-Miyaura coupling strategy, this innovation enables the construction of complex fluorinated biphenyl scaffolds with exceptional purity and yield. For R&D directors and procurement strategists in the optoelectronic sector, understanding the nuances of this patented process is essential for securing a competitive edge in the supply of high-performance liquid crystal precursors.
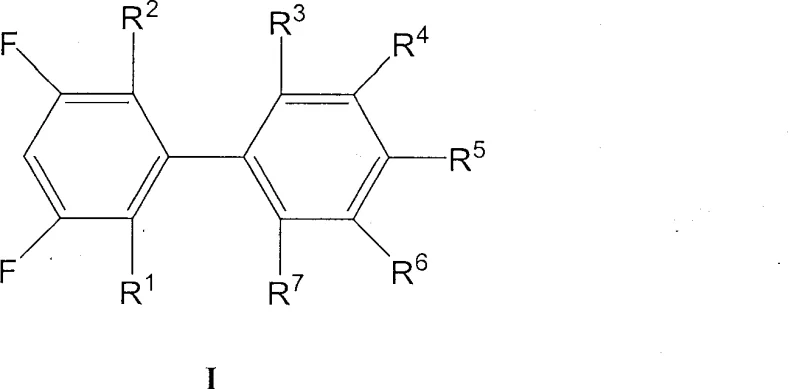
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-difluorobiphenyl derivatives has relied heavily on retrosynthetic disconnections that prioritize the formation of the biaryl bond using fluorinated boronic acid species. Traditional protocols often utilize 3,5-difluorophenylboronic acid as a primary building block, coupling it with various aryl halides to generate the target scaffold. While chemically feasible, this approach suffers from severe economic disadvantages due to the exorbitant cost and limited commercial availability of polyfluorinated boronic acids. Furthermore, conventional methods frequently necessitate high loadings of palladium catalysts to drive the reaction to completion, which not only inflates production costs but also complicates downstream purification processes required to meet stringent heavy metal specifications for electronic grade materials. The reliance on expensive starting materials creates a fragile supply chain vulnerable to price volatility and sourcing delays.
The Novel Approach
The methodology disclosed in CN100509722C fundamentally reverses the traditional coupling logic by employing inexpensive difluorohalobenzenes as the electrophilic partner and readily available phenylboronic acids as the nucleophilic counterpart. This strategic inversion allows manufacturers to bypass the premium pricing associated with fluorinated boron reagents, utilizing instead commodity-grade fluorinated halides which are produced on a massive industrial scale. The novel approach is characterized by its remarkable efficiency, operating effectively with catalyst loadings as low as 0.06 mol% to 0.1 mol%, a drastic reduction compared to standard literature procedures. By optimizing the reaction environment with a specific ternary solvent system, this method ensures high conversion rates while maintaining a clean impurity profile, thereby simplifying the isolation of the final high-purity product required for liquid crystal applications.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling
The core of this synthesis relies on the palladium-catalyzed cross-coupling mechanism, specifically the Suzuki-Miyaura reaction, which facilitates the formation of the carbon-carbon bond between the two aromatic rings. In this catalytic cycle, the active palladium(0) species undergoes oxidative addition into the carbon-halogen bond of the 3,5-difluorohalobenzene, forming an organopalladium(II) intermediate. This step is critical and is influenced by the electron-withdrawing nature of the fluorine substituents, which can enhance the reactivity of the aryl halide towards oxidative addition. Subsequently, the phenylboronic acid, activated by the base present in the reaction mixture, undergoes transmetallation with the palladium complex. The presence of the base is crucial as it converts the neutral boronic acid into a more nucleophilic boronate species, facilitating the transfer of the aryl group to the metal center.
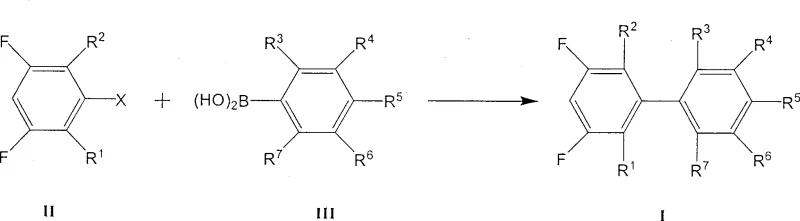
Following transmetallation, the final step involves reductive elimination, where the two aryl groups couple to release the desired 3,5-difluoro biphenyl derivative and regenerate the active palladium(0) catalyst. A key advantage of this specific patented process is the meticulous control over side reactions; the choice of ligands and the mild reaction conditions minimize homocoupling of the boronic acid or dehalogenation of the substrate. The use of a mixed solvent system containing water is particularly beneficial for the solubilization of the inorganic base, ensuring efficient activation of the boronic acid without compromising the solubility of the organic substrates. This mechanistic precision results in a product with a purity exceeding 99%, minimizing the need for extensive chromatographic purification which is often a bottleneck in the manufacturing of electronic chemicals.
How to Synthesize 3,5-Difluoro Biphenyl Derivative Efficiently
The operational protocol for this synthesis is designed for robustness and scalability, making it highly suitable for industrial adoption. The process begins with the careful preparation of the reaction vessel under an inert atmosphere to prevent catalyst deactivation by oxygen. Reagents are charged in specific molar ratios optimized to drive the equilibrium towards product formation while minimizing waste. The reaction is then heated to reflux, allowing the thermal energy to overcome the activation barrier for the coupling event. Following the reaction period, a straightforward workup procedure involving phase separation and solvent removal yields the crude product, which can be further purified by distillation to achieve electronic-grade specifications.
- Charge a reactor with 3,5-difluorohalobenzene, phenylboronic acid derivative, palladium catalyst, and base under inert atmosphere.
- Add a mixed solvent system comprising toluene, water, and ethanol, then heat the mixture to reflux temperature.
- Maintain reflux for approximately 10 hours, followed by phase separation, extraction, and distillation to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers transformative benefits that extend far beyond simple chemical yield improvements. The primary value proposition lies in the substantial reduction of raw material costs achieved by substituting high-value fluorinated boronic acids with low-cost fluorinated halobenzenes. This switch decouples the production cost from the volatile pricing of specialized boron reagents, stabilizing the cost of goods sold (COGS) and improving margin predictability. Additionally, the drastic reduction in catalyst loading means that less precious metal is consumed per kilogram of product, directly lowering the input cost for palladium, a commodity known for its significant price fluctuations. These factors combined create a much more resilient economic model for the manufacturing of liquid crystal intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the strategic selection of starting materials. By utilizing difluorohalobenzenes, which are commodity chemicals produced in vast quantities for various industries, the manufacturer avoids the premium markup associated with custom-synthesized fluorinated boronic acids. Furthermore, the ultra-low catalyst loading (down to 0.06 mol%) significantly reduces the expenditure on palladium complexes. This efficiency also translates to lower costs in waste treatment, as there is less heavy metal contamination to manage in the effluent streams. The cumulative effect is a significantly leaner cost structure that enhances competitiveness in the global market for electronic materials.
- Enhanced Supply Chain Reliability: From a supply chain perspective, relying on widely available difluorohalobenzenes mitigates the risk of single-source dependency often associated with specialized fine chemical intermediates. These halogenated starting materials are produced by multiple suppliers globally, ensuring continuity of supply even during market disruptions. The simplified reaction conditions and the use of common solvents like toluene and ethanol further reduce logistical complexities, as these materials are easy to source and store. This reliability is critical for maintaining consistent production schedules for downstream liquid crystal blenders who require uninterrupted delivery of high-purity intermediates to meet their own manufacturing commitments.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as reflux and distillation that are well-understood in chemical engineering. The high atom economy of the Suzuki coupling, combined with the ability to recover and recycle solvents from the ternary mixture, aligns with modern green chemistry principles. Reduced catalyst usage means lower levels of heavy metals in the final product and waste streams, simplifying compliance with increasingly stringent environmental regulations regarding metal discharge. This environmental compatibility not only reduces regulatory risk but also appeals to end-customers in the electronics sector who are increasingly prioritizing sustainable supply chains in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific advantages and operational parameters detailed in the patent documentation, providing clarity for technical teams evaluating the feasibility of this method for their specific production needs. Understanding these details is crucial for assessing the potential for technology transfer and process optimization within existing manufacturing facilities.
Q: What is the primary cost advantage of this synthesis method?
A: The method utilizes inexpensive difluorohalobenzenes instead of costly 3,5-difluorophenylboronic acids, significantly reducing raw material expenses.
Q: How does the catalyst loading compare to conventional methods?
A: This process achieves high conversion with extremely low catalyst loading (0.06-0.1 mol%), drastically lowering heavy metal residue and catalyst costs.
Q: What represents the optimal solvent system for this reaction?
A: A ternary mixed solvent system of toluene, water, and ethanol provides the ideal balance of solubility and reaction kinetics for high yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Difluoro Biphenyl Derivative Supplier
As the demand for advanced display technologies continues to surge, the need for reliable sources of high-purity liquid crystal intermediates has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in organometallic coupling reactions to deliver superior quality materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global display manufacturers. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3,5-difluoro biphenyl derivative meets the exacting standards required for high-performance liquid crystal formulations.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic benefits of switching to this more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition to a more cost-effective and reliable supply of critical electronic chemical intermediates.
