Revolutionizing Steroid Intermediate Production: A Deep Dive into Efficient Desoximetasone Synthesis
Revolutionizing Steroid Intermediate Production: A Deep Dive into Efficient Desoximetasone Synthesis
In the rapidly evolving landscape of pharmaceutical manufacturing, the demand for high-purity corticosteroid intermediates continues to surge, driven by the critical need for effective topical treatments for skin diseases. A significant breakthrough in this domain is detailed in patent CN114276406A, which outlines a novel preparation method for a key desoximetasone intermediate. This technology represents a paradigm shift from traditional, labor-intensive synthetic routes to a streamlined, fermentation-based approach that prioritizes atom economy and environmental sustainability. By leveraging 21-hydroxypregna-1, 4, 9(11), 16-tetraene-3, 20-diketone-21-acetate as a starting material, the process achieves a remarkable reduction in reaction steps while simultaneously enhancing product quality and yield. For R&D directors and procurement strategists alike, understanding the nuances of this synthesis is crucial for securing a competitive edge in the global supply of active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of desoximetasone and its precursors has been plagued by inefficiencies that drive up costs and complicate supply chains. As highlighted in the background of the patent, earlier methodologies, such as those disclosed in US3099654a1, relied on starting materials like 16 α-methyl-11 α, 21-dihydroxy-1, 4-pregnadiene-3, 20-dione-21-acetate. These conventional routes typically necessitated a cumbersome six-step modification process at the 9, 11, and 21 positions, resulting in a dismal overall yield of approximately 28% from the starting material to the final product. Furthermore, the starting materials for these legacy processes were often difficult to source, creating bottlenecks that jeopardized production continuity. Other prior art, including Chinese patent CN101397322A, attempted to address these issues but still suffered from excessively long reaction routes and unsatisfactory product yields, failing to meet the rigorous demands of modern commercial scale-up.
The Novel Approach
In stark contrast to these outdated techniques, the methodology presented in CN114276406A introduces a concise and robust synthetic strategy that fundamentally alters the economic equation of steroid production. This innovative approach initiates with a cheap, fermentation-derived starting material, 21-hydroxypregna-1, 4, 9(11), 16-tetraene-3, 20-diketone-21-acetate, which is not only cost-effective but also boasts a stable and reliable supply source. The process condenses the transformation into a sequence of bromohydroxylation, epoxidation, methylation, and hydrolysis reactions, effectively bypassing the complex multi-step modifications of the past. This reduction in synthetic complexity translates directly into a total weight yield that reaches an impressive 90.3%, a figure that dwarfs the performance of prior art. Moreover, the absence of dangerous chemical processes and the implementation of green, clean reagents ensure that the manufacturing footprint is minimized, aligning perfectly with contemporary environmental compliance standards.
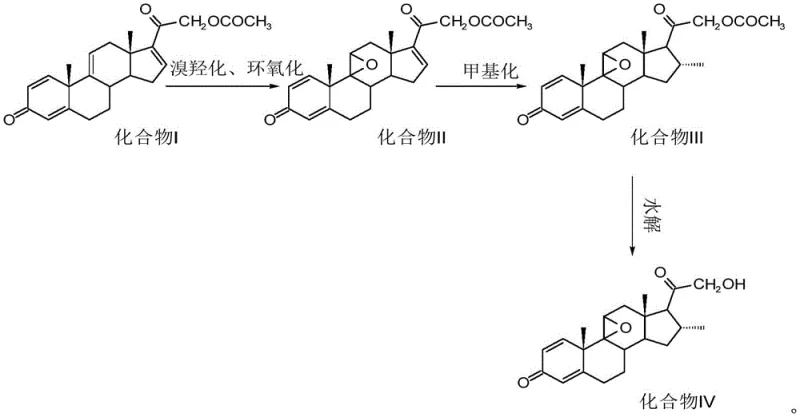
Mechanistic Insights into Bromohydroxylation and Copper-Catalyzed Methylation
The core of this technological advancement lies in the precise orchestration of chemical transformations that construct the complex steroid skeleton with high fidelity. The initial phase involves a tandem bromohydroxylation and epoxidation reaction where Compound I is treated with a brominating agent, such as N-bromosuccinimide (NBS), in an acidic acetone environment. This step is critical for establishing the 9 beta, 11 beta-epoxy functionality, a structural motif essential for the biological activity of the final corticosteroid. Following the bromination, the reaction mixture is subjected to an alkaline environment using agents like sodium carbonate or sodium hydroxide, which facilitates the intramolecular cyclization to form the epoxide ring in Compound II. This cascade reaction is meticulously controlled to prevent over-bromination or degradation of the sensitive diene system, ensuring that the stereochemistry at the 9 and 11 positions is established correctly from the outset.
Subsequent to the formation of the epoxy intermediate, the process employs a sophisticated methylation reaction to introduce the critical 16 alpha-methyl group. This step utilizes a Grignard reagent, specifically methyl magnesium bromide or chloride, in the presence of a copper halide catalyst within an anhydrous tetrahydrofuran solvent system. The inclusion of the copper catalyst is a masterstroke in process chemistry, as it moderates the reactivity of the Grignard reagent, promoting 1,4-addition or specific alkylation while suppressing unwanted side reactions that could lead to impurities. The reaction is conducted under strict nitrogen protection and low temperatures ranging from -15 to -10°C to maintain kinetic control. Finally, a hydrolysis step removes the acetate protecting group at the 21-position using an inorganic base in methanol, yielding the final Compound IV with a purity profile where the main peak area ratio exceeds 98%, demonstrating exceptional impurity control throughout the synthetic pathway.
How to Synthesize Desoximetasone Intermediate Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and high yield. The process is divided into three distinct operational stages: the initial bromination-epoxidation cascade, the copper-mediated methylation, and the final hydrolytic deprotection. Each stage demands precise control over temperature, pH, and reagent stoichiometry to maximize the conversion of the fermentation-derived precursor into the high-value intermediate. The following guide outlines the standardized procedure derived from the patent examples, serving as a foundational reference for process engineers aiming to replicate this efficient route in a pilot or commercial setting.
- Perform bromohydroxylation and epoxidation on Compound I using NBS and alkaline conditions to form the epoxy-ketone structure of Compound II.
- Execute a copper-catalyzed methylation reaction on Compound II using a Grignard reagent in anhydrous tetrahydrofuran to introduce the 16-alpha methyl group.
- Conduct a controlled hydrolysis reaction using an inorganic base/methanol solution to remove the acetate protecting group, yielding the final Compound IV.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers transformative benefits that extend far beyond simple chemical curiosity. The shift to a fermentation-derived starting material fundamentally decouples production costs from the volatility of complex chemical synthesis, providing a stable baseline for pricing negotiations. By eliminating the need for expensive, hard-to-source precursors and reducing the number of unit operations, manufacturers can achieve significant cost reductions in pharmaceutical intermediate manufacturing. This efficiency gain is not merely theoretical; the drastic improvement in yield from roughly 28% in legacy processes to over 90% in this new method implies a massive reduction in raw material consumption per kilogram of finished product, directly impacting the bottom line.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the utilization of cheap, fermentation-based starting materials that are readily available in bulk quantities. Unlike traditional routes that require costly and scarce precursors, this method leverages abundant biological feedstocks, thereby drastically lowering the input cost basis. Furthermore, the reduction in reaction steps minimizes the consumption of solvents, reagents, and energy, leading to substantial operational expenditure savings. The elimination of transition metal catalysts in certain steps, or the use of recoverable copper catalysts, further simplifies the purification process, removing the need for expensive heavy metal scavenging technologies that often inflate production costs in steroid synthesis.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the stability and accessibility of the raw materials employed in this synthesis. Since the starting material is derived from microbial fermentation, it is less susceptible to the geopolitical and logistical disruptions that often plague the supply of complex synthetic organic chemicals. The simplified process flow, characterized by fewer reaction vessels and shorter cycle times, allows for faster turnaround from order to delivery, effectively reducing lead time for high-purity pharmaceutical intermediates. This agility enables suppliers to respond more rapidly to market fluctuations and urgent demand spikes, ensuring a continuous flow of critical materials to downstream API manufacturers without the risk of prolonged stockouts.
- Scalability and Environmental Compliance: From an operational perspective, the process is designed with scalability in mind, utilizing standard reaction conditions that are easily transferable from laboratory to industrial scale. The absence of dangerous chemical processes and the use of green, clean reagents simplify waste management and reduce the burden of environmental compliance, which is increasingly a major cost center for chemical manufacturers. The ability to recycle and reuse mother liquor in actual industrial production further enhances the atom economy and minimizes waste generation, aligning with global sustainability goals. This eco-friendly profile not only mitigates regulatory risks but also enhances the brand value of the final product in markets that prioritize green chemistry and sustainable sourcing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when integrating this route into their supply chain. Understanding these details is essential for making informed decisions about sourcing strategies and process development investments.
Q: What are the primary advantages of the new synthesis route described in CN114276406A compared to traditional methods?
A: The new route utilizes a fermentation-derived starting material which is significantly cheaper and more stable than previous precursors. It reduces the synthetic steps compared to the six-step process in US3099654a1, resulting in a total weight yield reaching approximately 90.3%, vastly superior to the historical 28% yield.
Q: How does this process ensure high purity and minimize impurities?
A: The process employs specific reaction conditions, such as controlled pH adjustment and temperature regulation during the methylation and hydrolysis steps. The use of copper halides as catalysts and precise neutralization with glacial acetic acid helps suppress side reactions, ensuring the main peak area ratio exceeds 98% in liquid chromatography analysis.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the invention explicitly states that the process involves no dangerous chemical processes and uses green, clean reagents. The simplicity of the operation, combined with the ability to recycle mother liquor, makes it highly suitable for large-scale industrial amplification and commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Desoximetasone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of robust and efficient synthetic routes like the one described in CN114276406A for maintaining a competitive position in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and risk-free. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of desoximetasone intermediate adheres to the highest international standards required by top-tier drug developers.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to validate the superior quality and economic advantages of our supply chain solutions for your critical steroid programs.
