Advanced Manufacturing of 3-Halo-4,5-Dihydro-1H-Pyrazoles for Global Pharmaceutical and Agrochemical Supply Chains
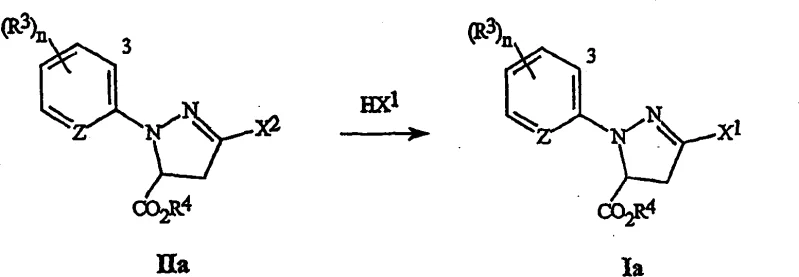
The chemical industry constantly seeks more efficient pathways to synthesize critical heterocyclic building blocks, and Patent CN1826332A presents a significant breakthrough in the preparation of 3-halo-4,5-dihydro-1H-pyrazole compounds. This intellectual property details a versatile method involving the reaction of a 4,5-dihydro-1H-pyrazole compound of formula (II) with a hydrogen halide (HX1) to yield the desired 3-halo derivative of formula (I). For R&D directors and procurement specialists alike, this technology represents a pivotal shift away from traditional, waste-intensive halogenation protocols towards a more streamlined halogen exchange methodology. The process is particularly notable for its ability to accommodate a wide range of substituents on the pyrazole ring, including various carbon moieties and heteroaryl groups, thereby expanding the chemical space accessible for drug discovery and crop protection agent development. By leveraging this specific transformation, manufacturers can access high-purity intermediates essential for the synthesis of advanced insecticides and pharmaceutical agents, ensuring a robust pipeline for downstream applications.
Historically, the synthesis of 3-halo-4,5-dihydro-1H-pyrazoles has relied on methods that present significant operational and environmental challenges. Conventional approaches, such as those reported by J.P. Chupp, often involve reacting oxopyrazolidines with phosphorus oxychloride (POCl3), a reagent known for generating substantial amounts of corrosive waste and requiring rigorous safety controls. Alternatively, methods described by M.V. Gorelik utilize diazonium salt intermediates derived from 3-amino precursors, which can be thermally unstable and pose safety risks during scale-up. Another route involving dipolar cycloaddition with hydrazine chloride intermediates, as published by K.K. Bach, requires multi-step sequences that can impact overall yield and cost efficiency. These limitations underscore the critical need for alternative synthetic strategies that offer broader chemical generality while utilizing low-cost, industrially available reagents. The method disclosed in CN1826332A addresses these pain points by introducing a direct halogen exchange mechanism that bypasses the need for hazardous diazonium species or excessive phosphorus reagents, thereby simplifying the manufacturing workflow.
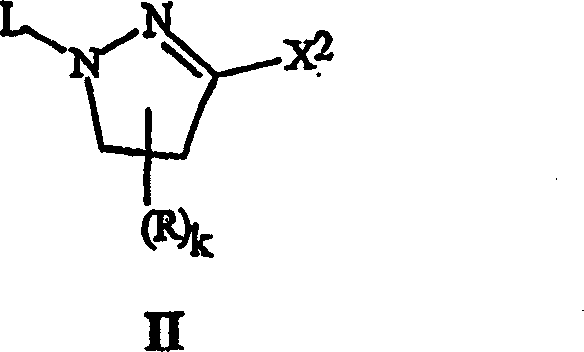
The novel approach centers on the activation of the pyrazole ring through the installation of a superior leaving group at the 3-position, denoted as X2 in formula (II). This leaving group can be a halogen different from the target halogen X1, or more advantageously, a sulfonate ester such as OS(O)mR1 or a phosphate ester like OP(O)p(OR2)2. When treated with a hydrogen halide (HX1), such as HBr or HCl, in a suitable non-nucleophilic solvent, the leaving group is displaced to install the desired halogen atom with high fidelity. This exchange reaction is remarkably efficient, often proceeding at temperatures ranging from 0°C to 100°C, with a strong preference for ambient conditions between 20°C and 30°C. The use of solvents like acetonitrile, dichloromethane, or acetic acid ensures that the reaction mixture remains homogeneous and manageable, facilitating easy workup procedures. Furthermore, the process allows for the use of gaseous HX1 or solutions thereof, providing flexibility in reactor design and reagent handling that is crucial for large-scale operations.
Mechanistically, this transformation relies on the nucleophilic displacement of the leaving group X2 by the halide anion derived from HX1. The efficiency of this substitution is heavily dependent on the nature of X2; sulfonates and phosphates are particularly effective because they stabilize the negative charge upon departure, driving the equilibrium towards the product. In cases where X2 is already a halogen, such as chlorine, the reaction can still proceed effectively if a large excess of the incoming hydrogen halide (e.g., 4 to 10 equivalents of HBr) is used to drive the equilibrium, or if a Lewis acid catalyst like aluminum bromide is employed to enhance the electrophilicity of the carbon center. This mechanistic insight is vital for process chemists aiming to optimize reaction conditions for specific substrates. Additionally, the patent notes that basic functional groups within the molecule may require additional equivalents of HX1 to form salts, ensuring that the free base is available for the substitution reaction. Understanding these nuances allows for precise control over impurity profiles, ensuring that the final 3-halo-4,5-dihydro-1H-pyrazole meets the stringent purity specifications required for regulatory approval in pharmaceutical and agrochemical sectors.
Impurity control is another critical aspect where this method excels compared to prior art. Traditional chlorination with POCl3 often leads to over-chlorination or degradation of sensitive functional groups, resulting in complex impurity spectra that are difficult to purge. In contrast, the halogen exchange method described here is highly selective. For instance, when converting a 3-chloro precursor to a 3-bromo product using HBr, the reaction conditions are mild enough to preserve other sensitive moieties on the ring, such as ester groups or substituted aryl rings. The patent examples demonstrate that products can be isolated with minimal impurities, often requiring only simple filtration or crystallization to achieve high purity. For example, the conversion of a 3-chloro-1-(3-chloro-2-pyridyl) derivative to the corresponding 3-bromo analog yielded a highly crystalline product with negligible starting material remaining, as confirmed by 1H NMR analysis. This level of cleanliness reduces the burden on downstream purification processes, directly translating to cost savings and higher throughput in a commercial setting.
How to Synthesize 3-Halo-4,5-Dihydro-1H-Pyrazoles Efficiently
The synthesis of these valuable intermediates begins with the preparation of the activated precursor, typically an oxopyrazolidine which is subsequently converted into the leaving group-containing species. As detailed in the patent, the precursor of formula (II) can be generated from the corresponding oxo-compound of formula (1) using halogenating agents like phosphorus oxyhalides or sulfonylating agents like tosyl chloride. Once the precursor is secured, the core transformation involves dissolving the compound in a solvent such as acetonitrile and treating it with the chosen hydrogen halide. The reaction progress is monitored until complete conversion is achieved, after which the mixture is neutralized, often with aqueous bicarbonate, and the product is extracted into an organic phase. Detailed standardized synthesis steps see the guide below.
- Preparation of the activated 4,5-dihydro-1H-pyrazole precursor (Formula II) containing a suitable leaving group such as a sulfonate, phosphate, or different halogen.
- Reaction of the Formula II precursor with a hydrogen halide (HX1) in a non-nucleophilic solvent like acetonitrile or dichloromethane at temperatures between 0°C and 100°C.
- Isolation of the final 3-halo-4,5-dihydro-1H-pyrazole product (Formula I) through standard workup procedures including extraction, washing, and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant simplification of the raw material supply chain. By utilizing common hydrogen halides and readily available sulfonating or phosphorylating reagents, manufacturers can reduce their dependency on specialized, high-cost halogenating agents that are subject to volatile market pricing and strict regulatory controls. This shift towards commodity chemicals enhances supply chain resilience, ensuring that production schedules are not disrupted by shortages of niche reagents. Furthermore, the ability to operate at or near ambient temperature reduces energy consumption associated with heating and cooling large reactors, contributing to a lower carbon footprint and reduced operational expenditures. The robustness of the reaction also minimizes batch failures, leading to more predictable delivery timelines for customers relying on these critical intermediates for their own API or agrochemical synthesis.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like phosphorus oxychloride in favor of hydrogen halides drastically simplifies the waste treatment process. Traditional methods generate large volumes of acidic phosphorus waste that require neutralization and disposal, incurring significant environmental compliance costs. In contrast, the halogen exchange method produces cleaner waste streams, primarily consisting of the displaced leaving group and excess acid, which are easier and cheaper to manage. Additionally, the high yields reported in the patent examples, often exceeding 90% and reaching up to 100% in optimized cases, mean that less raw material is wasted per kilogram of product produced. This material efficiency, combined with reduced waste disposal fees, results in a substantially lower cost of goods sold (COGS), allowing suppliers to offer more competitive pricing without sacrificing margins.
- Enhanced Supply Chain Reliability: The use of stable intermediates and mild reaction conditions mitigates the risks associated with thermal runaways or decomposition events that can halt production lines. Conventional diazonium routes are notoriously sensitive to temperature fluctuations, requiring specialized equipment and constant monitoring. The process disclosed in CN1826332A is far more forgiving, capable of being run in standard glass-lined or stainless steel reactors without the need for exotic containment systems. This operational simplicity translates to higher equipment utilization rates and the ability to scale production rapidly in response to market demand. Moreover, the versatility of the method allows for the production of a wide library of analogues using the same core platform, enabling suppliers to respond quickly to custom synthesis requests from R&D partners who are exploring new chemical space for drug candidates or pesticide formulations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns well with modern green chemistry principles. The solvents recommended, such as acetonitrile and ethyl acetate, are widely recycled in the industry, and the avoidance of heavy metal catalysts or stoichiometric oxidants reduces the toxic load of the effluent. The patent explicitly mentions that the reaction can be performed at atmospheric pressure, removing the need for high-pressure autoclaves which are capital-intensive and require rigorous inspection regimes. This ease of scale-up ensures that the transition from pilot plant to commercial manufacturing is seamless, reducing the time-to-market for new products. For global supply chains, this means a more reliable source of high-quality intermediates that comply with increasingly stringent international environmental regulations, safeguarding the reputation of both the supplier and the end-user.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this halogen exchange technology. These answers are derived directly from the experimental data and theoretical framework provided in Patent CN1826332A, offering clarity on how this method can be integrated into existing manufacturing workflows. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their specific product portfolios.
Q: What are the advantages of this halogen exchange method over conventional POCl3 chlorination?
A: Unlike conventional methods using phosphorus oxychloride which generate significant acidic waste and require harsh conditions, this patent-disclosed method utilizes hydrogen halides (HX1) under milder conditions (often ambient temperature), allowing for broader functional group tolerance and simplified waste management.
Q: Which leaving groups (X2) are most effective for this transformation?
A: The patent highlights that sulfonates (OS(O)mR1), phosphates (OP(O)p(OR2)2), and halogens different from the target halogen serve as excellent leaving groups. Specifically, mesylates, tosylates, and chlorides have demonstrated high conversion rates when treated with the appropriate hydrogen halide.
Q: Is this process scalable for commercial production of agrochemical intermediates?
A: Yes, the process is designed for scalability. It employs commercially available low-cost reagents and solvents like acetonitrile and dichloromethane, and operates effectively at or near atmospheric pressure, making it highly suitable for large-scale manufacturing of complex pharmaceutical and agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Halo-4,5-Dihydro-1H-Pyrazoles Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics and crop protection solutions. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is smooth and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 3-halo-4,5-dihydro-1H-pyrazoles meets the exacting standards required by global regulatory bodies. Our facility is equipped to handle the specific solvent systems and reagent handling requirements outlined in this patent, allowing us to deliver consistent quality regardless of order volume.
We invite you to collaborate with us to leverage this advanced synthetic technology for your projects. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific molecular targets, demonstrating how this halogen exchange method can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can accelerate your development timelines and secure a sustainable supply chain for your most critical chemical building blocks.
