Scalable Synthesis of Isolicoflavonol: A Technical Breakthrough for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust, scalable methods for producing bioactive flavonoids, particularly those with potent anti-ulcer and anti-tumor properties. Patent CN112047916A discloses a highly efficient method for synthesizing Isolicoflavonol, a prenylated flavonol originally isolated from Licorice (Glycyrrhiza uralensis). This compound has demonstrated significant biological activity, including inhibition of Helicobacter pylori without inducing drug resistance, making it a critical lead compound for next-generation gastrointestinal therapeutics. Unlike traditional extraction methods which suffer from low yields and supply chain volatility, this patented synthetic route offers a deterministic, chemistry-driven approach. By leveraging classic organic transformations such as the Houben-Hoesch reaction, oxidative cyclization, and Claisen rearrangement, the process ensures high purity and reproducibility. For R&D directors and procurement specialists, understanding this pathway is essential for securing a stable supply of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the supply of Isolicoflavonol has been entirely dependent on extraction from natural plant sources, primarily the roots of Licorice. This biological sourcing model presents inherent and severe limitations for industrial manufacturing. Firstly, the concentration of Isolicoflavonol in natural plants is extremely low, necessitating the processing of massive quantities of biomass to obtain gram-scale amounts, which drives up costs exponentially. Secondly, agricultural sourcing is subject to seasonal variations, climate change impacts, and geographical restrictions, leading to unpredictable supply continuity. Furthermore, the extraction process involves complex separation techniques to isolate the target molecule from hundreds of other co-extracted flavonoids and impurities, often requiring extensive chromatography that is difficult to scale. These factors combined result in a high-cost, low-yield supply chain that cannot meet the rigorous demands of modern pharmaceutical development.
The Novel Approach
The synthetic methodology described in CN112047916A fundamentally shifts the production paradigm from extraction to total synthesis, utilizing inexpensive and commercially available starting materials such as phloroglucinol and substituted benzaldehydes. The core innovation lies in the strategic construction of the flavonol skeleton followed by the precise installation of the isopentenyl side chain via a Claisen rearrangement strategy. Instead of relying on nature to assemble the complex structure, this route builds the molecule step-by-step with full control over regioselectivity and stereochemistry. The process employs a modular protection-deprotection strategy that allows for the selective functionalization of hydroxyl groups, ensuring that the final product matches the natural structure exactly. This chemical approach eliminates the variability of biological sources and enables true commercial scale-up.
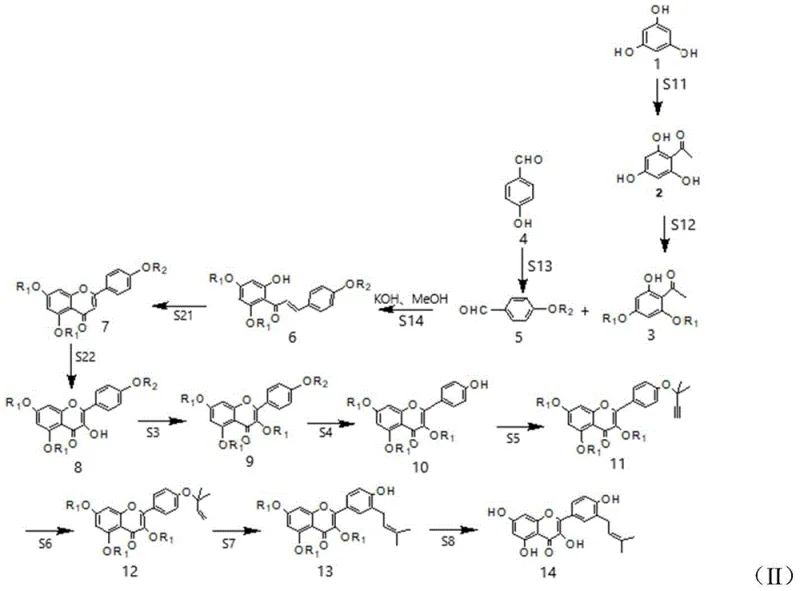
Mechanistic Insights into Oxidative Cyclization and Claisen Rearrangement
The chemical elegance of this synthesis is best observed in the oxidative cyclization and the subsequent rearrangement steps. The formation of the flavone core from the chalcone precursor is achieved through an iodine-mediated oxidative cyclization. In this mechanism, molecular iodine acts as both an oxidant and a Lewis acid promoter, facilitating the intramolecular attack of the phenolic oxygen onto the alpha,beta-unsaturated ketone system. This reaction typically proceeds in polar aprotic solvents like DMSO or pyridine at elevated temperatures (around 110°C), efficiently closing the C-ring to form the flavone scaffold. Following this, a second oxidation step converts the flavone into the corresponding flavonol (3-hydroxyflavone). The patent suggests using oxidants such as potassium hydrogen persulfate (Oxone) or m-CPBA, which selectively introduce the hydroxyl group at the C-3 position without over-oxidizing the sensitive aromatic rings.
Perhaps the most critical mechanistic feature for establishing the biological activity of Isolicoflavonol is the installation of the isopentenyl group. Direct prenylation of the flavonol ring often leads to mixtures of regioisomers. To circumvent this, the patent employs a clever "propargyl-Claisen" sequence. First, the 4'-hydroxyl group is alkylated with 3-chloro-3-methyl-1-butyne to form a propargyl ether. This alkyne is then partially hydrogenated using a poisoned palladium catalyst (Lindlar catalyst) to yield the corresponding allyl ether with high Z-selectivity. Upon heating in a high-boiling solvent like N,N-diethylaniline, this allyl ether undergoes a [3,3]-sigmatropic Claisen rearrangement. This pericyclic reaction migrates the isopentenyl chain from the oxygen atom to the ortho-carbon position on the B-ring, perfectly reconstructing the natural product's substitution pattern with high fidelity.
How to Synthesize Isolicoflavonol Efficiently
The synthesis of Isolicoflavonol requires precise control over reaction conditions, particularly during the protection and rearrangement phases. The process begins with the preparation of protected acetophenone and benzaldehyde derivatives, which are condensed to form the chalcone backbone. Subsequent oxidative cyclization builds the heterocyclic core, while selective methylation or benzylation protects sensitive hydroxyl groups during the harsh rearrangement conditions. The final deprotection steps utilize reagents like boron tribromide (BBr3) or catalytic hydrogenation to reveal the free phenolic hydroxyls, yielding the final active pharmaceutical ingredient. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below.
- Condense protected hydroxy-acetophenone with protected benzaldehyde to form a chalcone intermediate.
- Perform oxidative cyclization to generate the flavone core, followed by further oxidation to flavonol.
- Execute selective protection, propargylation, partial hydrogenation, and thermal Claisen rearrangement to install the isopentenyl group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from extraction to the synthetic route described in CN112047916A offers transformative economic and logistical benefits. The primary advantage is the decoupling of production from agricultural cycles. By synthesizing Isolicoflavonol from petrochemical-derived precursors like phloroglucinol and acetonitrile, manufacturers can guarantee year-round production stability, eliminating the risks associated with crop failures or seasonal shortages. This reliability is crucial for maintaining continuous API manufacturing lines and meeting strict Just-In-Time delivery schedules required by major pharmaceutical clients. Furthermore, the use of commodity chemicals significantly reduces the raw material cost base compared to purchasing expensive botanical extracts.
- Cost Reduction in Manufacturing: The synthetic route utilizes widely available, low-cost reagents such as potassium carbonate, iodine, and common organic solvents like acetone and methanol. By avoiding the capital-intensive infrastructure required for large-scale botanical extraction and purification, the overall cost of goods sold (COGS) is drastically reduced. Additionally, the high selectivity of the Claisen rearrangement minimizes the formation of difficult-to-separate regioisomers, thereby reducing waste disposal costs and improving overall process mass intensity (PMI).
- Enhanced Supply Chain Reliability: Relying on a defined chemical synthesis allows for better inventory planning and forecasting. Unlike natural extracts which can vary in potency and impurity profile from batch to batch, this synthetic method produces a consistent chemical entity with a defined impurity spectrum. This consistency simplifies quality control testing and regulatory filing, as the manufacturing process is fully validated and reproducible across different production sites, ensuring a resilient supply chain.
- Scalability and Environmental Compliance: The reaction conditions described, such as the use of heterogeneous catalysts (Pd/C) and recyclable solvents, are amenable to large-scale batch processing. The elimination of complex chromatographic purifications in favor of crystallization-based isolation steps makes the process inherently more scalable. Moreover, the ability to control waste streams chemically, rather than dealing with tons of plant biomass residue, aligns better with modern green chemistry principles and environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Isolicoflavonol. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy for R&D and procurement evaluation.
Q: What are the key advantages of this synthetic route over natural extraction?
A: This synthetic method overcomes the limitations of natural extraction, such as low yield, seasonal dependency, and complex purification. It utilizes readily available raw materials like phloroglucinol and allows for consistent, large-scale production with controlled impurity profiles.
Q: How is the isopentenyl group introduced in this synthesis?
A: The isopentenyl group is installed via a strategic sequence involving 1,1-dimethylpropargylation at the 4'-OH position, followed by Lindlar-catalyzed hydrogenation to the alkene, and finally a thermal Claisen rearrangement to migrate the group to the ortho-position on the B-ring.
Q: What catalysts are used in the oxidative cyclization steps?
A: The process employs iodine (I2) in solvents like DMSO or pyridine for the oxidative cyclization of chalcones to flavones, and oxidants such as potassium hydrogen persulfate (Oxone) or m-CPBA for the subsequent conversion to flavonols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isolicoflavonol Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of securing high-quality intermediates for your drug development pipeline. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the complexities of flavonoid synthesis, including the precise execution of Claisen rearrangements and sensitive hydrogenation steps. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to ensure every batch of Isolicoflavonol meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for this valuable compound. Our experts can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our synthetic capabilities can lower your overall procurement costs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability for your upcoming projects.
