Advancing Pharmaceutical Intermediate Production with Metal-Free Visible Light Asymmetric Catalysis
The pharmaceutical industry is constantly seeking innovative synthetic methodologies that balance high stereochemical precision with environmental sustainability and cost efficiency. Patent CN115108875A introduces a groundbreaking approach to constructing nitrogen-containing heteroaromatic chiral cyclic alcohols through visible light asymmetric catalysis. This technology represents a significant paradigm shift from traditional transition-metal-mediated processes to a more sustainable, metal-free photoredox strategy. By utilizing an organic photocatalyst (DPZ) in synergy with a spirocyclic chiral phosphoric acid (CPA), this method enables the direct reductive coupling of enones or enals with nitrogen-containing heteroaromatics. The resulting chiral cyclic alcohols serve as critical scaffolds for neuroprotective agents and other bioactive molecules, demonstrating potent activity in inhibiting oxidative stress in PC12 cells. For R&D directors and process chemists, this patent offers a robust pathway to access complex chiral architectures that were previously difficult to synthesize with high fidelity.
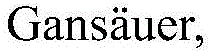
Historically, the synthesis of cyclic alcohols via reductive coupling of carbonyls and alkenes has relied heavily on stoichiometric amounts of reducing metals or toxic radical initiators. Conventional methods often employ reagents such as tributyltin hydride (Bu3SnH), samarium diiodide (SmI2), or various transition metal complexes to generate the necessary ketyl radical anions. While effective, these traditional approaches suffer from significant drawbacks, including the generation of hazardous waste, difficulties in removing toxic metal residues from the final product, and often poor control over stereoselectivity due to the high reactivity of free radicals. Furthermore, ionic synthesis methods frequently require harsh conditions, such as strong bases or cryogenic temperatures, which limit functional group tolerance and increase operational complexity. The reliance on heavy metals also poses a severe bottleneck for pharmaceutical manufacturing, where regulatory limits on residual metals are becoming increasingly stringent, necessitating costly and time-consuming purification steps.
In stark contrast, the novel approach disclosed in CN115108875A leverages the power of visible light photoredox catalysis combined with chiral hydrogen bonding to overcome these limitations. This dual-catalytic system operates under mild conditions, typically between -40°C and 25°C, using simple blue LED irradiation to drive the reaction. The use of an organic photocatalyst like DPZ eliminates the need for expensive and toxic transition metals, thereby inherently reducing the environmental footprint and simplifying the workup procedure. More importantly, the introduction of the chiral phosphoric acid catalyst provides a rigid chiral environment that effectively controls the stereochemical outcome of the radical addition. This allows for the simultaneous generation of two stereocenters with high enantioselectivity and diastereoselectivity, a feat that is exceptionally challenging in radical chemistry. The method exhibits broad substrate scope, tolerating various substituents on both the aromatic ketone and the heteroaromatic alkene components, making it a versatile tool for library synthesis and lead optimization.
Mechanistic Insights into Visible Light Dual Catalytic Cyclization
The success of this transformation lies in the intricate interplay between the photoredox cycle and the chiral hydrogen-bonding activation. Upon irradiation with blue light (450-455nm), the DPZ photocatalyst enters an excited state, enabling it to act as a potent single-electron transfer (SET) agent. It facilitates the reduction of the enone substrate, likely via interaction with the Hantzsch ester (HE) or benzothiazoline (BT) serving as the terminal reductant and hydrogen atom donor. This process generates a ketyl radical species which subsequently undergoes intramolecular addition to the tethered nitrogen-containing heteroaromatic olefin. The critical innovation is the role of the chiral phosphoric acid (CPA), which acts as a Brønsted acid catalyst. Through hydrogen bonding interactions, the CPA activates the imine or nitrogenous moiety of the heteroaromatic ring, organizing the transition state within a well-defined chiral pocket. This precise spatial arrangement directs the approach of the radical species, ensuring that the new carbon-carbon bond is formed with high stereocontrol. This synergistic mechanism avoids the use of strong bases required in ionic pathways and prevents the racemization often seen in uncontrolled radical processes.
From an impurity control perspective, this mechanism offers distinct advantages for process chemistry. Traditional radical reactions using tin hydrides often produce organotin byproducts that are notoriously difficult to separate from the desired product, leading to complex purification trains and yield losses. In the visible light protocol, the byproducts are primarily the oxidized forms of the organic reductants (e.g., pyridine derivatives from Hantzsch ester), which are generally more polar and easier to remove via standard aqueous workups or chromatography. Additionally, the mild reaction conditions minimize thermal degradation of sensitive functional groups, reducing the formation of decomposition impurities. The high diastereoselectivity observed (often >19:1 dr) implies that the formation of unwanted diastereomers is suppressed at the source, rather than requiring separation later. This inherent selectivity translates directly to higher overall process efficiency and reduced solvent consumption during purification, aligning with green chemistry principles.
How to Synthesize Chiral Cyclic Alcohols Efficiently
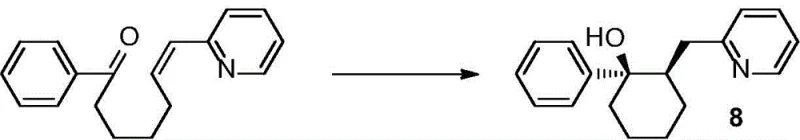
Implementing this visible light asymmetric catalysis protocol requires careful attention to reaction setup and light source calibration to ensure reproducibility and optimal yields. The general procedure involves dissolving the enone precursor, the chiral catalyst, the photocatalyst, and the reductant in a dry, degassed organic solvent such as 1,2-dichloroethane or toluene. The reaction vessel must be rigorously excluded from oxygen, typically achieved through multiple freeze-pump-thaw cycles followed by backfilling with inert gas like argon, as oxygen can quench the radical intermediates and deactivate the photocatalyst. Once prepared, the mixture is stirred under the irradiation of blue LEDs at a controlled distance to maintain consistent photon flux. The reaction progress is monitored until the starting material is consumed, after which standard isolation techniques are employed. The following reaction scheme illustrates a representative example of this transformation, highlighting the conversion of a phenyl-substituted enone to the corresponding chiral cyclic alcohol with high optical purity.
- Prepare the reaction mixture by dissolving the enone substrate, Hantzsch ester (HE) or benzothiazoline (BT) reductant, DPZ photocatalyst, and chiral phosphoric acid (CPA) in an organic solvent like 1,2-dichloroethane under inert atmosphere.
- Subject the reaction vessel to freeze-pump-thaw cycles to ensure complete removal of oxygen, then maintain the system under argon protection to prevent radical quenching.
- Irradiate the mixture with blue LED light (450-455nm) at controlled temperatures ranging from -40°C to 25°C until completion, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free visible light catalysis technology presents compelling economic and logistical benefits. The most significant advantage is the elimination of transition metal catalysts, which removes the necessity for expensive scavenging resins or complex filtration steps designed to meet strict residual metal specifications in active pharmaceutical ingredients (APIs). This simplification of the downstream processing workflow leads to substantial cost savings in terms of both materials and labor. Furthermore, the use of commercially available organic photocatalysts and reductants ensures a stable and reliable supply chain, reducing dependency on scarce or geopolitically sensitive precious metals. The mild reaction conditions also contribute to lower energy consumption compared to processes requiring cryogenic cooling or high-pressure hydrogenation, further enhancing the overall cost-effectiveness of the manufacturing process.
- Cost Reduction in Manufacturing: The transition to a metal-free catalytic system fundamentally alters the cost structure of producing these complex chiral intermediates. By avoiding the use of expensive noble metals and the associated validation costs for metal removal, manufacturers can significantly reduce the cost of goods sold (COGS). The high selectivity of the reaction minimizes the loss of valuable starting materials to side products, improving the overall mass balance and yield. Additionally, the simplified purification process reduces solvent usage and waste disposal costs, contributing to a leaner and more economical production model that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: Relying on organic catalysts and standard LED light sources mitigates supply chain risks associated with the volatility of the precious metals market. The reagents used in this protocol, such as Hantzsch esters and phosphoric acids, are readily accessible from multiple chemical suppliers, ensuring continuity of supply even during market disruptions. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process does not require specialized high-pressure equipment or extreme temperature infrastructure. This flexibility enables faster response times to market demand fluctuations and supports a more agile supply chain strategy for pharmaceutical partners.
- Scalability and Environmental Compliance: The scalability of photochemical reactions has historically been a concern, but advancements in flow chemistry and LED reactor design have made large-scale implementation feasible. This specific protocol operates at ambient or near-ambient temperatures, reducing the thermal load on reactors and simplifying heat management during scale-up. From an environmental standpoint, the absence of heavy metals and the use of visible light as a traceless reagent align perfectly with modern sustainability goals and regulatory requirements. This green profile facilitates smoother regulatory approvals and enhances the corporate social responsibility (CSR) standing of the manufacturing entity, making it an attractive option for environmentally conscious clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light asymmetric catalysis technology. These answers are derived from the detailed experimental data and mechanistic studies presented in the patent documentation, providing clarity on the practical aspects of adopting this synthetic route. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this method into their existing production pipelines.
Q: What are the advantages of using DPZ photocatalyst over traditional metal catalysts?
A: The DPZ photocatalyst is metal-free, eliminating the risk of heavy metal contamination in the final API, which simplifies downstream purification and ensures compliance with stringent pharmaceutical regulatory standards regarding residual metals.
Q: How does this method improve stereoselectivity compared to conventional radical reactions?
A: By combining photoredox catalysis with chiral hydrogen bond catalysis (CPA), the method achieves unprecedented control over the stereochemistry of the radical addition, resulting in high enantiomeric excess (ee) and diastereomeric ratios (dr) that are difficult to achieve with ionic methods.
Q: Is this synthesis method scalable for commercial production?
A: Yes, the reaction utilizes mild conditions (ambient to low temperature) and standard LED light sources, avoiding extreme pressures or temperatures, which facilitates safe and efficient scale-up from laboratory to industrial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Cyclic Alcohol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN115108875A can be reliably reproduced on an industrial scale. Our state-of-the-art facilities are equipped with specialized photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates. We understand the critical nature of chiral integrity in drug development and are committed to delivering high-quality chiral cyclic alcohols that support your R&D and commercial manufacturing needs with consistency and precision.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time to market with our superior synthesis capabilities and dedication to excellence in fine chemical manufacturing.
