Revolutionizing Agrochemical Intermediate Production via Novel Chlorination and Ring-Opening Strategies
Revolutionizing Agrochemical Intermediate Production via Novel Chlorination and Ring-Opening Strategies
The global demand for high-efficacy insecticides continues to drive innovation in the synthesis of key pharmaceutical and agrochemical intermediates. Patent CN1538953A, filed in October 2004, introduces a groundbreaking methodology for the preparation of 2-(hydroxyphenyl)-2-(alkoxyimino)-N-methylacetamide derivatives. These compounds serve as critical building blocks in the manufacture of advanced crop protection agents. The technical significance of this patent lies not merely in the final structure, but in the robustness of the synthetic pathway, which addresses long-standing challenges regarding scalability and raw material accessibility. For R&D directors and procurement specialists alike, understanding the nuances of this process is essential for securing a reliable supply chain of high-purity agrochemical intermediates. The invention specifically targets the efficient conversion of readily available enol acetates into complex amide structures through a novel chlorination and ring-opening sequence.
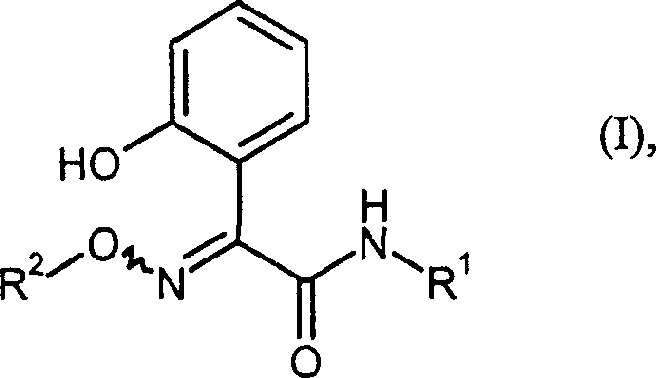
This report provides a deep technical dissection of the patented process, evaluating its potential for commercial scale-up and cost optimization. By shifting away from obscure starting materials and closed-vessel reactions, the described method offers a tangible pathway to reducing manufacturing lead times and enhancing overall process safety. As we analyze the specific reaction conditions and mechanistic insights, it becomes clear that this technology represents a significant leap forward for manufacturers seeking to optimize their production of insecticide precursors. The following sections will detail the comparative advantages over conventional methods, the specific chemical mechanisms at play, and the strategic commercial benefits for supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN1538953A, the synthesis of 2-(2-hydroxyphenyl)-2-(alkoxyimino)-N-methylacetamide derivatives was fraught with significant technical hurdles that hindered industrial adoption. Historical methods often relied on starting materials such as methyl 2-(2-hydroxyphenyl)-2-(methoxyimino)acetate or 1-benzofuran-2,3-dione 3-(O-methyloxime), which are notoriously difficult to obtain in bulk quantities with consistent quality. Furthermore, existing literature describes the preparation of dichlorobenzofuran-3-ones via chlorination of benzofuran-3-ones in closed vessels, a setup that poses severe safety risks and engineering challenges when attempting to scale to multi-ton production levels. The inability to perform these reactions in open systems limits heat dissipation and pressure management, creating bottlenecks in manufacturing throughput. Additionally, previous routes often generated dichlorobenzofuran-3-one merely as a by-product rather than a targeted intermediate, leading to poor atom economy and complex purification workflows that drive up operational costs.
The Novel Approach
The methodology outlined in the patent fundamentally reimagines the synthetic route by utilizing 3-acetoxybenzofuran (Formula II) as a primary feedstock. This starting material is not only more accessible but also reacts in a surprisingly efficient manner with chlorine gas. Unlike prior art, this novel process allows for the chlorination of the enol acetate to occur with the simultaneous removal of the acetate group, directly yielding the reactive 2,2-dichloro-1-benzofuran-3(2H)-one intermediate. This transformation is particularly noteworthy because it proceeds effectively in open vessels under atmospheric pressure, drastically simplifying the reactor engineering requirements. The subsequent ring-opening of this dichloro-intermediate with simple alkylamines, such as methylamine, provides a direct and high-yielding entry point to the core acetamide scaffold. By streamlining these initial steps, the process eliminates the need for exotic reagents and hazardous high-pressure equipment, paving the way for a more economical and safer manufacturing protocol.
Mechanistic Insights into Chlorination and Amine-Mediated Ring Opening
The core chemical innovation of this process resides in the unexpected reactivity observed during the chlorination step. Typically, the chlorination of enol acetates might be expected to result in alpha-chlorination while retaining the ester functionality. However, under the specific conditions defined in the patent—using glacial acetic acid as a diluent at temperatures between 15°C and 80°C—the reaction facilitates a concurrent deacetylation. This results in the formation of the gem-dichloro ketone structure within the benzofuran ring system. This mechanistic pathway is crucial because it generates a highly electrophilic carbonyl carbon flanked by two electron-withdrawing chlorine atoms, making the ring susceptible to nucleophilic attack. The stability of this intermediate is managed by the choice of solvent and temperature control, ensuring that the reactive species does not decompose before the next step.
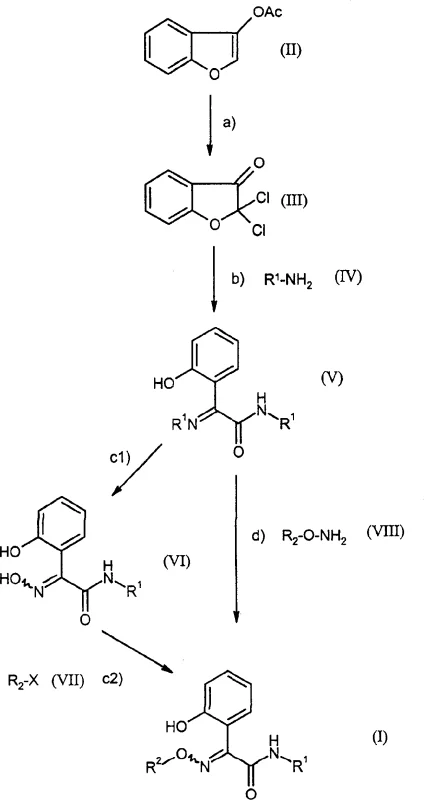
Following the formation of the dichloro-intermediate, the mechanism shifts to a nucleophilic substitution and ring-opening sequence. When treated with an amine of Formula IV (such as methylamine), the nucleophile attacks the carbonyl carbon, leading to the cleavage of the furan ring. This ring-opening event is facilitated by the electron-withdrawing nature of the chlorine atoms, which activates the carbonyl towards nucleophilic addition. The result is the formation of the 2-(2-hydroxyphenyl)-2-imino-N-alkylacetamide structure (Formula V). This intermediate serves as a versatile pivot point; it can be subsequently converted to the final alkoxyimino product either through a two-step oximation and alkylation sequence (Steps c1 and c2) or via a direct one-pot reaction with an alkoxyamine (Step d). The ability to control the stereochemistry (E/Z isomers) and minimize side reactions during this ring-opening phase is critical for maintaining the high purity required for downstream agrochemical applications.
How to Synthesize 2-(Hydroxyphenyl)-2-(Alkoxyimino)-N-Methylacetamide Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize impurity formation. The process begins with the careful introduction of chlorine gas into a solution of the enol acetate, where monitoring the exotherm is vital to prevent over-chlorination or degradation. Following the isolation of the dichloro-intermediate, the subsequent amination step must be conducted with stoichiometric precision to ensure complete ring opening without excessive consumption of the amine reagent. The final functionalization steps offer flexibility, allowing manufacturers to choose between a stepwise approach for maximum control or a telescoped one-pot procedure for increased throughput. Detailed standard operating procedures regarding solvent selection, temperature ramps, and workup protocols are essential for replicating the high success rates reported in the patent examples.
- Chlorinate 3-acetoxybenzofuran (Formula II) with chlorine gas in glacial acetic acid at 15-80°C to form 2,2-dichloro-1-benzofuran-3(2H)-one (Formula III).
- React the dichloro-intermediate (Formula III) with a C1-C4 alkylamine (Formula IV) in an ether or alcohol solvent to effect ring opening, yielding the hydroxyphenyl acetamide derivative (Formula V).
- Convert the intermediate (Formula V) to the final product (Formula I) either by reacting with hydroxylamine followed by alkylation, or directly with an alkoxyamine in a buffered medium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling strategic advantages that extend beyond simple chemistry. The primary value driver is the substantial reduction in raw material complexity. By utilizing 3-acetoxybenzofuran and common alkylamines, manufacturers can source inputs from a broader base of suppliers, mitigating the risk of single-source bottlenecks that often plague the agrochemical intermediate market. The elimination of closed-vessel high-pressure reactions further reduces capital expenditure requirements for specialized reactor infrastructure, allowing for production in standard glass-lined or stainless steel vessels commonly found in fine chemical facilities. This compatibility with existing infrastructure significantly shortens the timeline for technology transfer and commercial scale-up.
- Cost Reduction in Manufacturing: The process achieves cost optimization through improved atom economy and simplified unit operations. By combining chlorination and deacetylation into a single step, the method reduces the total number of processing stages, which directly correlates to lower labor, energy, and solvent consumption costs. Furthermore, the use of inexpensive solvents like glacial acetic acid and methyl tert-butyl ether, which can often be recovered and recycled, minimizes waste disposal expenses. The high yields reported in the patent examples suggest that less raw material is required per kilogram of finished product, driving down the variable cost of goods sold and improving overall margin potential for the final insecticide active ingredient.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of commodity chemicals that are less susceptible to market volatility compared to specialized heterocyclic starting materials used in older methods. The robustness of the reaction conditions—operating at atmospheric pressure and moderate temperatures—reduces the likelihood of unplanned shutdowns due to equipment failure or safety incidents. This operational stability ensures a consistent flow of intermediates to downstream formulation plants, safeguarding against production delays that could impact the seasonal availability of crop protection products. The ability to scale from kilogram to multi-ton batches without changing the fundamental chemistry provides a secure foundation for long-term supply agreements.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with modern green chemistry principles. The avoidance of heavy metal catalysts and the use of aqueous workups in certain steps simplify effluent treatment and reduce the burden of hazardous waste management. The open-vessel nature of the chlorination step, when properly engineered with scrubbing systems, allows for better containment of off-gases compared to batch venting from high-pressure autoclaves. These factors contribute to a lower environmental footprint, facilitating easier permitting and compliance with increasingly stringent global regulations on chemical manufacturing emissions and waste discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this technology for their specific production lines. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the primary advantage of the chlorination step in Patent CN1538953A?
A: The process uniquely achieves simultaneous chlorination and deacetylation of the enol acetate starting material in an open vessel, overcoming the limitations of prior art which required closed systems and yielded difficult-to-handle by-products.
Q: Why is this synthesis route considered more scalable for industrial production?
A: The reaction steps operate at atmospheric pressure and moderate temperatures (15-80°C), utilizing commercially available solvents like glacial acetic acid and methyl tert-butyl ether, which simplifies reactor requirements and enhances safety profiles compared to high-pressure alternatives.
Q: How does this method impact the purity profile of the final insecticide intermediate?
A: By avoiding complex starting materials and utilizing a direct ring-opening strategy, the process minimizes the formation of hard-to-remove side products, resulting in a cleaner crude profile that facilitates easier downstream purification and higher final assay values.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(Hydroxyphenyl)-2-(Alkoxyimino)-N-Methylacetamide Supplier
The technical potential of Patent CN1538953A is immense, but realizing its commercial value requires a partner with deep expertise in process development and scale-up engineering. NINGBO INNO PHARMCHEM stands ready to assist global agrochemical companies in translating this laboratory-scale innovation into robust commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We understand the critical importance of maintaining stringent purity specifications and rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for final API or agrochemical synthesis.
We invite you to engage with our technical procurement team to discuss how this novel chlorination and ring-opening technology can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this route for your operations. We encourage you to contact us today to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
