Advanced Manufacturing of O-Phenoxyalkylhydroxylamines: A Technical Breakthrough for Agrochemical Intermediates
The global demand for high-performance agrochemicals continues to drive innovation in intermediate synthesis, particularly for precursors used in cyclohexanone herbicides. Patent CN1150418A discloses a highly efficient process for producing isomer mixtures of O-phenoxyalkylhydroxyl amines and their corresponding oximes, representing a significant departure from traditional protecting-group strategies. This technology addresses critical pain points in fine chemical manufacturing by streamlining the synthetic route and enhancing isomeric purity without relying on cumbersome separation techniques. For R&D teams and procurement specialists alike, understanding this methodology is essential for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality. The core innovation lies in the direct conversion of O-(2-hydroxyethyl)-oximes into activated sulfonates, which subsequently undergo nucleophilic substitution with phenols, bypassing the need for nitrogen protection entirely.
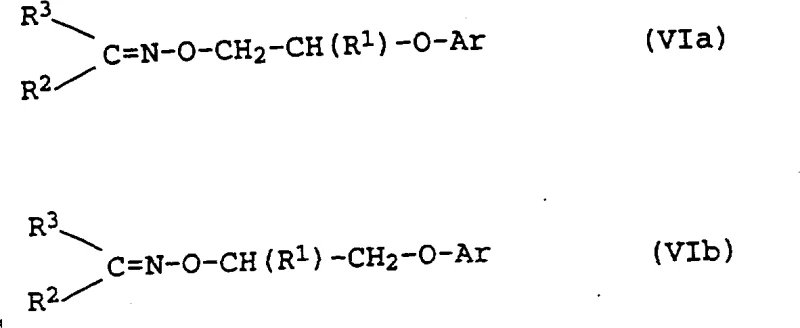
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of O-alkylated hydroxylamines has been plagued by selectivity issues and inefficient multi-step sequences. Conventional wisdom dictated the use of protecting groups, most notably N-hydroxyphthalimide, to prevent unwanted reactions at the nitrogen atom during alkylation. While chemically sound in theory, this approach suffers from substantial practical drawbacks in an industrial setting. The installation and subsequent removal of the phthalimide moiety add at least two distinct operational units to the process flow, increasing capital expenditure and operational complexity. Furthermore, the deprotection step often results in the loss of valuable protecting group compounds and generates stoichiometric amounts of phthalic acid waste, creating significant environmental disposal burdens. Yield losses are compounded at each stage, meaning that the overall throughput of the facility is drastically reduced compared to a linear, protection-free pathway.
The Novel Approach
The methodology outlined in the patent data revolutionizes this landscape by utilizing a sulfonate activation strategy that renders nitrogen protection obsolete. By converting the hydroxyl group of the starting oxime directly into a leaving group (such as a mesylate or tosylate), the molecule becomes primed for nucleophilic attack by a phenol species. This transformation allows for the direct formation of the ether linkage while maintaining the integrity of the oxime functionality. The process is not only shorter but also demonstrates a remarkable ability to enrich the desired isomer during the reaction sequence itself. Instead of fighting thermodynamic equilibria to separate isomers post-synthesis, this route leverages kinetic preferences and solubility differences during crystallization to deliver a product with superior purity profiles. This represents a paradigm shift in cost reduction in agrochemical manufacturing, turning a previously wasteful process into a lean, high-yielding operation.
Mechanistic Insights into Sulfonate Activation and Etherification
The heart of this synthetic strategy involves a two-stage activation and substitution mechanism that ensures high fidelity in bond formation. Initially, the O-(2-hydroxyethyl)-oxime reacts with a sulphonyl halogenide, such as methanesulfonyl chloride, in the presence of a tertiary amine base like triethylamine. This step converts the poor leaving group (hydroxyl) into an excellent leaving group (sulfonate ester), creating a highly electrophilic center at the alkyl chain. The reaction conditions are mild, typically proceeding between 0°C and 40°C, which minimizes side reactions such as elimination or decomposition of the sensitive oxime group. Following isolation or telescoping, the sulfonate intermediate is subjected to reaction with a phenol derivative in the presence of a strong inorganic base, such as potassium hydroxide. The phenoxide anion generated in situ acts as a potent nucleophile, displacing the sulfonate group via an SN2-type mechanism to form the O-phenoxyalkyloxime backbone.
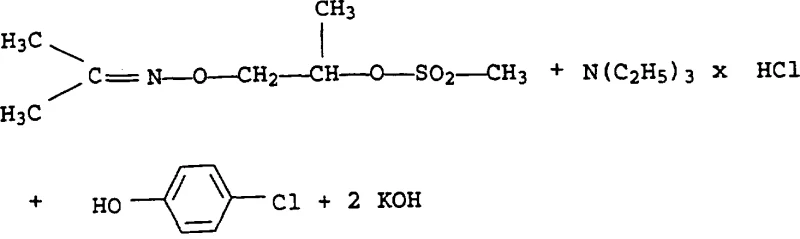
Crucially, the mechanism also accounts for the observed isomer enrichment. The patent data indicates that the content of the undesired 'b' isomer decreases progressively through steps a, b, and c. This suggests that either the reaction kinetics favor the formation of the 'a' isomer or that the 'b' isomer is more susceptible to degradation or remains in the mother liquor during crystallization. In the final hydrolysis step, the oxime group is cleaved under acidic conditions (using sulfuric or hydrochloric acid) to release the ketone byproduct and generate the hydroxylamine salt. This acidolysis is highly selective, breaking the N-O bond while leaving the newly formed ether linkage intact. The ability to control these mechanistic pathways allows manufacturers to achieve isomer ratios exceeding 99:1, which is critical for the biological activity of the final herbicide active ingredient.
How to Synthesize O-Phenoxyalkylhydroxylamine Efficiently
Implementing this synthesis requires precise control over stoichiometry and temperature to maximize yield and purity. The process begins with the activation of the hydroxy-oxime, followed by etherification with the chosen phenol, and concludes with acidic hydrolysis and neutralization. Each step has been optimized in the patent examples to demonstrate robustness on a kilogram scale, ensuring that the transition from laboratory bench to pilot plant is seamless. Operators must pay close attention to the molar ratios of base to substrate, particularly during the sulfonation step where 1 to 1.5 equivalents of base are preferred to scavenge the generated acid without promoting side reactions. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- React O-(2-hydroxyethyl)-oximes with a sulphonyl halogenide in the presence of a base to form a sulphonate mixture.
- React the resulting sulphonate mixture with a phenol derivative in the presence of a base to generate O-phenoxyalkyloximes.
- Hydrolyze the oxime mixture in the presence of an acid to cleave the oxime group and form hydroxylamine salts.
- Liberate the free O-phenoxyalkylhydroxylamines from the salts using a mineral base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates directly into enhanced operational efficiency and risk mitigation. By eliminating the need for specialized protecting group reagents, the bill of materials is simplified, reducing dependency on niche suppliers who might face availability constraints. The use of commodity chemicals such as methanesulfonyl chloride, triethylamine, and potassium hydroxide ensures that raw material sourcing remains stable and cost-effective even during market fluctuations. Furthermore, the reduction in synthetic steps shortens the overall production cycle time, allowing for faster turnaround on customer orders and improved inventory turnover rates. This agility is a key component of being a reliable agrochemical intermediate supplier in a volatile global market.
- Cost Reduction in Manufacturing: The most significant financial benefit arises from the complete removal of the N-hydroxyphthalimide protection strategy. In traditional routes, the cost of the protecting agent and the reagents required for its removal (hydrazine or similar) constitutes a major portion of the variable cost. By bypassing these steps, the new process drastically lowers the cost of goods sold (COGS). Additionally, the higher overall yield means that less starting material is required to produce the same amount of final product, further amplifying the economic advantage. The elimination of phthalic acid waste also reduces wastewater treatment costs, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available reagents and solvents. Unlike complex chiral catalysts or custom-synthesized protecting groups, the inputs for this process—toluene, methanol, inorganic bases—are produced globally in massive quantities. This ubiquity minimizes the risk of supply disruptions due to geopolitical issues or single-source vendor failures. Moreover, the process tolerance described in the patent suggests that strict anhydrous conditions are not always mandatory, simplifying logistics and storage requirements for raw materials. This flexibility ensures continuous production capability even when facing minor logistical hurdles.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the viability of this route on a multi-kilogram scale, proving its readiness for commercial scale-up of complex agrochemical intermediates. The reaction conditions operate at moderate temperatures and pressures, reducing the energy load on the facility and minimizing safety risks associated with high-pressure reactors. From an environmental perspective, the atom economy is superior to the protected route, generating less solid waste. The ability to recover and recycle excess acid from the hydrolysis step further aligns with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations without sacrificing productivity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis pathway. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for process evaluation. Understanding these nuances is vital for technical teams assessing the feasibility of integrating this chemistry into existing production lines.
Q: How does this process improve isomer purity compared to conventional methods?
A: The process described in CN1150418A inherently reduces the content of the unwanted 'b' isomer during the sulfonation and etherification steps. By starting with a mixture where the 'a' isomer is dominant (e.g., 70-100%), the final product can achieve an isomer ratio as high as 99.4:0.6, significantly reducing the need for difficult downstream purification.
Q: What are the economic advantages of avoiding N-hydroxyphthalimide protection?
A: Conventional methods often rely on N-hydroxyphthalimide as a protecting group, which introduces additional synthetic steps for installation and removal. This new route eliminates the protecting group entirely, thereby removing the cost of phthalic acid derivatives and avoiding the yield losses typically associated with deprotection steps.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent provides preparation examples demonstrating kilogram-scale synthesis (e.g., using 57.3 kg of starting oxime). The use of common solvents like toluene and standard inorganic bases like potassium hydroxide indicates that the process is robust and suitable for commercial scale-up of complex agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Phenoxyalkylhydroxylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of O-phenoxyalkylhydroxylamine meets the exacting standards required for downstream herbicide synthesis. Our commitment to quality assurance means that you can rely on us for high-purity agrochemical intermediates that drive the efficacy of your final products.
We invite you to engage with our technical procurement team to discuss how this patented technology can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient manufacturing route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive agrochemical sector.
