Advanced Synthesis of Triketone Bis(Oxime Ether) Derivatives for Crop Protection Intermediates
The chemical landscape of modern agrochemical development relies heavily on the availability of high-purity, structurally precise intermediates. Patent CN1321144A introduces a transformative methodology for the preparation of triketone bis(oxime ether) derivatives, specifically addressing the synthetic challenges associated with Formula I compounds. These molecules serve as critical precursors for the crop protection agents detailed in WO-A97/15552, representing a significant value chain in the agricultural sector. The core innovation lies in overcoming the historical difficulties of regioselectivity and stereoselectivity that have plagued direct oximation routes. By implementing a strategic ketal protection group on the central carbonyl, this process enables a directed synthesis that bypasses the formation of complex cyclic by-products such as N-oxidized oxadiazoles and isoxazoles. 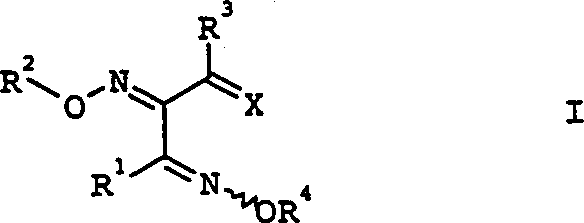 For procurement specialists and R&D directors alike, understanding this patented pathway is essential for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality. The ability to synthesize these derivatives without extensive purification steps translates directly into supply chain stability and cost efficiency. As we delve deeper into the technical specifics, it becomes clear that this method is not merely a laboratory curiosity but a robust industrial protocol designed for scalability.
For procurement specialists and R&D directors alike, understanding this patented pathway is essential for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality. The ability to synthesize these derivatives without extensive purification steps translates directly into supply chain stability and cost efficiency. As we delve deeper into the technical specifics, it becomes clear that this method is not merely a laboratory curiosity but a robust industrial protocol designed for scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ortho-triketone derivatives has been fraught with chemical inefficiencies that hinder commercial viability. Prior art literature, including early publications in Gazzetta Chimica Italiana, indicates that direct reaction of triketones with hydroxylamine results in a chaotic mixture of products. Instead of the desired linear oxime structures, the reaction tends to undergo intramolecular cyclization, yielding thermodynamically stable but undesired heterocyclic compounds like isoxazoles. Furthermore, even when linear products are formed, the lack of steric differentiation between the substituents on the central carbon atom leads to a non-selective formation of stereoisomers. This results in a complex matrix of E and Z isomers that are notoriously difficult to separate using standard crystallization or chromatography techniques. For a manufacturing entity, this implies low overall yields, high solvent consumption for purification, and significant batch-to-batch variability, all of which are detrimental to cost reduction in crop protection manufacturing.
The Novel Approach
The methodology disclosed in CN1321144A fundamentally alters the reaction trajectory by introducing a temporary masking group. By converting the central carbonyl of the 1,3-diketone precursor into a ketal (Formula III), the reactivity of the central position is effectively neutralized. This allows the subsequent oximation steps to occur exclusively at the terminal positions, ensuring regioselectivity. 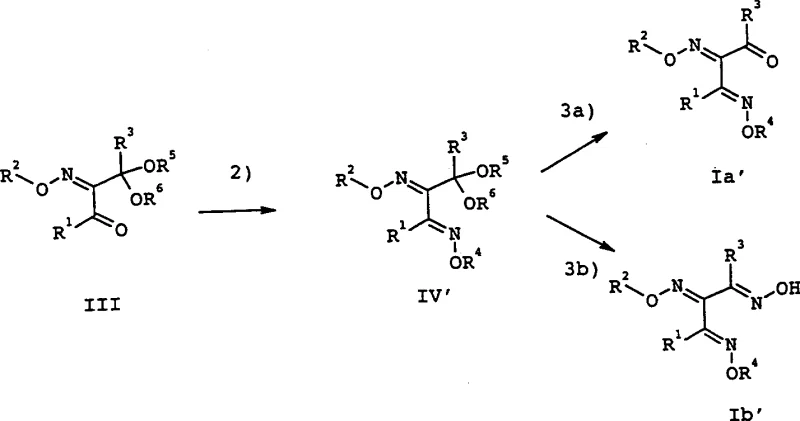 Once the side chains are functionalized to form the bis-oxime ether ketal (Formula IV), the protecting group can be cleanly removed under acidic conditions to reveal the target triketone structure. This stepwise approach prevents the formation of cyclic by-products entirely and offers superior control over the stereochemical outcome. The process transforms a previously unpredictable reaction into a deterministic sequence, enabling the production of isomerically pure materials suitable for high-performance agricultural applications without the need for resource-intensive separation protocols.
Once the side chains are functionalized to form the bis-oxime ether ketal (Formula IV), the protecting group can be cleanly removed under acidic conditions to reveal the target triketone structure. This stepwise approach prevents the formation of cyclic by-products entirely and offers superior control over the stereochemical outcome. The process transforms a previously unpredictable reaction into a deterministic sequence, enabling the production of isomerically pure materials suitable for high-performance agricultural applications without the need for resource-intensive separation protocols.
Mechanistic Insights into Ketal-Protection Directed Synthesis
The success of this synthetic route hinges on the precise manipulation of carbonyl reactivity through acid-catalyzed ketalization. In the first stage, the 1,3-diketone (Formula II) reacts with an alcohol or diol in the presence of a catalytic amount of a strong acid, such as p-toluenesulfonic acid or sulfuric acid. The mechanism involves the protonation of the central carbonyl oxygen, making it more electrophilic and susceptible to nucleophilic attack by the alcohol. 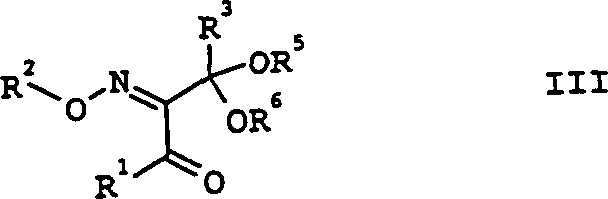 Crucially, the reaction equilibrium is driven forward by the continuous removal of water, either through azeotropic distillation using entrainers like toluene or cyclohexane, or by employing chemical dehydrating agents like trimethyl orthoformate. The choice of diol, such as ethylene glycol or neopentyl glycol, influences the steric environment of the intermediate, which subsequently impacts the stereoselectivity of the final deprotection step. This careful engineering of the intermediate ensures that the molecule remains stable during the harsh conditions of the subsequent oximation reactions.
Crucially, the reaction equilibrium is driven forward by the continuous removal of water, either through azeotropic distillation using entrainers like toluene or cyclohexane, or by employing chemical dehydrating agents like trimethyl orthoformate. The choice of diol, such as ethylene glycol or neopentyl glycol, influences the steric environment of the intermediate, which subsequently impacts the stereoselectivity of the final deprotection step. This careful engineering of the intermediate ensures that the molecule remains stable during the harsh conditions of the subsequent oximation reactions.
Following the formation of the ketal, the oximation step proceeds with high fidelity. The use of alkoxyamines (R4O-NH2) or hydroxylamine allows for the conversion of the remaining ketone groups into oxime ethers. The final deprotection step involves hydrolysis in an acidic medium, typically at a pH of 0.5 to 1.5. 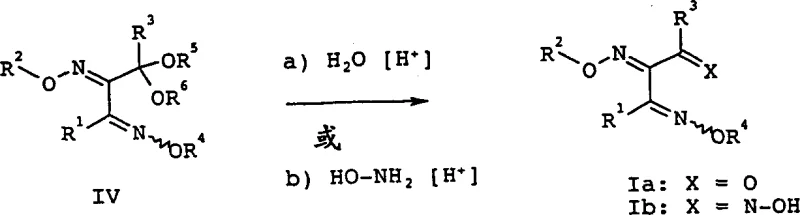 Interestingly, the patent data reveals that the temperature during this hydrolysis acts as a switch for stereocontrol. Conducting the hydrolysis at lower temperatures (20-40°C) favors the formation of the E,E-isomer or the biologically relevant E,Z,E-isomer. Conversely, higher temperatures can shift the equilibrium towards Z-isomers. This mechanistic nuance provides process chemists with a valuable lever to tune the impurity profile and maximize the yield of the desired active isomer, thereby enhancing the overall purity specifications of the final API intermediate.
Interestingly, the patent data reveals that the temperature during this hydrolysis acts as a switch for stereocontrol. Conducting the hydrolysis at lower temperatures (20-40°C) favors the formation of the E,E-isomer or the biologically relevant E,Z,E-isomer. Conversely, higher temperatures can shift the equilibrium towards Z-isomers. This mechanistic nuance provides process chemists with a valuable lever to tune the impurity profile and maximize the yield of the desired active isomer, thereby enhancing the overall purity specifications of the final API intermediate.
How to Synthesize Triketone Bis(Oxime Ether) Efficiently
The synthesis of these complex intermediates requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process is divided into three distinct operational phases: protection, functionalization, and deprotection. Each phase demands specific control over pH, temperature, and stoichiometry to prevent side reactions. For example, the initial ketalization must be monitored closely to ensure complete conversion before proceeding, as residual diketone can lead to impurities in the final step. Similarly, the work-up procedures involving extraction with non-polar solvents like methyl tert-butyl ether are critical for removing inorganic salts and acid residues. Detailed standard operating procedures regarding reagent addition rates and quenching protocols are essential for scaling this chemistry from the bench to the plant floor.
- Protect the central carbonyl group of the 1,3-diketone precursor by reacting it with an alcohol or diol in the presence of an acid catalyst to form a ketal intermediate.
- Perform directed oximation on the remaining ketone groups using alkoxyamines or hydroxylamine followed by alkylation to generate the bis-oxime ether ketal.
- Cleave the ketal protecting group under acidic conditions, optionally followed by amination with hydroxylamine, to yield the final triketone bis(oxime ether) or tris(oxime) derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ketal-protection strategy offers substantial advantages over traditional synthesis routes, particularly regarding cost structure and supply reliability. The primary economic driver is the drastic simplification of the purification process. By eliminating the formation of cyclic by-products and minimizing isomer mixtures, the need for expensive preparative chromatography or multiple recrystallization steps is significantly reduced. This streamlining of downstream processing directly lowers the cost of goods sold (COGS) and reduces solvent waste, aligning with modern green chemistry initiatives. Furthermore, the use of commodity chemicals such as simple diols and alkoxyamines ensures that raw material sourcing remains stable and unaffected by niche supply bottlenecks.
- Cost Reduction in Manufacturing: The elimination of complex separation processes translates to significant operational savings. Traditional methods often require extensive solvent usage and energy-intensive distillation to isolate the correct isomer from a complex mixture. In contrast, this directed synthesis produces a cleaner crude product, reducing the load on purification units. Additionally, the potential for one-pot sequences, where the solvent from the ketalization step is carried over into the oximation, further minimizes solvent exchange costs and processing time. These efficiencies compound to offer a much more competitive price point for high-purity agrochemical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to higher batch success rates and consistent output. Because the chemistry avoids unstable intermediates and sensitive reagents, the risk of batch failure due to minor parameter deviations is minimized. This reliability is crucial for maintaining continuous supply lines to downstream formulation plants. Moreover, the scalability of the acid-catalyzed steps means that production can be easily ramped up from pilot scale to multi-ton commercial batches without requiring specialized equipment, ensuring that lead times for high-purity agrochemical intermediates remain short and predictable.
- Scalability and Environmental Compliance: The process utilizes standard unit operations familiar to any fine chemical manufacturing facility, such as stirred tank reactors and liquid-liquid extraction columns. This ease of scale-up facilitates the commercial scale-up of complex agrochemical intermediates without the need for capital-intensive new infrastructure. From an environmental standpoint, the reduction in solvent volume and the avoidance of heavy metal catalysts (often used in alternative coupling reactions) simplify waste treatment protocols. The aqueous waste streams are primarily acidic or saline, which are easier to neutralize and treat compared to organic-heavy waste streams containing complex heterocyclic impurities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing supply chains. The answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Why is the ketal protection strategy superior to direct triketone oximation?
A: Direct reaction of triketones with hydroxylamine often yields complex mixtures of cyclic by-products like N-oxidized oxadiazoles and isoxazoles, along with difficult-to-separate stereoisomers. The ketal protection method described in CN1321144A blocks the central reactive site, allowing for regioselective functionalization of the side chains and preventing unwanted cyclization.
Q: Can this process control the stereochemistry of the final product?
A: Yes, the process allows for the preferential formation of specific isomers. For instance, hydrolysis at lower temperatures (20-40°C) favors the E,E-isomer or E,Z,E-isomer, whereas higher temperatures (>40°C) with certain ketals can increase the proportion of Z-isomers, offering tunable selectivity based on process conditions.
Q: What are the typical reaction conditions for the ketal formation step?
A: The ketalization is typically conducted in the presence of a Brönstedt acid like p-toluenesulfonic acid or sulfuric acid at temperatures ranging from 60°C to 110°C. Water removal via azeotropic distillation with toluene or cyclohexane, or using dehydrating agents like trimethyl orthoformate, is critical to drive the equilibrium towards the ketal product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triketone Bis(Oxime Ether) Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation crop protection solutions. Our technical team has extensively analyzed the pathways described in CN1321144A and possesses the expertise to execute this ketal-protection strategy with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical or field trials to full-scale market launch. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed isomer ratio analysis via HPLC and NMR, guaranteeing that every batch meets the exacting standards required for agrochemical registration.
We invite you to collaborate with us to optimize your supply chain for these vital intermediates. By leveraging our process engineering capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to chemical excellence and supply chain integrity can support your long-term strategic goals in the global agrochemical market.
