Advanced Manufacturing of Alpha-Difluoromethylornithine Intermediates for Global Pharmaceutical Supply Chains
Advanced Manufacturing of Alpha-Difluoromethylornithine Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective synthetic routes for critical active pharmaceutical ingredients (APIs) and their intermediates. Patent CN1564806A introduces a transformative methodology for the preparation of α-difluoromethylornithine (DFMO), also known as Eflornithine, which has gained significant traction for dermatological applications including the treatment of unwanted facial hair. This technical insight report analyzes the novel synthetic pathway disclosed in the patent, highlighting its potential to redefine supply chain dynamics for pharmaceutical intermediates. By shifting away from expensive natural amino acid precursors towards readily available synthetic building blocks, this process addresses key pain points regarding raw material volatility and process safety. The innovation lies not just in the chemical transformations but in the holistic design that facilitates commercial scale-up of complex pharmaceutical intermediates while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of DFMO has been plagued by significant economic and safety hurdles that hinder efficient large-scale production. Traditional routes, such as those disclosed in US Patent No. 4,309,442, rely heavily on ornithine as the starting material. Ornithine is a relatively high-cost amino acid, and its price volatility directly impacts the cost structure of the final API, making margin optimization difficult for procurement teams. Furthermore, alternative methods involving malonate and acrylonitrile often necessitate the use of the Hofmann reaction. The Hofmann rearrangement is notoriously difficult to control on an industrial scale due to the risk of exothermic runaway, posing severe safety liabilities for manufacturing facilities. Additionally, many legacy processes depend on halogenated solvents which require expensive waste disposal protocols and rigorous environmental monitoring, further eroding the economic viability of these older synthetic strategies.
The Novel Approach
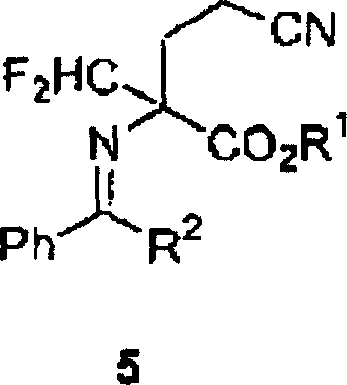
The methodology presented in CN1564806A offers a compelling alternative by utilizing glycine alkyl esters as the foundational building block. These starting materials are abundant, inexpensive, and chemically stable, providing a solid foundation for cost reduction in pharmaceutical intermediates manufacturing. The core of this innovation involves the formation of a Schiff base protected intermediate, followed by a Michael addition and a critical difluoromethylation step. This sequence allows for the precise installation of the difluoromethyl group without the need for hazardous rearrangement reactions. Moreover, the process is designed to minimize or eliminate the use of halogenated solvents, favoring greener alternatives like acetonitrile and ethers, which simplifies solvent recovery and waste management. This strategic shift not only enhances operational safety but also aligns with modern environmental compliance standards, making it an attractive option for forward-thinking supply chain leaders.
Mechanistic Insights into Difluoromethylation and Selective Reduction
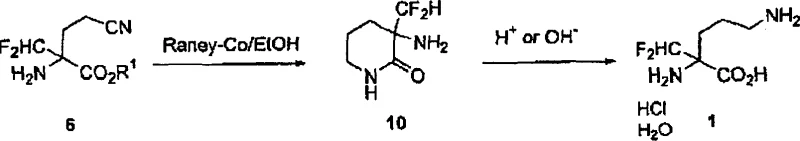
From a technical perspective, the success of this synthesis hinges on the efficient introduction of the difluoromethyl moiety and the subsequent selective reduction of the nitrile group. The difluoromethylation step involves the deprotonation of the α-position of the cyano-butyrates using strong bases such as sodium tert-butoxide or sodium ethoxide. This generates a reactive carbanion which then attacks chlorodifluoromethane (Freon-22), a widely available industrial gas. The reaction conditions are carefully optimized, typically operating between -35°C and 25°C for deprotonation and slightly higher temperatures for the alkylation, ensuring high conversion rates often exceeding 98%. The use of a Schiff base protecting group during this stage is crucial as it prevents side reactions at the amino group, thereby maintaining the integrity of the molecular scaffold throughout the harsh alkylation conditions.
Following the construction of the carbon backbone, the process requires the selective reduction of the nitrile functionality to a primary amine without affecting the ester group prematurely. This is achieved using heterogeneous transition metal catalysts, specifically platinum on carbon (Pt/C), under hydrogen pressure. The presence of acid, such as hydrochloric acid, in the reaction mixture aids in the formation of the amine salt, which helps drive the equilibrium and prevents over-reduction or side reactions. Alternatively, the patent describes the use of Raney cobalt catalysts which can facilitate the cyclization to a lactam intermediate before final hydrolysis. This mechanistic flexibility allows manufacturers to choose the catalytic system that best fits their existing infrastructure, whether they prefer direct hydrogenation to the diamino ester or a lactam-mediated pathway, both leading to the high-purity target molecule after final hydrolysis.
How to Synthesize Alpha-Difluoromethylornithine Efficiently
Implementing this synthesis requires a disciplined approach to reaction control and purification to maximize yield and purity. The process begins with the condensation of glycine ester and benzaldehyde, followed by the Michael addition of acrylonitrile. The subsequent difluoromethylation is the key value-adding step, requiring precise temperature control and gas handling capabilities. Once the protected difluoromethyl intermediate is obtained, the final stages involve hydrolysis of the Schiff base and ester groups, coupled with the reduction of the nitrile. Detailed standard operating procedures for each unit operation, including specific stoichiometry, temperature ramps, and workup protocols, are essential for reproducibility. For the complete standardized synthesis steps and technical parameters, please refer to the guide below.
- Condense glycine ester with benzaldehyde to form a Schiff base protected intermediate.
- Perform Michael addition with acrylonitrile followed by difluoromethylation using chlorodifluoromethane.
- Hydrolyze the protecting group and reduce the nitrile moiety to yield the final diamino acid structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the decoupling of production costs from the volatile pricing of natural amino acids like ornithine. By switching to synthetic glycine esters, manufacturers can secure a more predictable and lower cost base for their raw materials. This stability is crucial for long-term contract negotiations and budget forecasting. Furthermore, the elimination of dangerous reagents and the reduction of halogenated solvent usage significantly lower the operational overhead associated with safety compliance and environmental waste disposal. These factors combine to create a more resilient and cost-efficient supply chain capable of withstanding market fluctuations.
- Cost Reduction in Manufacturing: The substitution of expensive ornithine with commodity-grade glycine esters fundamentally alters the cost of goods sold. Since glycine esters are produced on a massive scale for various industries, their pricing is stable and competitive. Additionally, the avoidance of complex and hazardous rearrangement reactions reduces the need for specialized, high-maintenance reactor equipment. The simplified downstream processing, characterized by straightforward crystallization and filtration steps, minimizes solvent consumption and energy usage. These cumulative efficiencies translate into substantial cost savings that can be passed down the supply chain or retained as improved margins.
- Enhanced Supply Chain Reliability: Relying on widely available petrochemical-derived starting materials rather than fermentation-derived amino acids reduces the risk of supply disruptions. Glycine and acrylonitrile are produced by multiple global suppliers, ensuring a diversified sourcing strategy that mitigates single-point failures. The robustness of the chemical steps, which do not rely on sensitive biocatalysts or unstable reagents, further ensures consistent batch-to-batch production. This reliability is paramount for maintaining continuous inventory levels and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as hydrogenation and alkylation that are standard in fine chemical manufacturing. The ability to run these reactions in common solvents like ethanol or THF simplifies the technology transfer from pilot plant to commercial production. Moreover, the reduced environmental footprint, achieved by minimizing halogenated waste and avoiding explosive reaction conditions, facilitates easier regulatory approval and permitting. This alignment with green chemistry principles future-proofs the manufacturing asset against tightening environmental regulations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding safety, purity, and scalability based on the patent data. These insights are derived directly from the experimental examples and summary sections of the intellectual property, providing a factual basis for decision-making. We encourage technical teams to review these points when assessing the feasibility of integrating this route into their existing production portfolios.
Q: How does this new DFMO synthesis method improve safety compared to traditional routes?
A: Traditional methods often rely on the Hofmann reaction which can be uncontrollable and explosive. This novel route utilizes standard hydrogenation and alkylation conditions, significantly mitigating safety risks associated with runaway reactions.
Q: What are the cost advantages of using glycine esters as starting materials?
A: Glycine esters are commercially available, inexpensive bulk chemicals compared to ornithine. This shift in raw material sourcing drastically reduces the baseline cost of goods sold for the final API intermediate.
Q: Can this process achieve the high purity required for pharmaceutical applications?
A: Yes, the patent demonstrates that through careful control of hydrolysis and crystallization steps, DFMO purity can exceed 98%, meeting stringent regulatory standards for active pharmaceutical ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Difluoromethylornithine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of advanced pharmaceutical intermediates like DFMO. Our team of expert chemists has extensively analyzed the pathways described in CN1564806A and possesses the technical capability to execute this synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the high standards required for downstream API manufacturing.
We invite you to collaborate with us to optimize your supply chain for alpha-difluoromethylornithine and related intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable partner dedicated to driving efficiency and innovation in your pharmaceutical manufacturing operations.
