Scalable Synthesis of Substituted Imidazole Derivatives for Cognitive Disorder Therapeutics
Scalable Synthesis of Substituted Imidazole Derivatives for Cognitive Disorder Therapeutics
The pharmaceutical landscape for treating cognitive disorders continues to evolve, driven by the demand for highly selective alpha-2 adrenoceptor antagonists. Patent CN100339368C introduces a robust and innovative synthetic pathway for preparing substituted imidazole derivatives of Formula (I), which serve as critical intermediates for these therapeutic agents. Unlike previous methodologies that struggled with yield and purity, this invention outlines a comprehensive strategy starting from readily available indanone precursors. The core innovation lies in the strategic construction of the imidazole ring through a sequence involving controlled halogenation, thiocyanate-mediated cyclization, and subsequent catalytic refinement. This approach not only enhances the chemical efficiency but also addresses the scalability challenges inherent in producing complex heterocyclic scaffolds.
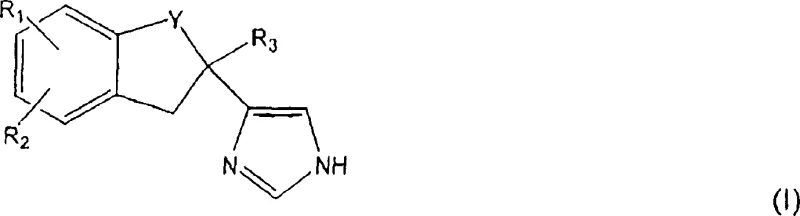
These compounds, characterized by their specific substitution patterns on the indane or tetralin rings, exhibit excellent oral bioavailability and long-acting antagonistic properties. The versatility of the synthetic route allows for the introduction of various functional groups, such as halogens and hydroxyls, at precise positions on the aromatic ring. This flexibility is paramount for medicinal chemists aiming to optimize the pharmacokinetic profiles of drug candidates. By establishing a reliable supply chain for these high-purity intermediates, manufacturers can significantly accelerate the development timeline for next-generation cognitive enhancers and neuroprotective agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Formula (I) compounds has been fraught with significant technical hurdles that impede commercial viability. Prior art, such as that disclosed in EP0618906B1, primarily focused on modifying the benzene ring portion of the indane scaffold through various substitution routes. However, these methods failed to provide a total synthesis capable of delivering the target compounds in high yields, often resulting in complex mixtures that were difficult to separate. Furthermore, another prominent method described in EP0310745B1 relied heavily on the use of formamide to construct the imidazole ring in the final step. While chemically feasible on a small scale, the use of formamide necessitates extremely harsh reaction conditions, typically involving high temperatures that promote degradation and the formation of numerous by-products.
The reliance on such aggressive conditions creates a bottleneck for industrial manufacturing. The generation of substantial impurities during the formamide cyclization step mandates rigorous and costly purification processes, such as extensive chromatography or multiple recrystallizations, which drastically reduce the overall mass balance. Additionally, the handling of formamide at elevated temperatures poses safety and environmental concerns, making it less desirable for large-scale production facilities adhering to modern green chemistry principles. Consequently, there has been a persistent need for a synthetic alternative that avoids these pitfalls while maintaining structural integrity and stereochemical control.
The Novel Approach
The process detailed in CN100339368C represents a paradigm shift by decoupling the ring formation from harsh thermal conditions. Instead of forcing the cyclization with formamide, this novel route employs a stepwise assembly beginning with the alpha-halogenation of a ketone precursor (Formula II). This activation step is performed under mild conditions, utilizing bromine in a methanolic solvent system at temperatures ranging from -8°C to +25°C. This controlled environment ensures high selectivity for the mono-brominated species, setting the stage for a clean subsequent transformation. The avoidance of extreme heat at this early stage preserves the integrity of sensitive functional groups on the indane ring.
Following halogenation, the synthesis proceeds through a condensation reaction with a protected amine and an alkali metal thiocyanate. This cyclization step effectively constructs the imidazole core under reflux conditions in alcohol solvents, which are far more manageable than molten formamide. The resulting mercapto-imidazole intermediate (Formula IV) is then subjected to a two-stage refinement process: desulfurization using Raney Nickel and deprotection of the nitrogen atom. This modular approach allows for the isolation and characterization of stable intermediates, providing multiple quality control checkpoints that are absent in one-pot formamide methods. The result is a process that is not only higher yielding but also inherently safer and more adaptable to kilogram and tonne-scale manufacturing.
Mechanistic Insights into Halogenation and Thiocyanate-Mediated Cyclization
The success of this synthetic route hinges on the precise execution of the initial activation and cyclization steps. The mechanism begins with the electrophilic alpha-halogenation of the acetyl-indan derivative. In the presence of a protic solvent like methanol, molecular bromine generates an enol or enolate equivalent which attacks the bromine species. The low temperature control (-8°C to -5°C) is critical here to suppress poly-halogenation, a common side reaction that would render the intermediate useless for the subsequent nucleophilic substitution. The resulting alpha-bromo ketone (Formula III) is a highly reactive electrophile, primed for attack by nitrogen and sulfur nucleophiles.
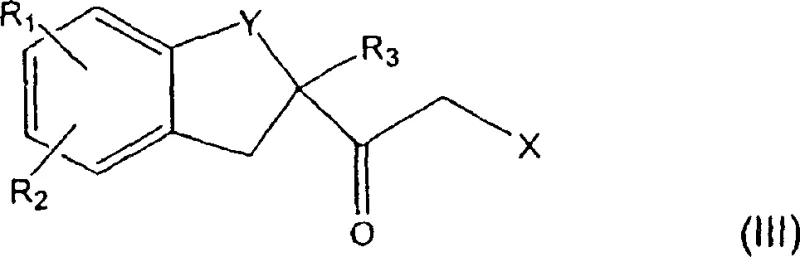
In the subsequent cyclization phase, the alpha-bromo ketone reacts with a primary amine (where the nitrogen is protected by a removable group like benzyl) and potassium thiocyanate. The mechanism likely involves the initial displacement of the bromide by the sulfur of the thiocyanate or the nitrogen of the amine, followed by an intramolecular cyclization to close the five-membered imidazole ring. The use of a protected amine is a strategic masterstroke; it prevents polymerization and ensures that the imidazole ring forms with the correct regiochemistry. The sulfur atom, initially part of the thiocyanate, ends up as a mercapto group at the 2-position of the imidazole, which serves as a temporary handle for further manipulation. This intermediate stability is key to the process's robustness, allowing the reaction to proceed to completion without the rapid decomposition seen in formamide routes.
Impurity control is inherently built into this mechanism. By avoiding the high-energy environment of formamide cyclization, the formation of fused ring by-products or degraded aromatic systems is minimized. The desulfurization step, utilizing Raney Nickel at moderate temperatures (40°C to 60°C), cleanly removes the thiol group via hydrogenolysis, leaving behind the unsubstituted imidazole carbon. Finally, the removal of the nitrogen protecting group, achievable via catalytic hydrogenation with Pd/C or transfer hydrogenation with ammonium formate, yields the final free base. Each step is designed to be chemoselective, ensuring that the fluorine or other halogen substituents on the indane ring remain untouched throughout the synthesis.
How to Synthesize 4-(2-Ethyl-5-Fluoro-Indan-2-Yl)-1H-Imidazole Efficiently
The practical implementation of this chemistry requires careful attention to reagent stoichiometry and thermal management, particularly during the exothermic halogenation phase. Operators must ensure that the bromine addition is slow and that the cooling capacity is sufficient to maintain the sub-zero temperature window. Following the workup of the bromo-ketone, the cyclization step benefits from the use of dry alcohol solvents to prevent hydrolysis of the reactive intermediate. The subsequent catalytic steps require strict exclusion of air to prevent catalyst poisoning and ensure efficient hydrogen transfer. For a detailed breakdown of the specific operational parameters, reagent grades, and workup procedures validated by the patent examples, please refer to the standardized synthesis guide below.
- Halogenate the starting ketone (Formula II) using bromine in methanol at low temperatures (-8°C to 25°C) to form the alpha-bromo ketone (Formula III).
- React the alpha-bromo ketone with a protected amine (e.g., benzylamine) and potassium thiocyanate in refluxing alcohol to form the mercapto-imidazole intermediate (Formula IV).
- Remove the mercapto group using Raney Nickel catalyst, followed by removal of the protecting group (e.g., benzyl) via hydrogenation or transfer hydrogenation to yield the final imidazole (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for specialized high-temperature formamide processing equipment, facilities can utilize standard glass-lined or stainless steel reactors equipped with standard reflux condensers. This compatibility with existing infrastructure reduces capital expenditure requirements for scaling up production. Furthermore, the reagents employed—bromine, potassium thiocyanate, benzylamine, and common hydrogenation catalysts—are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source bottlenecks that often plague specialty reagent procurement.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the significant improvement in yield and the reduction in purification costs. Conventional methods often suffer from low mass recovery due to the formation of intractable tars and impurities during harsh cyclization. In contrast, the milder conditions of this new route preserve the carbon skeleton, leading to higher crude yields. Additionally, the ability to use crude intermediates directly in subsequent steps, as noted in the patent examples, eliminates the need for expensive and time-consuming chromatographic purifications at every stage. This streamlining of the workflow translates directly into lower cost of goods sold (COGS) and improved margin potential for the final API.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the robustness of the reaction conditions. The tolerance for slight variations in temperature and the use of stable intermediates mean that batch-to-batch consistency is easier to achieve. In a commercial setting, this reduces the rate of batch failures and the need for re-processing, which are major disruptors to supply continuity. Moreover, the avoidance of hazardous high-pressure or high-temperature operations simplifies regulatory compliance and safety audits, ensuring that production schedules are not delayed by safety incidents or regulatory hold-ups. The process is designed for continuity, supporting a steady flow of material from pilot plant to commercial manufacturing.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route aligns well with modern sustainability goals. The solvents used, primarily alcohols like methanol and ethanol, are easier to recover and recycle compared to the complex waste streams generated by formamide degradation. The catalytic nature of the final steps (using Raney Nickel or Pd/C) minimizes the generation of stoichiometric metal waste. As production scales from kilograms to tonnes, the ability to efficiently recycle solvents and catalysts becomes a critical factor in maintaining environmental compliance and reducing waste disposal costs. This green chemistry profile makes the process attractive for manufacturing in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline candidates.
Q: Why is this new process superior to the formamide method described in EP0310745B1?
A: The conventional formamide method requires harsh reaction conditions that often lead to the formation of significant impurities, complicating purification. The novel process disclosed in CN100339368C utilizes milder conditions, specifically avoiding high-temperature formamide cyclization, which results in a cleaner reaction profile and higher overall yields suitable for industrial scale-up.
Q: What are the critical temperature controls required for the halogenation step?
A: Precision temperature control is vital during the initial halogenation of the indanone derivative. The patent specifies a range of -8°C to +25°C, with a preferred window of -8°C to -5°C. Maintaining these low temperatures prevents over-halogenation and side reactions, ensuring the formation of the desired alpha-bromo ketone intermediate with high selectivity.
Q: How is the final deprotection of the imidazole nitrogen achieved?
A: The removal of the protecting group (such as a benzyl group) from the nitrogen atom can be achieved through catalytic hydrogenation using Pd/C or Raney Nickel. Alternatively, transfer hydrogenation using ammonium formate in the presence of a palladium catalyst offers a safe and effective route to generate the free NH-imidazole without requiring high-pressure hydrogen gas equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(2-Ethyl-5-Fluoro-Indan-2-Yl)-1H-Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of cognitive disorder therapeutics depends on the availability of high-quality, scalable intermediates. Our technical team has extensively analyzed the pathway described in CN100339368C and possesses the expertise to execute this chemistry with precision. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facility is equipped with state-of-the-art hydrogenation reactors and cryogenic cooling systems necessary to manage the specific temperature requirements of the halogenation and cyclization steps, guaranteeing adherence to stringent purity specifications.
We invite you to collaborate with us to optimize this synthesis for your specific project needs. Our rigorous QC labs are prepared to validate the identity and purity of every batch against your specific COA data requirements. Whether you require custom route feasibility assessments or immediate scale-up of this imidazole derivative, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis and discover how our manufacturing capabilities can support your journey from clinical trials to market launch.
