Scalable Synthesis of High-Purity Quercetin-3-O-Acyl Esters for Advanced Drug Development
Scalable Synthesis of High-Purity Quercetin-3-O-Acyl Esters for Advanced Drug Development
The pharmaceutical industry continuously seeks novel derivatives of natural flavonoids to overcome limitations such as poor bioavailability and rapid metabolic clearance. Patent CN102659735A introduces a robust and scalable chemical synthesis method for Quercetin-3-O-acyl esters, utilizing inexpensive Rutin as the starting material. This innovative approach addresses the critical need for stable, high-potency flavonoid intermediates capable of exhibiting superior antitumor activity against cell lines such as EC9706, PC-3, and MGC-803. By employing a strategic sequence of benzylation, acid hydrolysis, regioselective esterification, and catalytic hydrogenolysis, the process achieves excellent selectivity and yield under mild conditions. The resulting compounds represent a significant advancement over native quercetin, offering enhanced pharmacokinetic profiles suitable for next-generation therapeutic development.
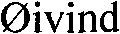
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the modification of quercetin has relied heavily on enzymatic catalysis or non-selective chemical reactions that often suffer from significant drawbacks in an industrial setting. Enzymatic methods, while selective, are frequently constrained by the high cost of biocatalysts, limited substrate tolerance, and difficulties in scaling up to multi-kilogram production volumes. Furthermore, previous chemical attempts to modify quercetin often lacked regioselectivity, leading to complex mixtures of poly-substituted products that are difficult and expensive to purify. Some prior art methods involved harsh reaction conditions that could degrade the sensitive flavonoid backbone, reducing overall yield and compromising the quality of the final pharmaceutical intermediate. These inefficiencies create bottlenecks in the supply chain, driving up costs and extending lead times for drug developers seeking reliable sources of modified flavonoids.
The Novel Approach
The methodology described in patent CN102659735A circumvents these traditional hurdles by implementing a highly efficient four-step chemical synthesis that prioritizes both selectivity and scalability. By starting with Rutin, a widely available and cost-effective natural glycoside, the process leverages a protection-deprotection strategy that ensures the acyl group is installed exclusively at the 3-hydroxyl position. The use of standard organic reagents such as benzyl chloride, dicyclohexylcarbodiimide (DCC), and palladium on carbon (Pd-C) allows for precise control over reaction parameters without the need for specialized biological equipment. This chemical route not only simplifies the purification process through crystallization but also ensures that the final product maintains the structural integrity of the flavonoid core. Consequently, this approach offers a commercially viable pathway for producing high-purity Quercetin-3-O-acyl esters that meet the stringent quality standards required for clinical applications.
Mechanistic Insights into Regioselective Esterification and Deprotection
The core of this synthesis lies in its sophisticated manipulation of the flavonoid hydroxyl groups to achieve perfect regioselectivity. The process begins with the selective benzylation of the 7, 3', and 4' hydroxyl groups of Rutin using benzyl chloride and anhydrous potassium carbonate in DMF. This step is crucial as it masks the reactive sites that could otherwise compete during the subsequent esterification, effectively directing the incoming acyl group solely to the 3-position. Following this protection, the rutinose sugar moiety is cleaved via acid-catalyzed hydrolysis in an ethanol solution containing hydrochloric acid. This hydrolysis step is carefully controlled to prevent degradation of the aglycone, yielding tribenzyl quercetin as a stable intermediate ready for functionalization.
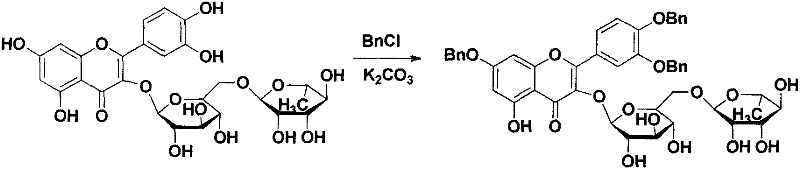
Once the tribenzyl quercetin intermediate is obtained, the critical esterification step is performed using DCC and DMAP in dichloromethane. The carbodiimide coupling agent activates the carboxylic acid, forming an O-acylisourea intermediate which is then attacked by the nucleophilic 3-hydroxyl group of the flavonoid. The presence of DMAP acts as a potent nucleophilic catalyst, accelerating the acylation rate and ensuring high conversion even with sterically hindered acids. This mechanism allows for the introduction of a diverse range of acyl groups, from short-chain fatty acids to complex aromatic moieties, providing a versatile platform for structure-activity relationship studies. The robustness of this coupling reaction ensures consistent yields across different substrates, making it ideal for generating libraries of derivatives.
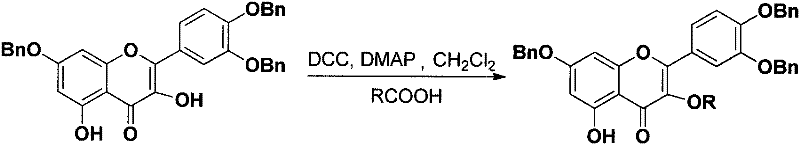
The final stage of the synthesis involves the removal of the benzyl protecting groups via catalytic hydrogenolysis. Using Pd-C as the catalyst under hydrogen pressure, the benzyl ethers are cleanly cleaved to regenerate the free phenolic hydroxyl groups at the 7, 3', and 4' positions without affecting the newly formed ester bond at the 3-position. This chemoselective deprotection is vital for restoring the antioxidant capacity of the B-ring while maintaining the metabolic stability conferred by the 3-acyl modification. The use of heterogeneous catalysis facilitates easy removal of the catalyst by filtration, simplifying downstream processing and minimizing metal contamination in the final product. This comprehensive mechanistic approach guarantees a high-purity end product suitable for sensitive biological assays and pharmaceutical formulation.
How to Synthesize Quercetin-3-O-Acyl Esters Efficiently
Implementing this synthesis route requires careful attention to reaction stoichiometry and purification techniques to maximize yield and purity. The process is designed to be operationally simple, utilizing common laboratory glassware and standard workup procedures such as aqueous washing and recrystallization. Detailed protocols for each step, including specific solvent ratios, temperature controls, and reaction times, are essential for reproducing the high yields reported in the patent data. For large-scale manufacturing, parameters such as hydrogen pressure and catalyst loading must be optimized to ensure safety and efficiency. The following guide outlines the standardized operational framework derived from the patent examples to assist process chemists in scaling this technology.
- Protect the 7, 3', and 4' hydroxyl groups of Rutin using benzyl chloride and potassium carbonate to form tribenzyl rutin.
- Perform acid-catalyzed hydrolysis in ethanol using hydrochloric acid to remove the rutinose sugar moiety, yielding tribenzyl quercetin.
- Conduct regioselective esterification at the 3-position using DCC and DMAP with the desired fatty or aromatic acid.
- Execute catalytic hydrogenolysis using Pd-C under hydrogen pressure to remove benzyl protecting groups and obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers substantial advantages by leveraging commodity chemicals and eliminating reliance on volatile biological reagents. The primary cost driver in flavonoid modification is often the starting material and the complexity of purification; however, by utilizing Rutin, which is abundantly sourced from agricultural byproducts, the raw material costs are significantly minimized. Furthermore, the avoidance of expensive enzymes removes a major variable from the supply chain, ensuring consistent availability of reagents regardless of biological harvest cycles. The use of standard organic solvents and catalysts means that production can be easily transferred between different manufacturing sites without requiring specialized fermentation infrastructure, thereby enhancing supply chain resilience and reducing logistical risks.
- Cost Reduction in Manufacturing: The elimination of enzymatic catalysts and the use of inexpensive protecting groups like benzyl chloride drastically reduce the bill of materials for this synthesis. Traditional enzymatic routes often require costly downstream processing to remove proteins and biological impurities, whereas this chemical method allows for straightforward crystallization and filtration. Additionally, the high regioselectivity minimizes the formation of isomeric byproducts, reducing the waste associated with extensive chromatographic purification. This efficiency translates directly into lower production costs per kilogram, making the final Quercetin-3-O-acyl esters more economically viable for inclusion in broad-spectrum therapeutic formulations.
- Enhanced Supply Chain Reliability: Sourcing stability is critical for long-term drug development projects, and this route relies entirely on petrochemical-derived reagents and plant-extracted Rutin, both of which have mature and stable global supply chains. Unlike specialized biocatalysts that may have single-source suppliers and long lead times, reagents such as DCC, DMAP, and Pd-C are commoditized and readily available from multiple vendors worldwide. This diversification of supply sources mitigates the risk of production stoppages due to vendor shortages. Moreover, the robustness of the chemical steps ensures that minor variations in reagent quality do not compromise the final product, allowing for greater flexibility in procurement strategies.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind, utilizing reaction conditions that are easily managed in large reactors. The solvent systems employed, primarily dichloromethane and ethanol, are well-understood in terms of recovery and recycling, facilitating compliance with environmental regulations regarding volatile organic compound emissions. The heterogeneous nature of the final deprotection step allows for the recovery and reuse of the palladium catalyst, further reducing waste and environmental impact. This alignment with green chemistry principles not only satisfies regulatory requirements but also appeals to environmentally conscious stakeholders in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Quercetin-3-O-acyl esters. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating these intermediates into your specific drug development pipeline.
Q: Why is the 3-OH position specifically targeted for esterification in this synthesis?
A: The 3-OH position is targeted because metabolic degradation of quercetin often occurs at the 3-OH and 7-OH sites. Modifying the 3-OH position with acyl groups significantly improves bioavailability and antitumor activity compared to unmodified quercetin, as demonstrated in pharmacological screening.
Q: What are the advantages of this chemical route over enzymatic methods?
A: Unlike enzymatic methods which require expensive enzymes and have strict reaction conditions, this chemical synthesis utilizes cost-effective reagents like benzyl chloride and DCC. It offers better scalability, milder reaction conditions, and higher yields suitable for industrial production.
Q: How does this synthesis ensure high purity for pharmaceutical use?
A: The process employs a robust protection-deprotection strategy using benzyl groups which are stable during esterification but easily removed via catalytic hydrogenolysis. This ensures high regioselectivity and minimizes byproduct formation, resulting in a high-purity final intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quercetin-3-O-Acyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Quercetin-3-O-acyl ester synthesis route in developing next-generation antitumor agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of flavonoid chemistry, including rigorous moisture control for coupling reactions and specialized hydrogenation suites for safe deprotection. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to optimize your supply chain for these high-value compounds. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By partnering with us, you gain access to our deep expertise in process optimization and regulatory compliance. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your upcoming projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
