Advanced One-Step Synthesis of 2-Nitroglycal Alkenes for Commercial Glycosylation Applications
Advanced One-Step Synthesis of 2-Nitroglycal Alkenes for Commercial Glycosylation Applications
The landscape of carbohydrate chemistry is constantly evolving to meet the rigorous demands of modern glycoprotein and therapeutic synthesis, where the availability of high-quality building blocks is paramount. Patent CN113200951A introduces a groundbreaking methodology for the efficient synthesis of 2-nitroglycal alkenes, a class of compounds that serves as versatile intermediates in the construction of complex glycoconjugates. This innovation addresses long-standing challenges in generating conjugated nitroolefin and enol ether moieties, which are critical for downstream transformations such as Michael additions and Diels-Alder reactions. By leveraging a novel TBAN-Tf2O-DTBMP catalytic system, the disclosed method achieves high efficiency in a single synthetic step, significantly streamlining the production workflow for these valuable sugar derivatives.
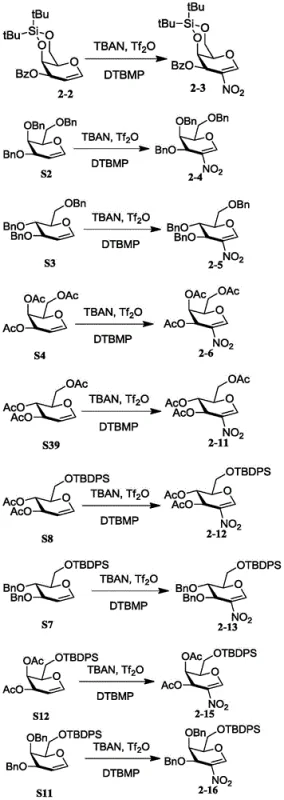
For research and development teams focused on glycosyl donor synthesis, the ability to access 2-nitrosugars with diverse protecting group patterns is essential for tuning reactivity and selectivity in subsequent glycosylation events. The patent details a robust protocol that accommodates a wide array of substrates, including those protected with acetyl, benzyl, benzoyl, and bulky silyl groups like TBDPS and TIPS. This versatility ensures that chemists can tailor the electronic and steric properties of the glycal scaffold to suit specific synthetic targets, whether they are developing new antibiotics, anticancer agents, or vaccine candidates. The technical depth of this disclosure provides a reliable foundation for scaling up the production of these critical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-nitroglycals has been plagued by significant inefficiencies and substrate limitations that hindered their widespread adoption in industrial settings. Traditional approaches often relied on harsh nitrating agents or multi-step sequences that resulted in poor yields and extensive byproduct formation, particularly when dealing with sterically demanding protecting groups. For instance, earlier attempts using acetyl nitrate generated from nitric acid and acetic anhydride at low temperatures frequently failed to eliminate properly to form the desired double bond, leading to complex mixtures that were difficult to purify. Furthermore, methods employing silver nitrate and acetyl chloride often struggled with substrates containing silicon-based protecting groups, as the strong electron-donating nature and steric bulk of silyl ethers interfered with the elimination step necessary to generate the nitroolefin functionality.
These conventional limitations created a bottleneck in the supply chain for advanced glycosylation building blocks, forcing manufacturers to rely on costly and time-consuming purification processes or to abandon promising synthetic routes altogether. The inability to effectively nitrate and eliminate silyl-protected glycals meant that chemists were restricted to a narrower range of substrates, limiting the structural diversity of the final glycoconjugates. This lack of flexibility was particularly problematic in the synthesis of proteobacterial glycoantigens and other complex targets where specific protection patterns are required to achieve the desired stereochemical outcomes. Consequently, there was an urgent need for a more reactive and tolerant system that could overcome these steric and electronic barriers.
The Novel Approach
The inventive method described in the patent overcomes these historical hurdles by utilizing a highly reactive TBAN-Tf2O-DTBMP system that facilitates the in situ generation of nitronium trifluoroacetate species. This powerful electrophile reacts rapidly with the saccharide alkene to form a 2-nitro-1-(trifluoroacetyl) pyranose intermediate, which subsequently undergoes elimination in the presence of the base DTBMP to yield the target 2-nitroglycene. Unlike previous systems that faltered with silyl groups, this new approach successfully navigates the steric hindrance associated with bulky protecting groups, enabling the efficient synthesis of a broad spectrum of 2-nitrosugars. The reaction conditions are meticulously optimized, typically operating at temperatures between -70°C and -30°C in dry dichloromethane, ensuring high selectivity and minimal degradation of sensitive functional groups.
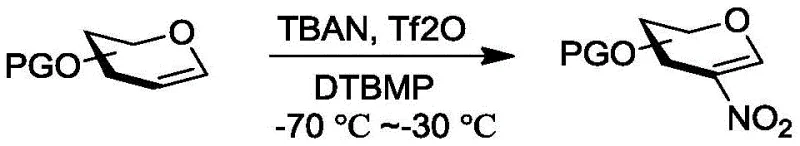
This streamlined one-pot process not only simplifies the operational complexity but also dramatically improves the overall atom economy and throughput of the synthesis. The patent demonstrates the efficacy of this method across various substrates, including glucals, galactals, and even disaccharides, achieving yields that are substantially higher than those reported for legacy methods. For example, substrates protected with tert-butyl diphenyl silyl (TBDPS) groups, which were previously problematic, are now converted to their corresponding 2-nitro derivatives with impressive efficiency. This breakthrough represents a significant leap forward in carbohydrate chemistry, providing a reliable and scalable route for producing high-purity intermediates that are essential for the next generation of glycan-based therapeutics.
Mechanistic Insights into TBAN-Tf2O-DTBMP Catalyzed Nitration
The core of this technological advancement lies in the unique mechanistic pathway enabled by the combination of tetrabutylammonium nitrate (TBAN), trifluoroacetic anhydride (Tf2O), and 2,6-di-tert-butyl-4-methylpyridine (DTBMP). Upon mixing, TBAN and Tf2O react to generate a highly electrophilic nitronium ion species, likely stabilized as a trifluoroacetyl nitrate complex, which attacks the electron-rich double bond of the glycal substrate. This initial addition forms a transient cationic intermediate that is rapidly trapped to create the 2-nitro-1-trifluoroacetyl species. The presence of DTBMP, a non-nucleophilic base with significant steric bulk, is crucial for the subsequent elimination step; it abstracts the proton at the C-1 position without interfering with the sensitive nitro group or the protecting groups on the sugar ring. This precise balance of reactivity ensures that the elimination proceeds smoothly to restore the double bond, resulting in the conjugated 2-nitroglycal alkene structure.
Impurity control is inherently built into this mechanism through the careful selection of reaction parameters and reagents. The use of dry dichloromethane and molecular sieves minimizes hydrolysis side reactions that could degrade the trifluoroacetyl intermediate or the final product. Furthermore, the low temperature range (-70°C to -30°C) suppresses competing pathways such as polymerization or over-nitration, which are common pitfalls in traditional nitration chemistry. The patent data reveals that optimizing the molar ratios of the reagents is key to maximizing yield; for instance, increasing the equivalents of TBAN and Tf2O to 2.0 and 4.0 respectively, along with 4.0 equivalents of DTBMP, was found to boost yields for sterically hindered substrates like compound 2-7 up to 75.5%. This level of mechanistic understanding allows for fine-tuning the process to accommodate different substrate classes, ensuring consistent quality and purity profiles.
How to Synthesize 2-Nitroglycal Alkene Efficiently
The practical implementation of this synthesis route is designed for reproducibility and ease of operation, making it suitable for both laboratory-scale discovery and pilot-scale production. The process begins with the preparation of a reaction mixture containing the glycal substrate, TBAN, and DTBMP in anhydrous DCM, activated by the addition of molecular sieves to maintain strict anhydrous conditions. After stirring under nitrogen to ensure homogeneity and activation, the system is cooled to the optimal temperature window, typically starting at -70°C, before the slow addition of Tf2O initiates the nitration cascade. Reaction progress is monitored via thin-layer chromatography (TLC), allowing operators to quench the reaction precisely upon completion to prevent over-reaction or decomposition. The detailed standardized synthesis steps for specific compounds are outlined in the guide below.
- Mix the glycal substrate, TBAN, and DTBMP in dry DCM with molecular sieves under nitrogen protection.
- Cool the reaction system to between -70°C and -30°C and add Tf2O dropwise to initiate nitration.
- Monitor by TLC, then quench, filter, wash, and purify via column chromatography to obtain the target 2-nitroglycal.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis method offers substantial strategic advantages by addressing key pain points associated with the sourcing of complex carbohydrate intermediates. The one-pot nature of the reaction eliminates the need for isolating unstable intermediates, thereby reducing the number of unit operations and the associated labor and equipment costs. This simplification of the workflow translates directly into shorter manufacturing cycles and reduced lead times, enabling suppliers to respond more agilely to fluctuating market demands. Furthermore, the high yields reported across a diverse range of substrates indicate a robust process with excellent atom economy, minimizing raw material waste and lowering the overall cost of goods sold (COGS) for the final active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of commercially available reagents like TBAN and Tf2O significantly lowers the input costs compared to precious metal-catalyzed alternatives. Additionally, the high efficiency of the reaction reduces the burden on downstream purification processes, as fewer byproducts are generated, leading to substantial savings in solvent usage and chromatography media. This economic efficiency is further enhanced by the ability to recycle solvents and recover unreacted starting materials, creating a more sustainable and cost-effective production model.
- Enhanced Supply Chain Reliability: By utilizing a synthetic route that tolerates a wide variety of protecting groups, manufacturers are not constrained by the availability of specific niche substrates, thereby diversifying the supply base and mitigating risks associated with single-source dependencies. The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and avoiding costly production delays. This reliability fosters stronger partnerships between chemical suppliers and pharmaceutical companies, ensuring a steady flow of high-quality intermediates for critical drug development programs.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful application to disaccharide substrates, indicating its potential for large-scale commercial production without significant re-engineering. The use of standard organic solvents and the absence of heavy metals simplify waste treatment protocols, aligning with increasingly stringent environmental regulations and corporate sustainability goals. This alignment not only reduces the environmental footprint of the manufacturing process but also future-proofs the supply chain against evolving regulatory landscapes regarding chemical safety and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2-nitroglycal alkenes, based on the detailed experimental data provided in the patent documentation. These insights are intended to clarify the operational parameters and potential benefits for stakeholders evaluating this technology for integration into their existing workflows. Understanding these nuances is vital for making informed decisions about process adoption and resource allocation.
Q: Why is the TBAN-Tf2O-DTBMP system superior for silyl-protected glycals?
A: Conventional nitration methods often fail with bulky silyl protecting groups due to steric hindrance and strong electron donation. This novel system generates a highly reactive nitronium species in situ that successfully overcomes these barriers, enabling efficient elimination to form the nitroolefin moiety without degrading the sensitive silicon-based protections.
Q: What are the typical yields for this synthesis method?
A: The patent data indicates robust yields across various substrates. For example, glucal derivatives protected with benzyl or acetyl groups achieve yields ranging from 58% to 71.6%, while more complex silyl-protected substrates can reach up to 75.5% yield under optimized conditions.
Q: Is this method scalable for industrial production?
A: Yes, the process utilizes common organic solvents like DCM and standard reagents (TBAN, Tf2O). The reaction times are short (often minutes to hours), and the workup involves standard filtration and chromatography, making it highly amenable to commercial scale-up for producing complex carbohydrate intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nitroglycal Alkene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development pipelines, and we are committed to delivering excellence in every batch we produce. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every shipment of 2-nitroglycal alkene meets the highest industry standards, providing you with the confidence needed to advance your projects.
We invite you to engage with our technical procurement team to discuss how our advanced synthesis capabilities can support your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our optimized manufacturing processes. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the synthesis of complex pharmaceutical intermediates.
