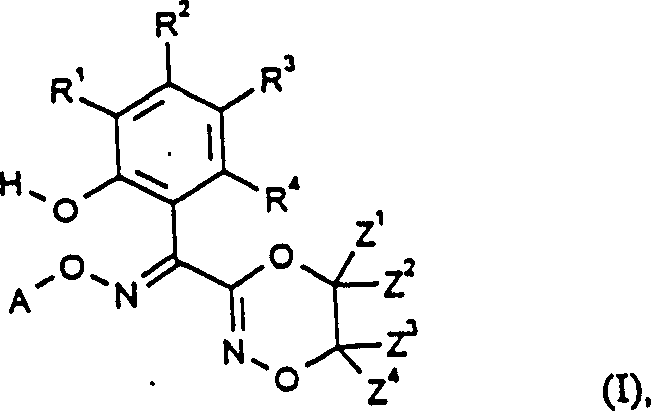
Advanced Manufacturing of 3-(1-Hydroxyphenyl-1-Alkoxyiminomethyl)Dioxazines for Global Supply Chains
Introduction to Advanced Dioxazine Manufacturing
The pharmaceutical and agrochemical industries continuously demand more efficient pathways for synthesizing complex heterocyclic intermediates, particularly those serving as precursors for antifungal agents. Patent CN1082955C introduces a groundbreaking methodology for the preparation of 3-(1-hydroxyphenyl-1-alkoxyiminomethyl)dioxazines, addressing critical bottlenecks in existing manufacturing technologies. This innovation leverages a novel rearrangement of O-hydroxyethyl-O'-alkyl-benzofurandione dioximes to achieve superior yields and purity profiles. By circumventing the cumbersome protection-deprotection sequences characteristic of older methodologies, this process offers a robust platform for the commercial scale-up of complex pharmaceutical intermediates. The technical depth of this patent provides a clear roadmap for optimizing reaction conditions, solvent systems, and catalytic environments to ensure consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art techniques, such as those disclosed in WO 95-04728, rely on a linear synthesis starting from hydroxyphenyl acetates that necessitates extensive functional group manipulation. A typical conventional route involves protecting the phenolic hydroxyl group as a tetrahydropyranyl ether, followed by nitrosation, alkylation, and subsequent reaction with hydroxylamine. This multi-step sequence not only increases the consumption of reagents and solvents but also introduces significant opportunities for yield loss at each stage. Furthermore, the final deprotection step requires acidic conditions that can compromise the stability of sensitive oxime functionalities. The cumulative effect of these inefficiencies results in a process that is economically disadvantageous for large-scale production, with occasional low yields impacting the overall cost ratio and supply reliability for downstream drug manufacturers.
The Novel Approach
In stark contrast, the methodology described in CN1082955C streamlines the synthesis by utilizing a direct rearrangement strategy that bypasses the need for temporary protecting groups. The core innovation involves the transformation of O-hydroxyethyl-O'-alkyl-benzofurandione dioximes into the target dioxazine ring system under basic conditions. This approach allows for the use of readily available starting materials like omega-nitro-2-hydroxyacetophenones or benzofuranones, which react efficiently with alkoxyamines. The process is characterized by its operational simplicity, often proceeding in aqueous or alcoholic media at moderate temperatures ranging from 0°C to 100°C. By eliminating several isolation and purification steps, this novel route drastically reduces waste generation and processing time, thereby enhancing the overall sustainability and economic viability of producing these high-value antifungal intermediates.
Mechanistic Insights into Base-Catalyzed Rearrangement and Cyclization
The heart of this technological advancement lies in the unique reactivity of the benzofurandione dioxime intermediates under basic conditions. When treated with bases such as sodium hydroxide or potassium carbonate in aqueous or alcoholic solutions, the O-hydroxyethyl-O'-alkyl-benzofurandione dioxime undergoes a surprising and efficient rearrangement. Unlike traditional expectations where such compounds might decompose into salicylic acid derivatives, this specific substrate architecture facilitates an intramolecular cyclization to form the 1,4,2-dioxazine ring. The mechanism likely involves the deprotonation of the hydroxyl group followed by nucleophilic attack on the adjacent oxime carbon, driven by the release of ring strain and the formation of a stable aromatic system. This rearrangement is highly selective, minimizing the formation of by-products and ensuring that the crude product obtained after simple filtration possesses high purity, often exceeding 90% as verified by HPLC analysis.
Impurity control is inherently built into the reaction design through the careful selection of reaction parameters and the stability of the intermediates. The patent highlights that even in the presence of water, the cyclization proceeds cleanly, which is unexpected for moisture-sensitive oxime chemistry. Furthermore, the process includes a dedicated isomerization step where the initial mixture of E and Z stereoisomers is converted predominantly into the thermodynamically stable E-isomer using acid catalysis. This ensures that the final product meets stringent stereochemical specifications required for biological activity. The use of common solvents like ethyl acetate, methanol, or water further simplifies the workup procedure, allowing for the removal of inorganic salts and minor organic impurities through straightforward crystallization or extraction techniques without the need for complex chromatographic separations.
How to Synthesize 3-(1-Hydroxyphenyl-1-Alkoxyiminomethyl)Dioxazines Efficiently
The synthesis protocol outlined in the patent provides a detailed framework for executing this transformation with high reproducibility. The process typically begins with the formation of the oxime intermediate from the corresponding ketone, followed by cyclization to the benzofurandione system. Subsequent reaction with ethylene oxide introduces the necessary hydroxyethyl side chain, which then triggers the final rearrangement to the dioxazine core. Operators must pay close attention to temperature control during the exothermic addition of ethylene oxide and the pH adjustment during the final acidification step to maximize yield. The detailed standardized synthesis steps见下方的指南 ensure that laboratory-scale success can be translated effectively to pilot and commercial production environments.
- React omega-nitro-2-hydroxyacetophenone with alkoxyamine to form the corresponding oxime intermediate.
- Perform base-catalyzed cyclization to generate the benzofurandione dioxime derivative.
- Execute rearrangement using ethylene oxide and base, followed by acid-catalyzed isomerization to obtain the final E-isomer dioxazine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic benefits regarding cost structure and vendor reliability. The elimination of expensive protecting group reagents and the reduction in total unit operations directly lower the manufacturing cost base, allowing for more competitive pricing in the global market. Additionally, the use of commodity chemicals like ethylene oxide and simple inorganic bases reduces dependency on specialized or scarce raw materials, thereby mitigating supply chain risks associated with raw material volatility. The robustness of the reaction conditions, which tolerate the presence of water and proceed at moderate temperatures, further enhances process safety and reduces energy consumption, contributing to a lower carbon footprint and improved environmental compliance.
- Cost Reduction in Manufacturing: The streamlined synthetic route eliminates multiple isolation and purification stages, significantly reducing solvent usage and labor hours per kilogram of product. By avoiding the use of costly tetrahydropyranyl protecting groups and hazardous alkylating agents like dibromoethane found in older methods, the raw material costs are substantially optimized. This efficiency gain allows manufacturers to offer high-purity intermediates at a more attractive price point, improving the margin structure for downstream pharmaceutical formulations without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as omega-nitro-2-hydroxyacetophenones and benzofuranones ensures a stable supply base that is less susceptible to geopolitical or logistical disruptions. The simplified process flow reduces the lead time required for batch production, enabling faster response to market demand fluctuations. Furthermore, the high yields reported in the examples, such as 91.3% for the cyclization step and 67.7% for the final rearrangement, guarantee consistent output volumes, which is critical for maintaining uninterrupted supply to major drug manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory glassware to industrial reactors. The ability to perform key steps in aqueous media minimizes the generation of hazardous organic waste, simplifying effluent treatment and reducing disposal costs. The high selectivity of the rearrangement reaction reduces the burden on downstream purification systems, aligning with modern green chemistry principles and regulatory requirements for sustainable manufacturing practices in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on process capabilities and product specifications. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of the new synthesis route over conventional methods?
A: The new process significantly reduces the number of synthetic steps compared to the prior art (WO 95-04728), eliminating the need for protecting group strategies like tetrahydropyranyl ethers. This results in higher overall yields, with specific steps achieving over 90% conversion, and simplifies purification protocols.
Q: How is stereochemical purity controlled in this manufacturing process?
A: The process initially produces a mixture of stereoisomers, but includes a dedicated isomerization step using acid catalysis (e.g., hydrochloric acid in ethyl acetate) to convert the mixture into the thermodynamically stable E-isomer with high purity, often exceeding 94% as confirmed by HPLC analysis.
Q: Are the starting materials commercially scalable?
A: Yes, the route utilizes readily available starting materials such as omega-nitro-2-hydroxyacetophenone, benzofuranones, and ethylene oxide. These commodities are accessible in bulk quantities, ensuring supply chain stability and reducing raw material costs for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(1-Hydroxyphenyl-1-Alkoxyiminomethyl)Dioxazines Supplier
At NINGBO INNO PHARMCHEM, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this advanced synthesis technology to the global market. Our state-of-the-art facilities are equipped to handle the specific reaction conditions required for this process, including the safe handling of ethylene oxide and precise temperature control for the rearrangement steps. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 3-(1-hydroxyphenyl-1-alkoxyiminomethyl)dioxazines meets the highest international standards for pharmaceutical intermediates. Our commitment to quality assurance guarantees that our clients receive products with consistent stereochemical profiles and minimal impurity levels.
We invite potential partners to engage with our technical procurement team to discuss how this innovative manufacturing route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient process. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition to a more reliable and cost-effective source of critical antifungal intermediates.
