Advanced Catalytic Process for High-Purity Thiazole Derivatives and Commercial Scalability
Advanced Catalytic Process for High-Purity Thiazole Derivatives and Commercial Scalability
The global demand for high-efficiency neonicotinoid insecticides has driven significant innovation in the synthesis of their key heterocyclic precursors. Patent CN1447800A introduces a groundbreaking catalytic process for the preparation of thiazole derivatives, specifically targeting the production of compounds like 2-chloro-5-chloromethyl-thiazole, which serves as a critical building block for active pharmaceutical ingredients and agrochemicals such as thiamethoxam and clothianidin. This technology represents a paradigm shift from traditional stoichiometric chlorination methods to a more sustainable, catalytic approach utilizing sulfur dioxide. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic advantages of this patent is essential for securing supply chains that prioritize both purity and environmental compliance. The invention addresses long-standing issues regarding yield optimization, thermal safety, and waste management that have historically plagued the industrial manufacturing of these valuable insecticides.
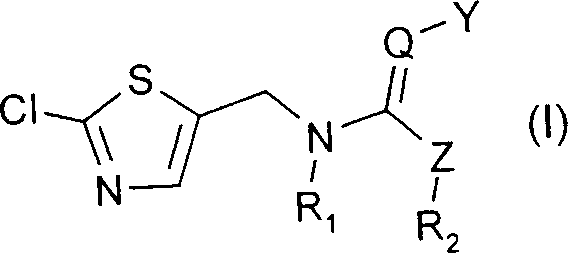
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in EP-A-446913, relied heavily on either direct chlorination with chlorine gas or the use of sulfuryl chloride (SO2Cl2) as a stoichiometric reagent. While the use of SO2Cl2 offered improved yields compared to chlorine alone, it introduced severe downstream processing challenges. Specifically, the stoichiometric consumption of SO2Cl2 generates equivalent molar amounts of sulfur dioxide byproduct, which often requires oxidation to sulfate for disposal. These sulfate wastes are ecologically harmless in isolation but are highly corrosive to the concrete infrastructure of wastewater purification systems, leading to substantial maintenance costs and regulatory hurdles. Furthermore, batch processes involving these reagents often resulted in the accumulation of thermally unstable intermediates, creating a hazardous thermal potential that could lead to serious safety accidents if not meticulously controlled. The crude yields in these conventional methods typically hovered around 73% to 82%, leaving significant room for improvement in volumetric efficiency and raw material utilization.
The Novel Approach
The methodology outlined in CN1447800A revolutionizes this synthesis by employing catalytic amounts of sulfur dioxide (ranging from 1 mol% to 50 mol%) in the presence of a chlorinating agent. This catalytic cycle allows for the efficient conversion of formula (II) compounds into the desired formula (III) intermediates without the burden of stoichiometric waste generation. By utilizing a continuous flow reaction setup, the process ensures that only minimal amounts of reactive intermediates accumulate at any given time, drastically mitigating the risk of thermal runaway. Experimental data from the patent demonstrates that this approach can achieve theoretical yields exceeding 90%, a significant leap from the historical baselines. This novel pathway not only enhances the economic viability of the production but also aligns with modern green chemistry principles by minimizing toxic waste streams and improving the overall safety profile of the manufacturing plant.

Mechanistic Insights into SO2-Catalyzed Cyclization
The core innovation lies in the catalytic role of sulfur dioxide during the chlorination and cyclization steps. In the absence of the catalyst, the reaction between 2-chloro-3-isocyanothio-1-propene and chlorine proceeds sluggishly and with poor selectivity. The introduction of SO2 or SO2Cl2 facilitates the formation of reactive chlorosulfite intermediates that accelerate the ring closure to form the thiazole nucleus. Mechanistically, the catalyst likely activates the chlorinating species, allowing for a more controlled electrophilic attack on the substrate. This controlled reactivity is crucial for suppressing side reactions that lead to poly-chlorinated impurities or polymeric byproducts. The patent specifies that the reaction can be conducted in polar aprotic solvents like acetonitrile or chlorobenzene, with temperatures carefully maintained between 20°C and 50°C to balance reaction rate and selectivity. The ability to tune the catalyst loading between 10 mol% and 40 mol% provides process engineers with a robust handle to optimize kinetics for different reactor scales.
Following the synthesis of the crude intermediate, the patent emphasizes a purification strategy that avoids the pitfalls of traditional distillation. Since 2-chloro-5-chloromethyl-thiazole is thermally sensitive and possesses a 'thermal memory' where stability decreases with heat stress, high-temperature distillation is detrimental. Instead, the process utilizes melt crystallization, often at temperatures as low as 0°C to 25°C. This technique leverages the differences in melting points between the product and impurities, allowing for the isolation of the target compound with purities exceeding 98% without exposing the molecule to degrading thermal conditions. This gentle purification method is critical for maintaining the integrity of the intermediate before it undergoes subsequent coupling reactions to form the final insecticide active ingredient.
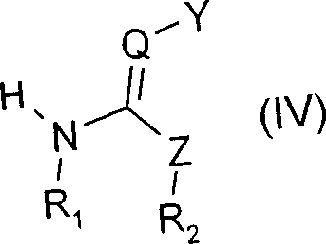
How to Synthesize 2-Chloro-5-chloromethyl-thiazole Efficiently
Implementing this catalytic process requires precise control over reagent addition and thermal management to replicate the high yields reported in the patent examples. The synthesis begins with the dissolution of the starting isocyanothio-propene derivative in a suitable solvent, followed by the introduction of the sulfur dioxide catalyst. Chlorine gas is then introduced subsurface over a controlled period to manage the exotherm. The reaction mixture is subsequently heated to drive the cyclization to completion. For the highest efficiency, the patent recommends a continuous loop reactor configuration where reagents are fed continuously, and the product stream is harvested steadily. This approach maximizes space-time yield and ensures consistent product quality. The detailed standardized synthesis steps for replicating this high-purity route are provided in the guide below.
- Prepare the reaction vessel with 2-chloro-3-isocyanothio-1-propene dissolved in a polar aprotic solvent such as acetonitrile or chlorobenzene.
- Introduce catalytic amounts of sulfur dioxide (1-50 mol%) or sulfuryl chloride into the mixture at temperatures between 20°C and 30°C.
- Slowly add chlorine gas while maintaining temperature control, followed by heating to 50°C to complete the cyclization and isolate the product via melt crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalytic technology offers profound strategic benefits beyond simple yield improvements. The elimination of stoichiometric sulfuryl chloride usage directly translates to a reduction in raw material costs and a simplification of the waste treatment infrastructure. By avoiding the generation of large volumes of sulfate waste, manufacturers can significantly reduce the frequency of costly repairs to wastewater treatment facilities caused by chemical corrosion. This operational reliability ensures uninterrupted production schedules, which is vital for meeting the demanding delivery timelines of the global agrochemical market. Furthermore, the implementation of continuous processing technologies enhances the scalability of the operation, allowing for flexible production volumes that can respond rapidly to market fluctuations without the need for massive capital investment in new batch reactors.
- Cost Reduction in Manufacturing: The shift from stoichiometric to catalytic reagent usage fundamentally alters the cost structure of the synthesis. By utilizing catalytic amounts of SO2, the process eliminates the need to purchase and dispose of large quantities of sulfuryl chloride. This reduction in reagent consumption lowers the direct variable costs per kilogram of product. Additionally, the higher yields achieved (over 90% versus the historical 73%) mean that less raw material is wasted, further driving down the cost of goods sold. The avoidance of complex waste neutralization steps for sulfate byproducts also reduces utility and labor costs associated with environmental compliance, resulting in substantial overall cost savings for the manufacturing entity.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain continuity. The conventional batch methods carried inherent risks of thermal runaway due to intermediate accumulation, which could lead to plant shutdowns or regulatory inspections following incidents. The new continuous flow method mitigates these risks by maintaining low inventory of reactive species within the reactor at any one time. This inherent safety design reduces the likelihood of unplanned downtime caused by safety incidents. Moreover, the robustness of the catalytic system allows for consistent production of high-purity intermediates, reducing the rejection rates of batches and ensuring a steady flow of material to downstream formulation plants, thereby strengthening the reliability of the entire supply network.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing continuous reactors that can be easily numbered up or scaled out to meet increasing demand. Unlike batch processes that face mixing and heat transfer limitations at large scales, continuous flow systems maintain consistent reaction parameters regardless of total volume. Environmentally, the process aligns with stringent global regulations by minimizing the release of toxic sulfur compounds and eliminating the generation of corrosive sulfate waste streams. This proactive approach to environmental stewardship future-proofs the manufacturing site against tightening ecological laws, ensuring long-term operational viability and reducing the risk of fines or shutdowns related to non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic thiazole synthesis. These insights are derived directly from the experimental data and claims within patent CN1447800A, providing clarity on how this technology resolves historical pain points in insecticide intermediate manufacturing. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What is the primary advantage of using catalytic SO2 over stoichiometric SO2Cl2?
A: Using catalytic SO2 eliminates the generation of stoichiometric amounts of sulfate waste, which is known to cause severe chemical erosion to wastewater purification concrete structures, thereby reducing environmental compliance costs.
Q: How does the continuous flow method improve safety in this synthesis?
A: Continuous processing prevents the accumulation of thermally unstable intermediates that occur in batch reactions, significantly lowering the hazardous thermal potential and risk of runaway exotherms.
Q: Why is melt crystallization preferred over distillation for purification?
A: Melt crystallization operates at lower temperatures, avoiding the thermal stress that causes decomposition in thermally sensitive thiazole compounds, while also eliminating the need for additional solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-chloromethyl-thiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the production of high-value agrochemical intermediates. Our technical team has extensively analyzed the catalytic processes described in CN1447800A and possesses the expertise to implement these advanced methodologies at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to full-scale manufacturing is seamless and efficient. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 2-chloro-5-chloromethyl-thiazole meets the exacting standards required for the synthesis of next-generation insecticides like thiamethoxam.
We invite global partners to collaborate with us to leverage these technological advancements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the highest levels of quality, safety, and economic efficiency available in the market.
