Advanced C-Cl Bond Activation Technology for High-Purity Pharmaceutical Intermediates Manufacturing
Introduction to Next-Generation C-Cl Activation Technology
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the need for more atom-economical and cost-effective processes. Patent CN101693723A introduces a breakthrough class of organic phosphonium salts, specifically meta-trialkoxyphenyl dialkylphosphine tetrafluoroborate, which addresses the longstanding challenge of activating inert carbon-chlorine bonds. Traditionally, the reliance on expensive and less atom-economical aryl bromides and iodides has been a bottleneck in the manufacturing of complex pharmaceutical intermediates. This novel technology leverages a uniquely structured phosphine ligand that coordinates with palladium catalysts to achieve high-selectivity activation of sp2 carbon-chlorine bonds. By enabling the use of inexpensive aryl chlorides while maintaining exceptional reaction efficiency and stereochemical control, this innovation offers a compelling value proposition for R&D directors seeking to optimize synthetic routes for high-purity API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In conventional Suzuki coupling reactions, the industry has historically depended on aryl bromides and iodides due to their higher reactivity compared to chlorides. However, these halides are significantly more expensive and generate heavier waste streams, impacting both the cost reduction in pharmaceutical intermediates manufacturing and environmental compliance. Furthermore, when attempting to use cheaper aryl chlorides, standard ligands often fail to provide sufficient activity or selectivity. A critical failure point identified in prior art involves the synthesis of optically active compounds; common ligands such as tricyclohexylphosphine tetrafluoroborate or dicyclohexyl(2',6'-dimethoxy-2-biphenyl)phosphine frequently lead to racemization. For instance, in the coupling of optically active beta-chloro-alpha,beta-unsaturated five-membered cyclic lactones, these traditional catalysts result in completely racemic products (0% ee), rendering them useless for chiral drug synthesis where enantiomeric purity is paramount.
The Novel Approach
The novel approach presented in this patent utilizes a specifically engineered meta-trialkoxyphenyl dialkylphosphine tetrafluoroborate ligand. This molecule is designed with steric and electronic properties that facilitate the oxidative addition of palladium into the strong C-Cl bond. Unlike its predecessors, this ligand allows for the high-selective activation of inert chlorides without compromising the stereochemical integrity of the substrate. The synthesis of the ligand itself is remarkably streamlined, involving a concise one-step process that yields an air-stable salt. This stability is a game-changer for logistics, as it removes the necessity for stringent inert atmosphere handling during storage and transportation. Consequently, this method not only broadens the scope of usable substrates to include cheap chlorides but also ensures that complex chiral structures are preserved, delivering high-purity OLED material or pharmaceutical precursors with superior quality profiles.
Mechanistic Insights into Palladium-Catalyzed C-Cl Bond Activation
The core mechanism driving this technology involves the in situ generation of a highly active organic phosphine species from the stable tetrafluoroborate salt. Upon introduction into the reaction system containing a palladium source such as palladium acetate, the phosphine salt coordinates with the metal center to form a reactive catalytic complex. This complex possesses the unique ability to undergo oxidative addition with the inert sp2 carbon-chlorine bond of the aryl chloride substrate. The electron-donating alkoxy groups on the phenyl ring of the ligand likely enhance the electron density at the phosphorus atom, thereby increasing the nucleophilicity of the palladium center towards the electrophilic carbon of the C-Cl bond. This step is typically the rate-determining step in cross-coupling reactions involving chlorides, and the enhanced activity of this specific ligand overcomes the high bond dissociation energy of the C-Cl bond effectively.
Furthermore, the steric environment created by the dialkyl groups (such as cyclohexyl or adamantyl) and the trialkoxyphenyl backbone plays a crucial role in stabilizing the monoligated palladium species, which is often the active catalyst in these transformations. This stabilization prevents the formation of inactive palladium black and ensures a sustained catalytic cycle. In the context of chiral synthesis, the ligand architecture appears to shield the chiral center of the substrate from racemization pathways that are prevalent with bulkier or less specific ligands. This precise control over the reaction environment allows for the retention of optical activity, as evidenced by the conversion of 97% ee chlorolactones to 98% ee coupled products, a feat unachievable with conventional phosphine systems.

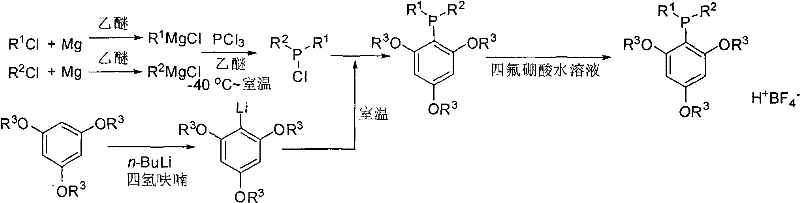
How to Synthesize Meta-Trialkoxyphenyl Dialkylphosphine Tetrafluoroborate Efficiently
The preparation of this advanced ligand is designed for operational simplicity and scalability, making it highly attractive for commercial scale-up of complex polymer additives or fine chemicals. The process begins with the lithiation of meta-trialkoxybenzene using n-butyllithium in tetrahydrofuran, followed by a controlled reaction with a dialkylchlorophosphine intermediate generated from phosphorus trichloride and a Grignard reagent. The final quenching with aqueous tetrafluoroboric acid precipitates the stable salt directly. For detailed operational parameters, stoichiometry, and safety protocols required for GMP-compliant production, please refer to the standardized synthesis guide below.
- Preparation of Lithium Reagent: React meta-trialkoxybenzene with n-butyllithium in dry tetrahydrofuran at room temperature to generate the corresponding aryl lithium species.
- Formation of Dialkylchlorophosphine: React phosphorus trichloride with alkylmagnesium chloride in dry ether at low temperature (-40°C) followed by warming to room temperature.
- Coupling and Quenching: Combine the lithium reagent with the dialkylchlorophosphine, stir, and quench with aqueous tetrafluoroboric acid to precipitate the stable phosphine salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic advantages beyond mere technical performance. The ability to substitute expensive aryl bromides and iodides with ubiquitous and low-cost aryl chlorides represents a fundamental shift in raw material sourcing strategies. This substitution drastically reduces the bill of materials for large-scale campaigns, directly impacting the bottom line without sacrificing yield or quality. Moreover, the air stability of the phosphine ligand simplifies inventory management, reducing the need for specialized storage infrastructure and minimizing the risk of material degradation during transit, thereby enhancing supply chain reliability.
- Cost Reduction in Manufacturing: The primary economic driver is the replacement of high-cost halides with inexpensive chlorides. Since aryl chlorides are often commodity chemicals with robust global supply chains, their utilization mitigates price volatility associated with specialty bromides. Additionally, the high turnover number of the catalyst system implies that lower loading levels of the expensive palladium catalyst may be feasible in optimized processes, further driving down the cost of goods sold. The simplified workup procedures, often requiring only standard extraction and crystallization, also reduce solvent consumption and waste disposal costs, contributing to substantial cost savings in fine chemical manufacturing.
- Enhanced Supply Chain Reliability: The synthesis of the ligand itself relies on readily available starting materials such as meta-trialkoxybenzene, phosphorus trichloride, and common Grignard reagents. This reliance on commodity feedstocks ensures that the production of the catalyst is not bottlenecked by scarce precursors. The air-stable nature of the final phosphine salt means that it can be stocked in standard warehouses without the need for gloveboxes or inert gas blanketing, significantly reducing lead time for high-purity pharmaceutical intermediates and ensuring continuity of supply even during logistical disruptions.
- Scalability and Environmental Compliance: The reaction conditions described, typically operating around 110°C in solvents like toluene or dioxane, are well within the capabilities of standard stainless steel reactors found in multipurpose chemical plants. The high selectivity of the reaction minimizes the formation of by-products, simplifying downstream purification and reducing the load on wastewater treatment facilities. This aligns with modern green chemistry principles by improving atom economy and reducing the E-factor of the process, making it easier to meet increasingly stringent environmental regulations while scaling up to multi-ton production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this C-Cl activation technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a clear perspective on its practical utility.
Q: Why is this phosphine salt superior to Tricyclohexylphosphine Tetrafluoroborate for chiral synthesis?
A: Unlike common ligands like tricyclohexylphosphine tetrafluoroborate which cause racemization (0% ee) in the coupling of optically active chlorolactones, this novel meta-trialkoxyphenyl derivative maintains high enantiomeric excess (up to 98% ee), preserving the stereochemical integrity of the pharmaceutical intermediate.
Q: Can this catalyst system activate inert aryl chlorides effectively?
A: Yes, the system is specifically designed to activate inert sp2 carbon-chlorine bonds with high selectivity, allowing manufacturers to utilize cheaper and more abundant aryl chlorides instead of expensive bromides or iodides.
Q: Is the ligand stable for long-term storage and logistics?
A: Yes, a key advantage of this invention is the synthesis of a phosphine salt that exists stably in air, eliminating the need for rigorous inert atmosphere handling during storage and transport, which simplifies supply chain management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meta-Trialkoxyphenyl Dialkylphosphine Tetrafluoroborate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented technology in streamlining the synthesis of complex molecular architectures. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical enantiomeric excess required for chiral pharmaceutical intermediates. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global market.
We invite you to leverage our technical expertise to evaluate the feasibility of this C-Cl activation route for your specific product portfolio. Our team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to chloride-based feedstocks. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for the future.
