Advanced Organic Catalysis for 1,2-cis-2-nitro-glucoside and Galactoside Manufacturing
Advanced Organic Catalysis for 1,2-cis-2-nitro-glucoside and Galactoside Manufacturing
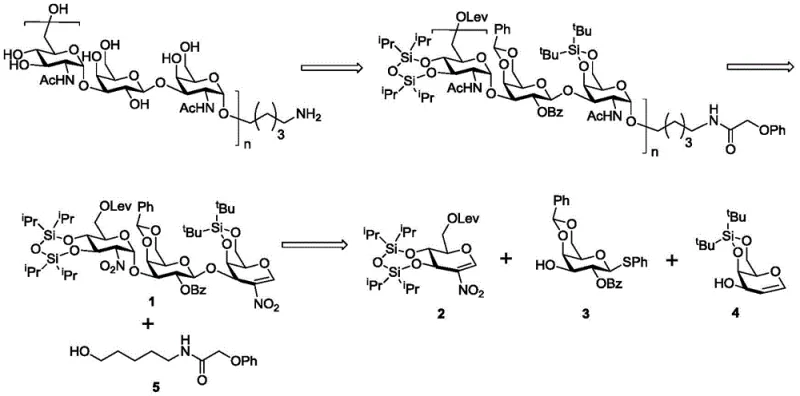
The pharmaceutical industry continuously seeks robust synthetic pathways for complex carbohydrate structures, particularly those serving as critical epitopes for vaccine development. Patent CN113234113A introduces a groundbreaking method for the efficient construction of 1,2-cis-2-nitro-glucoside and 1,2-cis-2-nitro-galactoside, addressing a long-standing challenge in glycochemistry. This technology is specifically designed to synthesize the trisaccharide repeating unit found in the O-antigen of Proteus mirabilis and Proteus vulgaris, pathogens responsible for significant urinary tract and wound infections globally. By leveraging a novel organic catalytic system, this approach bypasses the limitations of traditional biosynthesis and low-selectivity chemical methods, offering a streamlined route to high-purity vaccine intermediates. The ability to construct 1,2-cis glycosidic bonds with high stereoselectivity in a single step represents a major leap forward for the reliable pharmaceutical intermediates supplier market, ensuring that critical immunological research and vaccine development projects can proceed without synthetic bottlenecks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of high-purity polysaccharides for vaccine antigens has been plagued by significant technical and economic hurdles. Biological synthesis methods, while specific, suffer from poor substrate universality and extreme sensitivity to external environmental conditions, leading to inconsistent batch quality and high culture costs. Furthermore, the downstream processing required to separate sugar chains of different lengths from biological cultures is operationally difficult and expensive, often resulting in low overall yields. On the chemical synthesis front, traditional glycosylation strategies frequently struggle with stereocontrol, particularly when attempting to form 1,2-cis linkages which are thermodynamically less favored than their trans counterparts. Conventional promoters often require harsh conditions or expensive transition metal catalysts that introduce toxic impurities, necessitating rigorous and costly purification steps to meet regulatory standards for cost reduction in vaccine antigen manufacturing. These cumulative inefficiencies create substantial supply chain risks for manufacturers relying on outdated synthetic technologies.
The Novel Approach
The methodology disclosed in CN113234113A revolutionizes this landscape by applying a stereoselective glycosylation method driven by organic catalysis. This novel approach utilizes a nitroglucene donor activated by a specific thiourea-based organocatalyst, which facilitates the formation of the glycosidic bond under mild conditions. Unlike traditional methods that may yield complex mixtures of anomers, this system demonstrates exceptional alpha-selectivity, effectively solving the problem of constructing 1,2-cis glycosidic bonds among the most critical sugar units. The process allows for the direct assembly of the core trisaccharide unit from readily available precursors, significantly simplifying the synthetic route. By eliminating the need for transition metals and reducing the number of protection-deprotection steps, this technology lays a solid foundation for the completion of the subsequent complete synthesis of poly-O-antigen. This shift towards organocatalysis not only enhances chemical efficiency but also aligns with modern green chemistry principles, making it an ideal candidate for the commercial scale-up of complex glycosides.
Mechanistic Insights into Thiourea-Catalyzed Stereoselective Glycosylation
The core of this technological breakthrough lies in the precise activation of the nitroglucene donor by catalyst 29, a chiral thiourea derivative designed to engage in specific hydrogen-bonding interactions. The mechanism involves the catalyst binding to the nitro group of the glucene, thereby increasing the electrophilicity of the anomeric carbon while simultaneously directing the approach of the glycosyl acceptor. This dual activation mode ensures that the nucleophilic attack occurs preferentially from the alpha-face, resulting in the formation of the 1,2-cis-glycosidic linkage with high fidelity. Experimental data indicates that the choice of solvent plays a crucial role in this mechanistic pathway, with fluorobenzene and toluene providing the optimal balance between reaction rate and stereoselectivity. The reaction typically proceeds at 80°C, a temperature that is high enough to overcome the activation energy barrier but low enough to prevent the decomposition of sensitive carbohydrate structures. This level of mechanistic control is essential for R&D directors who require consistent impurity profiles and reproducible reaction outcomes for high-purity 1,2-cis-2-nitro-glucoside production.
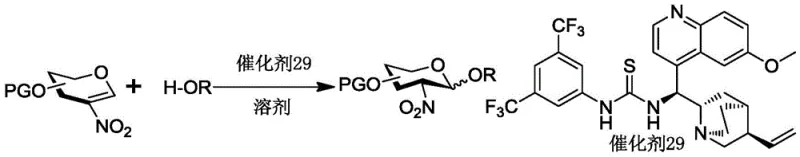
Impurity control is another critical aspect where this organic catalytic system excels over traditional Lewis acid-promoted glycosylations. In conventional metal-catalyzed processes, side reactions such as hydrolysis of the donor or formation of orthoesters are common, leading to difficult-to-remove byproducts that compromise the purity of the final API intermediate. The organocatalytic method described in the patent minimizes these side reactions by operating under neutral conditions without strong acids or metals that could degrade the sugar backbone. The use of a nitro group as a participating neighboring group further enhances stereocontrol and suppresses the formation of the unwanted beta-anomer. Furthermore, the reaction conditions allow for the use of molar ratios such as 1:1.5 for donor to acceptor, ensuring that the limiting reagent is fully consumed while minimizing waste. This precise control over the reaction environment translates directly into a cleaner crude product, reducing the burden on downstream purification processes and ensuring that the final material meets stringent purity specifications required for immunological research.
How to Synthesize 1,2-cis-2-nitro-glucoside Efficiently
Implementing this synthesis route requires a systematic approach to preparing the key nitroglucene donor and executing the catalytic glycosylation step with precision. The process begins with the modification of a simple sugar precursor, involving sequential protection of hydroxyl groups with silyl ethers and the introduction of a levulinyl ester to mask specific positions. The critical nitration step is performed at low temperatures, typically -70°C, using tetrabutylammonium nitrate and trifluoroacetic anhydride to install the nitro group at the anomeric center with high regioselectivity. Once the donor is prepared, it is coupled with various alcohol acceptors in the presence of catalyst 29 in fluorobenzene at 80°C. The detailed standardized synthesis steps see the guide below, which outlines the specific reagents, molar ratios, and workup procedures necessary to achieve the reported yields and selectivity.
- Prepare the nitroglucene donor by protecting hydroxyl groups with silyl ethers and introducing the nitro group at the anomeric position using tetrabutylammonium nitrate and trifluoroacetic anhydride.
- Mix the nitroglucene donor with the glycosyl acceptor and catalyst 29 in fluorobenzene or toluene solvent under nitrogen atmosphere.
- Heat the reaction mixture to 80°C to facilitate stereoselective glycosylation, then purify the resulting 1,2-cis-glycoside via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organic catalytic process offers transformative benefits that extend far beyond simple chemical yield. The elimination of transition metal catalysts from the synthetic route removes the need for expensive and time-consuming heavy metal scavenging steps, which are a standard requirement for pharmaceutical intermediates intended for human use. This simplification of the downstream processing workflow leads to substantial cost savings in manufacturing by reducing both the consumption of specialized purification resins and the associated labor hours. Additionally, the reagents used in this process, such as toluene and fluorobenzene, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages that often plague specialty reagent-dependent syntheses. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in temperature or moisture, enhancing supply chain reliability and ensuring consistent delivery schedules for critical vaccine components.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this technology is the removal of heavy metal catalysts, which eliminates the entire unit operation dedicated to metal clearance and validation. Traditional glycosylation methods often rely on gold, platinum, or palladium complexes that leave trace residues requiring complex filtration and testing, adding significant overhead to the cost of goods. By switching to an organocatalytic system, manufacturers can bypass these regulatory hurdles and reduce the overall solvent consumption associated with extensive washing protocols. Furthermore, the high stereoselectivity of the reaction minimizes the formation of isomeric impurities, which reduces the loss of material during chromatographic purification and improves the overall mass balance of the process. These factors combine to create a leaner, more cost-effective manufacturing model that maximizes resource utilization without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for vaccine production, and this synthetic route enhances reliability by utilizing widely available starting materials and solvents. The reliance on commodity solvents like toluene instead of specialized, low-volume halogenated solvents ensures that production is not halted due to niche reagent shortages. The reaction's tolerance to standard industrial equipment and conditions means that it can be easily transferred between different manufacturing sites without extensive re-validation, providing flexibility in sourcing. Moreover, the stability of the nitroglucene donor intermediates allows for the strategic stocking of key precursors, enabling manufacturers to respond rapidly to surges in demand for reducing lead time for high-purity pharmaceutical intermediates. This resilience against supply chain disruptions makes the technology a strategic asset for long-term procurement planning.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden environmental and safety challenges, but this method is designed with scalability in mind. The use of organic catalysts avoids the generation of heavy metal waste streams, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. The reaction operates at moderate temperatures and pressures, reducing the energy footprint associated with heating and cooling large-scale reactors. Additionally, the high atom economy of the glycosylation step minimizes the generation of organic waste, aligning with green chemistry principles that are becoming a prerequisite for supplier qualification in the global pharmaceutical market. These environmental advantages not only reduce disposal costs but also enhance the corporate sustainability profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this glycosylation technology in industrial settings. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production lines for vaccine antigens. The information covers catalyst performance, substrate scope, and process robustness to provide a comprehensive overview of the technology's capabilities.
Q: What is the primary advantage of using organic catalyst 29 over traditional metal catalysts?
A: Organic catalyst 29 eliminates the need for heavy metal reagents, thereby removing the costly and complex step of residual metal clearance required for pharmaceutical grade intermediates.
Q: How does this method improve stereoselectivity for 1,2-cis-glycosidic bonds?
A: The thiourea-based catalyst activates the nitroglucene donor through hydrogen bonding, favoring the formation of the alpha-anomer (1,2-cis) with high selectivity ratios up to 13.2:1.
Q: Is this synthesis route scalable for commercial vaccine production?
A: Yes, the process uses standard solvents like toluene and fluorobenzene and operates at moderate temperatures (80°C), making it highly suitable for scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-cis-2-nitro-glucoside Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the race to develop next-generation vaccines and therapeutics. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN113234113A and is fully prepared to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications for complex carbohydrate intermediates, guaranteeing that every batch meets the highest industry standards. By partnering with us, you gain access to a CDMO expert that understands the nuances of stereoselective glycosylation and the regulatory requirements of the pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain and reduce your time-to-market for critical vaccine components. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how our implementation of this organic catalytic method can enhance your manufacturing efficiency. Let us help you overcome synthetic bottlenecks and secure a reliable source of high-quality 1,2-cis-2-nitro-glucoside intermediates for your vital research and development programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
