Advanced Synthesis of Chiral Optical Pure Alkynylamide Substituted α,β-Diamino Acid Derivatives for Oncology
Advanced Synthesis of Chiral Optical Pure Alkynylamide Substituted α,β-Diamino Acid Derivatives for Oncology
The pharmaceutical industry continuously seeks novel scaffolds that offer enhanced biological activity and improved pharmacokinetic profiles, particularly in the realm of oncology. Patent CN111004145A introduces a groundbreaking class of chiral optical pure acetylenic amide substituted α,β-diamino acid derivatives, represented by Formula (I). These compounds are not merely structural variations but represent a significant leap in medicinal chemistry, targeting critical pathways in human colon cancer, non-small cell lung cancer, osteosarcoma, and acute promyelocytic leukemia. The core innovation lies in the efficient construction of molecules containing both carbon atom chiral centers and amide groups, structural motifs ubiquitous in high-value natural products and established therapeutics such as statins and quinolones. By leveraging a sophisticated catalytic system, this technology enables the precise assembly of complex molecular architectures that were previously challenging to access with high stereochemical fidelity.
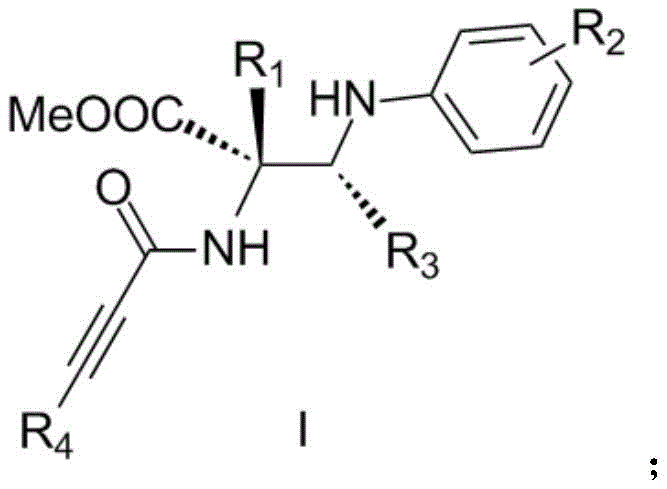
The significance of this patent extends beyond mere biological potency; it addresses the fundamental challenges of synthesizing such complex chiral entities. The presence of two aryl all-carbon chiral centers alongside an alkyne amide group creates a dense functional landscape that demands exceptional control during synthesis. The disclosed derivatives exhibit remarkable antitumor effects, with specific compounds demonstrating IC50 values as low as 0.004 μM against osteosarcoma cells. This level of potency underscores the potential of these intermediates to serve as the foundation for next-generation anticancer agents, providing a reliable pharmaceutical intermediate supplier with a distinct competitive edge in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α-diamino acid derivatives has been fraught with inefficiencies that hinder cost reduction in API manufacturing. Traditional routes often rely on multi-step sequences involving extensive protection and deprotection strategies to manage the reactivity of amino and carboxyl groups. These conventional methods frequently suffer from poor atom economy, generating substantial chemical waste and requiring large volumes of solvents for purification. Furthermore, achieving high enantiomeric excess (ee) in the formation of all-carbon quaternary chiral centers typically necessitates the use of stoichiometric chiral auxiliaries or resolution processes, which inherently limit the maximum theoretical yield to 50% unless dynamic kinetic resolution is employed. Such limitations result in prolonged lead times and inflated production costs, making the commercial scale-up of complex polymer additives or pharmaceutical intermediates economically unviable for many manufacturers.
The Novel Approach
In stark contrast, the methodology described in CN111004145A revolutionizes the synthetic landscape by employing a direct three-component coupling reaction. This novel approach utilizes a diazo compound, an aryl imine, and an alkynylamide as the primary building blocks, converging in a single operational step to form the target α,β-diamino acid derivative. 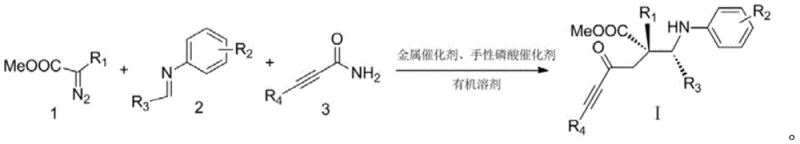
This strategy eliminates the need for pre-functionalized chiral starting materials and bypasses the tedious protection/deprotection cycles associated with classical peptide synthesis. The reaction proceeds with high selectivity and yield, driven by a dual catalytic system that ensures the simultaneous formation of multiple bonds and stereocenters. By streamlining the synthesis into a one-pot procedure, the process drastically simplifies the workflow, reduces solvent consumption, and minimizes the generation of hazardous waste, thereby aligning perfectly with modern green chemistry principles and enhancing supply chain reliability.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Three-Component Coupling
The success of this transformation hinges on the intricate interplay between a metal catalyst and a chiral organocatalyst. The reaction mechanism initiates with the activation of the diazo compound by the rhodium catalyst, typically a dirhodium tetracarboxylate species such as rhodium acetate or rhodium octanoate. This activation generates a reactive rhodium-carbenoid intermediate, a highly electrophilic species capable of undergoing subsequent transformations. However, the true brilliance of this system lies in the cooperative catalysis provided by the chiral phosphoric acid. 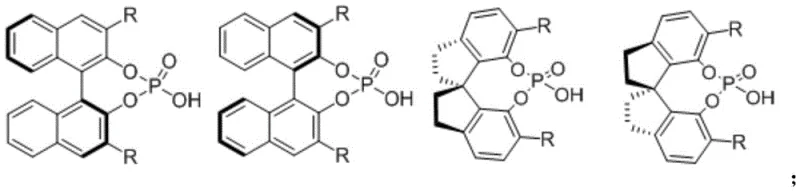
The chiral phosphoric acid acts as a Brønsted acid, activating the imine component through hydrogen bonding interactions while simultaneously creating a chiral environment around the reactive center. This dual activation mode ensures that the nucleophilic attack of the carbenoid or the subsequent rearrangement occurs with strict stereocontrol, leading to the formation of the desired enantiomer with high optical purity. The specific structure of the phosphoric acid, often featuring bulky substituents like 9-phenanthryl or 3,5-bis(trifluoromethyl)phenyl groups at the 3,3'-positions, is critical for shielding one face of the reacting species, thereby enforcing the observed enantioselectivity. This mechanistic understanding allows for the fine-tuning of catalyst structures to accommodate a wide range of substrates, ensuring robust impurity control mechanisms are in place to meet stringent purity specifications required for clinical applications.
Furthermore, the tolerance of this catalytic system towards various functional groups—such as halogens, ethers, and heterocycles like thiophene and furan—demonstrates its versatility. The ability to incorporate diverse R1, R2, and R3 substituents without compromising yield or selectivity indicates a highly optimized catalytic cycle that minimizes side reactions such as dimerization of the diazo compound or hydrolysis of the imine. This robustness is essential for maintaining consistent product quality across different batches, a key concern for procurement managers overseeing long-term supply contracts.
How to Synthesize Chiral Alkynylamide Substituted α,β-Diamino Acid Derivatives Efficiently
The practical execution of this synthesis is designed for operational simplicity and safety, addressing common pain points in laboratory and pilot plant settings. The protocol involves dissolving the aryl imine, metal rhodium catalyst, alkynylamide, chiral phosphoric acid, and activated 4Å molecular sieves in an anhydrous organic solvent, with methyl tert-butyl ether (MTBE) being the preferred medium due to its favorable safety profile and solubility characteristics. The molecular sieves play a crucial role in scavenging trace moisture, which could otherwise deactivate the sensitive rhodium-carbenoid intermediate or hydrolyze the imine substrate. To manage the exothermic nature of diazo decomposition and prevent the accumulation of unreacted diazo species, the diazo compound solution is added slowly via a syringe pump at a controlled temperature of 0°C. Detailed standardized synthesis steps follow below.
- Dissolve aryl imine, metal rhodium catalyst, alkynylamide, chiral phosphoric acid, and 4Å molecular sieves in anhydrous methyl tert-butyl ether.
- Slowly add the diazo compound solution dropwise to the mixture at 0°C over 1 hour using a syringe pump.
- Stir the reaction mixture at 0°C for 5-8 hours until the diazo compound is consumed, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this technology offers tangible strategic benefits that extend beyond simple unit price considerations. The streamlined nature of the three-component reaction directly translates into a more resilient and cost-effective supply chain. By consolidating multiple synthetic steps into a single operation, manufacturers can significantly reduce the infrastructure footprint required for production, lowering capital expenditure and operational overheads. This efficiency is paramount when aiming for cost reduction in electronic chemical manufacturing or high-value pharmaceutical sectors where margin compression is a constant challenge.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps inherent in multi-step syntheses leads to substantial cost savings. Since the reaction utilizes cheap and easily obtained raw materials such as simple diazo compounds and aryl imines, the overall material cost is kept low. Furthermore, the high atom economy of the reaction means that a greater proportion of the starting mass ends up in the final product, reducing the cost of goods sold (COGS) associated with raw material waste. The low catalyst loading, often in the range of 0.001 to 0.02 equivalents for the metal and 0.001 to 0.05 equivalents for the chiral acid, ensures that expensive catalytic species do not become a prohibitive cost factor, even at large scales.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials mitigates the risk of supply disruptions. Unlike specialized chiral pool materials that may have limited suppliers and long lead times, the precursors for this reaction are commodity chemicals produced by multiple vendors globally. This diversity in sourcing options enhances supply chain security, ensuring that production schedules are not compromised by raw material shortages. Additionally, the robustness of the reaction conditions allows for flexible manufacturing planning, as the process is not overly sensitive to minor fluctuations in environmental parameters, thereby reducing the risk of batch failures.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with straightforward equipment setups. The use of safer solvents like MTBE and the avoidance of highly toxic reagents simplify waste management and regulatory compliance. The high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the burden on downstream purification units and lowering the volume of solvent waste generated. This alignment with green chemistry principles not only reduces disposal costs but also supports corporate sustainability goals, making the final product more attractive to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral derivatives. Understanding these aspects is crucial for integrating this technology into existing development pipelines and ensuring seamless technology transfer.
Q: What is the role of the chiral phosphoric acid in this synthesis?
A: The chiral phosphoric acid acts as a chiral Brønsted acid catalyst that works synergistically with the rhodium metal catalyst to induce high enantioselectivity, ensuring the formation of the desired optical pure stereocenters in the α,β-diamino acid backbone.
Q: Can this reaction be scaled up for commercial production?
A: Yes, the process is designed for scalability. It utilizes a one-pot procedure with readily available raw materials and operates under mild conditions (0°C to 40°C), making it suitable for commercial scale-up of complex pharmaceutical intermediates without requiring extreme pressure or temperature equipment.
Q: What represents the primary advantage of this method over traditional amino acid synthesis?
A: The primary advantage is the construction of two all-carbon chiral centers and an amide group in a single step with high atom economy. Traditional methods often require multiple protection and deprotection steps, whereas this three-component coupling significantly simplifies the synthetic route.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral α,β-Diamino Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chiral optical pure alkynylamide substituted α,β-diamino acid derivatives described in CN111004145A. As a premier CDMO partner, we possess the technical expertise and infrastructure necessary to translate this innovative academic research into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move smoothly from gram-scale optimization to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest standards of quality and consistency required for clinical and commercial use.
We invite you to collaborate with us to unlock the full commercial potential of this technology. Whether you require a Customized Cost-Saving Analysis for your specific target molecule or need to evaluate the feasibility of this route for your portfolio, our technical procurement team is ready to assist. Contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the rapidly evolving oncology market.
