Advanced Benzimidazole Cyclophane Synthesis for High-Performance Optoelectronic Applications
The chemical industry is constantly evolving, driven by the need for more efficient and selective materials for advanced applications. Patent CN108822130B introduces a significant breakthrough in the field of supramolecular chemistry with the disclosure of a novel benzimidazole cyclophane and its preparation method. This specific molecular architecture combines the robust electronic properties of an anthraquinone core with the versatile coordination chemistry of benzimidazole units. For R&D directors and procurement specialists in the optoelectronic sector, this development represents a pivotal shift towards more tunable and sensitive fluorescent sensor materials. The patent details a robust synthetic pathway that avoids complex catalytic systems, relying instead on precise nucleophilic substitutions and macrocyclization techniques. This approach not only enhances the structural integrity of the final product but also streamlines the manufacturing process, making it highly relevant for commercial scale-up of complex optoelectronic intermediates. The ability of this cyclophane to act as a highly selective host for specific anions, particularly dihydrogen phosphate, underscores its potential value in environmental analysis and clinical diagnostics. By leveraging this technology, manufacturers can achieve higher purity specifications and more consistent batch-to-batch performance in fluorescent probe production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing cyclophane structures often suffer from significant drawbacks that hinder their widespread commercial adoption in high-tech industries. Many conventional routes rely heavily on transition metal-catalyzed cross-coupling reactions, which introduce the risk of metal contamination that is difficult and costly to remove to ppm levels. Furthermore, these traditional pathways frequently require harsh reaction conditions, such as extremely high temperatures or pressures, which can degrade sensitive functional groups like the benzimidazole moiety. The lack of regioselectivity in older alkylation methods often leads to complex mixtures of isomers, necessitating extensive and yield-reducing purification steps like preparative HPLC. Additionally, the starting materials for many legacy processes are often proprietary or expensive specialty chemicals, creating supply chain vulnerabilities and inflating the cost reduction in fluorescent sensor material manufacturing. These inefficiencies result in longer lead times and inconsistent product quality, which are critical pain points for supply chain heads managing just-in-time inventory for electronic material production lines.
The Novel Approach
The methodology outlined in patent CN108822130B offers a transformative solution by utilizing a stepwise alkylation strategy that maximizes yield and minimizes impurity formation. This novel approach begins with the efficient etherification of 1,4-dihydroxy-9,10-anthraquinone using 1,2-dibromoethane, a reaction that proceeds under mild basic conditions with high conversion rates. Subsequently, the benzimidazole units are linked via a propyl chain using 1,3-dibromopropane, creating a pre-organized linker that facilitates the final macrocyclization step. The use of common organic solvents such as acetone, tetrahydrofuran, and acetonitrile ensures that the process is easily adaptable to existing reactor infrastructure without requiring specialized equipment. This synthetic route eliminates the need for expensive catalysts, thereby significantly reducing raw material costs and simplifying the downstream workup procedure. The final ion exchange step with ammonium hexafluorophosphate ensures the formation of a stable salt form, enhancing the solubility and handling properties of the final fluorescent sensor material. 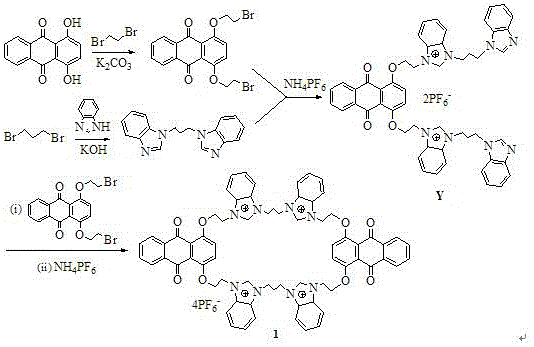
Mechanistic Insights into Host-Guest Fluorescence Recognition
The core value of this benzimidazole cyclophane lies in its sophisticated supramolecular mechanism, which allows for precise molecular recognition through non-covalent interactions. The anthraquinone unit serves as the fluorophore, emitting light at specific wavelengths, while the benzimidazole nitrogen atoms act as hydrogen bond donors or acceptors depending on the pH and guest species. When a target anion, such as H2PO4-, enters the cavity of the cyclophane, it forms a stable host-guest complex that alters the electronic environment of the anthraquinone core. This interaction typically results in a measurable red shift in the emission spectrum and a change in fluorescence intensity, providing a clear optical signal for detection. The rigid yet flexible nature of the cyclophane ring allows it to adapt to the size and shape of the guest molecule, ensuring high selectivity against competing anions like chloride or nitrate. Understanding this mechanism is crucial for R&D teams aiming to modify the structure for sensing other specific analytes in complex biological or environmental matrices. The crystal structure data confirms the spatial arrangement of the benzimidazole arms, validating the theoretical models of the binding pocket.
Controlling impurities in such complex macrocycles is essential to maintain the fidelity of the fluorescence signal and ensure reliable performance in sensing applications. The stepwise synthesis described in the patent inherently limits the formation of oligomeric byproducts, which are common pitfalls in macrocyclization reactions. By carefully controlling the molar ratios of the intermediates, specifically maintaining a 1:1 ratio during the cyclization step, the process favors the formation of the monomeric cyclophane over linear polymers. The use of recrystallization from ethyl acetate and methanol further purifies the intermediates, removing unreacted starting materials and salts that could quench fluorescence. Rigorous QC labs analyze the final product using NMR and mass spectrometry to confirm the absence of these impurities, ensuring that the high-purity fluorescent sensor material meets stringent specifications. This level of purity is vital for procurement managers who need to guarantee the performance of the final electronic or diagnostic devices to their end customers. The stability of the hexafluorophosphate salt form also contributes to the long-term shelf life of the product, reducing waste and improving inventory management.
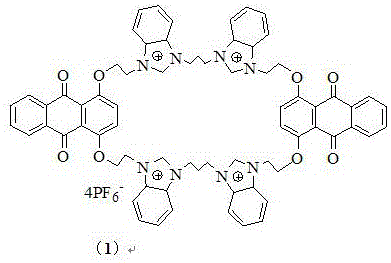
How to Synthesize Benzimidazole Cyclophane Efficiently
Implementing this synthesis in a commercial setting requires a clear understanding of the critical process parameters and safety considerations associated with the reagents involved. The patent provides a detailed roadmap that begins with the preparation of the bromoethoxy-anthraquinone intermediate, which serves as the electrophilic core for the subsequent coupling. Operators must ensure that the reaction temperature is maintained at reflux for the specified duration to drive the alkylation to completion while monitoring for any signs of decomposition. The subsequent coupling with the bis-benzimidazolyl propane linker requires precise addition rates to prevent local concentration spikes that could lead to polymerization.
- React 1,4-dihydroxy-9,10-anthraquinone with 1,2-dibromoethane in acetone with K2CO3 to form the bromoethoxy intermediate.
- Alkylate benzimidazole with 1,3-dibromopropane in THF using KOH to generate the bis-benzimidazolyl propane linker.
- Perform macrocyclization by reacting the intermediates in acetonitrile under reflux, followed by ion exchange with ammonium hexafluorophosphate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers substantial strategic advantages beyond mere technical performance. The reliance on commodity chemicals like 1,2-dibromoethane and 1,3-dibromopropane means that raw material sourcing is robust and less susceptible to geopolitical disruptions or single-supplier bottlenecks. This availability translates directly into enhanced supply chain reliability, allowing manufacturers to maintain consistent production schedules even during market fluctuations. Furthermore, the elimination of transition metal catalysts removes the need for expensive scavenging resins and complex filtration steps, leading to significant cost reduction in fluorescent sensor material manufacturing. The simplified workup procedure also reduces solvent consumption and waste generation, aligning with increasingly strict environmental compliance regulations and lowering disposal costs. These efficiencies collectively improve the overall margin profile of the product, making it a more attractive option for high-volume applications in the optoelectronic and diagnostic sectors.
- Cost Reduction in Manufacturing: The synthetic pathway described eliminates the need for precious metal catalysts such as palladium or platinum, which are traditionally used in cross-coupling reactions for similar structures. By avoiding these expensive reagents, the direct material cost is drastically lowered, and the associated costs of metal removal and validation are completely removed from the budget. Additionally, the use of standard solvents and ambient pressure conditions reduces energy consumption and equipment wear, contributing to lower overheads per kilogram of product. This economic efficiency allows for more competitive pricing strategies without compromising on the quality or purity of the final fluorescent sensor material.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are bulk commodity chemicals that are produced by multiple manufacturers globally, ensuring a diversified and secure supply base. This reduces the risk of production stoppages due to raw material shortages, a common issue with specialized intermediates that have single-source suppliers. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, further stabilizing the supply chain. For supply chain heads, this reliability is crucial for maintaining long-term contracts with downstream electronics and pharmaceutical clients who demand uninterrupted delivery.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction types that are well-understood and easily transferred from laboratory to pilot and commercial scales. The absence of toxic heavy metals simplifies the waste treatment process, making it easier to meet environmental discharge standards and reducing the regulatory burden on the manufacturing site. The high atom economy of the alkylation steps minimizes waste generation, supporting sustainability goals and reducing the carbon footprint of the manufacturing operation. This alignment with green chemistry principles enhances the corporate social responsibility profile of the supplier, a key factor for modern procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this benzimidazole cyclophane. These answers are derived directly from the experimental data and claims within patent CN108822130B to ensure accuracy and relevance for technical decision-makers. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their existing product lines or research pipelines.
Q: What is the primary application of the benzimidazole cyclophane described in patent CN108822130B?
A: The primary application is as a host molecule in fluorescent molecular recognition systems, specifically demonstrating high selectivity for dihydrogen phosphate anions (H2PO4-) through distinct fluorescence spectral shifts.
Q: How does the novel synthesis route improve upon conventional methods for cyclophane production?
A: The novel route utilizes readily available starting materials like 1,4-dihydroxy-9,10-anthraquinone and employs a stepwise alkylation strategy that simplifies purification and avoids the use of expensive transition metal catalysts often required in traditional cross-coupling methods.
Q: What are the key structural features enabling fluorescence sensing in this compound?
A: The compound features a rigid anthraquinone core linked by flexible benzimidazole-containing chains, creating a specific cavity that facilitates host-guest interactions, resulting in observable red shifts and intensity changes in fluorescence spectra upon anion binding.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole Cyclophane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating advanced patent technologies like CN108822130B into reliable commercial realities for our global partners. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We are committed to delivering stringent purity specifications and maintaining rigorous QC labs to verify the structural integrity and fluorescence performance of every batch. Our expertise in handling complex macrocycles and fluorescent materials allows us to optimize the process further, potentially improving yields and reducing cycle times beyond the baseline patent data. By partnering with us, you gain access to a supply chain that is both resilient and responsive to the dynamic needs of the optoelectronic and fine chemical markets.
We invite you to collaborate with us to explore how this benzimidazole cyclophane can enhance your product portfolio and drive innovation in your sensing applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us to request specific COA data and route feasibility assessments that will demonstrate our capability to meet your exacting demands. Let us help you secure a stable supply of high-performance fluorescent sensor materials that will give you a competitive edge in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
