Streamlined Synthesis of Coumarin 3-Carboxamide Derivatives for Advanced Antiviral Drug Development
The global pharmaceutical landscape is constantly seeking novel scaffolds to combat viral resistance, particularly in the realm of influenza treatment where neuraminidase inhibitors play a pivotal role. Patent CN113461648A introduces a significant advancement in this domain by disclosing a series of coumarin 3-carboxamide derivatives designed as potent neuraminidase inhibitors. Unlike traditional approaches that often rely on complex natural product extraction or multi-step chiral synthesis, this invention leverages a concise two-step synthetic pathway to access a diverse library of bioactive molecules. The core innovation lies in the structural novelty of the coumarin backbone functionalized at the 3-position with an amide linkage, providing a robust platform for structure-activity relationship (SAR) studies. 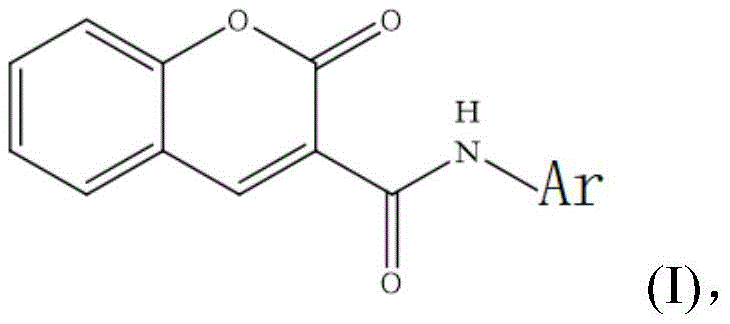 As depicted in the general formula, the versatility of the 'Ar' group allows for extensive chemical modification, enabling researchers to fine-tune pharmacokinetic properties and binding affinity without altering the core synthetic strategy. This patent represents a critical opportunity for R&D teams looking to diversify their antiviral pipelines with synthetically accessible, non-natural scaffolds that bypass existing intellectual property barriers associated with first-generation drugs.
As depicted in the general formula, the versatility of the 'Ar' group allows for extensive chemical modification, enabling researchers to fine-tune pharmacokinetic properties and binding affinity without altering the core synthetic strategy. This patent represents a critical opportunity for R&D teams looking to diversify their antiviral pipelines with synthetically accessible, non-natural scaffolds that bypass existing intellectual property barriers associated with first-generation drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of effective neuraminidase inhibitors like Oseltamivir (Tamiflu) has been plagued by significant supply chain vulnerabilities and synthetic inefficiencies. The industrial synthesis of Oseltamivir typically involves a nine-step sequence that is heavily dependent on shikimic acid, a natural product extracted from star anise or produced via fermentation, leading to volatile pricing and supply bottlenecks. Furthermore, the conventional routes often require harsh reaction conditions, expensive chiral catalysts, and rigorous purification protocols to ensure stereochemical purity, which drastically inflates the cost of goods sold (COGS). From a process chemistry perspective, the reliance on transition metal catalysis or hazardous reagents in older methodologies introduces substantial environmental burdens and complicates waste management, making large-scale manufacturing increasingly difficult to justify under modern green chemistry standards. These legacy processes also suffer from long lead times, as the cumulative yield losses over nine distinct chemical transformations necessitate massive starting material inputs to achieve commercially viable output quantities.
The Novel Approach
In stark contrast, the methodology outlined in CN113461648A offers a paradigm shift towards efficiency and simplicity by utilizing a direct condensation strategy. The novel approach bypasses the need for chiral pool starting materials entirely, instead constructing the bioactive coumarin core through a classic yet highly optimized Knoevenagel condensation followed by a straightforward amidation. This two-step sequence not only reduces the operational complexity but also significantly minimizes the accumulation of impurities that typically arise in longer synthetic chains. By employing common organic solvents like absolute ethanol and benign catalysts such as piperidine and acetic acid, the process aligns much more closely with sustainable manufacturing principles. The ability to generate the final active pharmaceutical ingredient (API) intermediate in just two steps from commodity chemicals means that production capacity can be scaled up rapidly without the need for specialized equipment or exotic reagents. This streamlined workflow effectively decouples the production of antiviral intermediates from the fluctuations of the agricultural market, offering a more predictable and stable manufacturing baseline for pharmaceutical companies.
Mechanistic Insights into the Two-Step Condensation Strategy
The chemical elegance of this synthesis lies in its mechanistic simplicity, which facilitates high purity and reproducibility. The first step involves the reaction of salicylaldehyde with diethyl malonate, catalyzed by a piperidine-acetic acid system in ethanol at mild temperatures ranging from 77°C to 79°C. This Knoevenagel-type condensation proceeds through the formation of an enamine intermediate, which subsequently undergoes intramolecular cyclization and elimination of ethanol to form the coumarin-3-carboxylate ester intermediate (Formula II). The use of acetic acid as a co-catalyst is crucial here, as it helps to moderate the basicity of piperidine, preventing side reactions such as polymerization of the aldehyde while ensuring rapid conversion to the cyclic ester. The reaction mixture is then subjected to a simple workup involving cooling and dilution with ice water, causing the intermediate to precipitate as a solid filter cake, which inherently purifies the material by excluding soluble byproducts.
The second transformation converts this ester intermediate into the final amide target through nucleophilic acyl substitution. By reacting the coumarin ester with various substituted anilines in the presence of sodium ethoxide, the ethoxy leaving group is displaced by the aniline nitrogen. 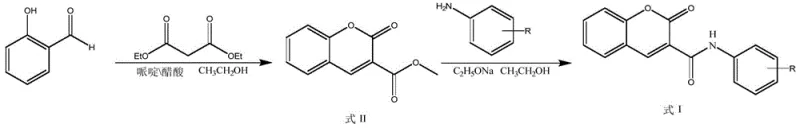 This amidation step is driven by the formation of the stable amide bond and is facilitated by the basic conditions provided by sodium ethoxide, which activates the aniline nucleophile. The reaction is typically conducted at similar mild temperatures (77-79°C) over a period of 24 to 36 hours to ensure complete conversion. A key advantage of this mechanism is the ease of isolation; upon completion, the reaction mixture is cooled and poured into an ice-water bath, inducing precipitation of the final product. This 'precipitation-driven' purification strategy eliminates the need for column chromatography or complex extraction procedures, which are often the most costly and time-consuming aspects of fine chemical synthesis. The result is a high-purity solid product suitable for direct biological evaluation or further formulation development.
This amidation step is driven by the formation of the stable amide bond and is facilitated by the basic conditions provided by sodium ethoxide, which activates the aniline nucleophile. The reaction is typically conducted at similar mild temperatures (77-79°C) over a period of 24 to 36 hours to ensure complete conversion. A key advantage of this mechanism is the ease of isolation; upon completion, the reaction mixture is cooled and poured into an ice-water bath, inducing precipitation of the final product. This 'precipitation-driven' purification strategy eliminates the need for column chromatography or complex extraction procedures, which are often the most costly and time-consuming aspects of fine chemical synthesis. The result is a high-purity solid product suitable for direct biological evaluation or further formulation development.
How to Synthesize Coumarin 3-Carboxamide Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the thermal and stoichiometric parameters defined in the patent to maximize yield and purity. The process is designed to be operationally simple, utilizing standard glassware and heating mantles, making it highly accessible for process development teams. The critical control points involve maintaining the reaction temperature within the narrow 77-79°C window to prevent thermal degradation of the sensitive coumarin ring while ensuring sufficient energy for the condensation and amidation events. Additionally, the ratio of reagents, particularly the slight excess of diethyl malonate and the precise amount of sodium ethoxide, must be controlled to drive the equilibrium towards the desired product without generating excessive salt waste. For detailed operational instructions, please refer to the standardized protocol below.
- Condense salicylaldehyde with diethyl malonate using piperidine and acetic acid catalysts in ethanol at 78°C to form the coumarin ester intermediate.
- React the resulting coumarin ester intermediate with substituted anilines in the presence of sodium ethoxide in ethanol.
- Isolate the final coumarin 3-carboxamide inhibitor via cooling, ice-water precipitation, filtration, and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the economic implications of adopting this two-step synthetic route are profound, primarily driven by the radical simplification of the manufacturing process. The reduction from a multi-step, resource-intensive protocol to a concise two-step sequence translates directly into lower operational expenditures and reduced capital tie-up in work-in-progress inventory. By eliminating the need for scarce natural precursors and complex chiral resolution steps, manufacturers can secure a more resilient supply chain that is less susceptible to external shocks such as crop failures or geopolitical trade restrictions. Furthermore, the use of ethanol as the primary solvent throughout both steps simplifies solvent recovery and recycling operations, contributing to a smaller environmental footprint and lower waste disposal costs. This process intensification allows for faster batch turnover times, enabling suppliers to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The most significant financial benefit arises from the drastic reduction in unit operations. Traditional antiviral syntheses often incur high costs due to the cumulative loss of material across numerous steps and the expense of specialized catalysts. In this novel route, the starting materials—salicylaldehyde, diethyl malonate, and substituted anilines—are bulk commodity chemicals available at competitive prices from a wide range of global vendors. The elimination of transition metal catalysts removes the necessity for expensive metal scavenging and testing, which is a major cost driver in API manufacturing. Moreover, the simplified workup procedure, which relies on precipitation and filtration rather than chromatography, significantly reduces labor hours and consumable costs associated with purification. This lean manufacturing approach ensures that the cost of goods sold remains low, even when scaling to metric ton quantities, providing a substantial margin advantage for downstream drug developers.
- Enhanced Supply Chain Reliability: Supply chain stability is paramount in the pharmaceutical industry, and this synthesis offers superior reliability compared to natural product-dependent routes. Since all raw materials are synthetic and petrochemical-derived, their availability is not subject to the seasonal variability or agricultural risks associated with star anise harvesting. The robustness of the chemical process means that production can be maintained continuously throughout the year, ensuring a steady flow of intermediates to formulation plants. Additionally, the simplicity of the reaction conditions allows for production to be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without requiring highly specialized infrastructure. This flexibility mitigates the risk of single-source dependency and allows procurement teams to diversify their supplier base, thereby strengthening the overall resilience of the drug supply network against disruptions.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but the exothermic nature and mild conditions of this reaction make it highly amenable to scale-up. The use of ethanol, a green solvent with a favorable safety profile, simplifies regulatory compliance regarding volatile organic compound (VOC) emissions. The precipitation-based isolation method generates a solid product with high purity, minimizing the generation of liquid waste streams that require complex treatment. This aligns well with modern environmental, social, and governance (ESG) goals, reducing the burden on wastewater treatment facilities. The ability to run the reaction at near-atmospheric pressure and moderate temperatures also lowers energy consumption compared to high-pressure hydrogenation or cryogenic reactions found in other antiviral syntheses. Consequently, this process offers a sustainable pathway for commercial scale-up that meets stringent environmental regulations while maintaining high production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and potential of this coumarin-based technology. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, aiming to clarify the feasibility of adoption for industrial partners. Understanding the specific advantages of this scaffold over existing therapies is crucial for strategic decision-making in R&D portfolio management.
Q: What is the primary advantage of this coumarin synthesis route compared to Oseltamivir?
A: The primary advantage is the drastic reduction in synthetic complexity. While Oseltamivir requires a complex 9-step synthesis often dependent on scarce shikimic acid, this patented method achieves the target scaffold in only two straightforward steps using readily available commodity chemicals.
Q: Are the starting materials for these inhibitors commercially scalable?
A: Yes, the synthesis relies on salicylaldehyde, diethyl malonate, and various substituted anilines. These are bulk commodity chemicals available from multiple global suppliers, ensuring robust supply chain continuity and mitigating the risk of raw material shortages common with natural product-derived drugs.
Q: What is the reported biological activity of these compounds?
A: The patent reports that specific derivatives, such as N-(3,4-dimethoxyphenyl)-2-oxo-2H-chromene-3-carboxamide, exhibit neuraminidase inhibitory activity with an IC50 value of 23.78 μM. While lower than the positive control Oseltamivir acid, the novel scaffold offers a distinct chemical space for further medicinal chemistry optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin 3-Carboxamide Supplier
As the pharmaceutical industry continues to evolve, the demand for innovative and cost-effective antiviral intermediates has never been higher. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this promising technology to market. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of the coumarin synthesis route, ensuring that every batch meets stringent purity specifications required for clinical and commercial applications. With our rigorous QC labs and commitment to process optimization, we can guarantee the consistent quality and supply continuity that global health initiatives demand, positioning us as a strategic partner for your antiviral drug development programs.
We invite you to explore the potential of this novel scaffold for your next-generation influenza therapeutics. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this streamlined synthesis can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data for our reference standards and to discuss route feasibility assessments for your custom analogues. Let us collaborate to accelerate the delivery of life-saving medicines to patients worldwide through superior chemistry and reliable supply chain execution.
