Advanced Synthesis of Pinoxaden Metabolite M3 for Regulatory Compliance and Commercial Scale
Advanced Synthesis of Pinoxaden Metabolite M3 for Regulatory Compliance and Commercial Scale
The global agrochemical industry is facing increasingly stringent regulatory landscapes, particularly concerning the environmental fate and toxicological profiles of active ingredients. As highlighted in the recent technical disclosure CN115894518A, the demand for high-purity reference standards and metabolites has surged to meet these compliance requirements. Pinoxaden, a widely used phenylpyrazoline herbicide, generates various metabolites upon application, among which Metabolite M3 is of paramount importance for safety assessments. This patent introduces a groundbreaking synthetic pathway that transforms the commercially available parent compound, Pinoxaden, directly into Metabolite M3 through a streamlined hydrolysis-nitration-rehydrolysis sequence. For R&D directors and procurement specialists, this innovation represents a significant leap forward, offering a route that bypasses the complexities of de novo synthesis while delivering product purity exceeding 99% and overall yields greater than 90%. The ability to secure reliable supplies of such critical intermediates is no longer just a logistical advantage but a strategic necessity for maintaining product registration and market continuity in the competitive herbicide sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of specific pesticide metabolites like Pinoxaden Metabolite M3 has been fraught with significant technical and economic challenges. Conventional approaches often involve multi-step de novo synthesis starting from basic petrochemical feedstocks, which necessitates the construction of the complex bicyclic pyrazolone core from scratch. These traditional routes are typically characterized by low atom economy, the requirement for harsh reaction conditions, and the use of expensive, specialized reagents that drive up production costs substantially. Furthermore, the purification of intermediates in these lengthy sequences often results in substantial material loss, leading to overall yields that are commercially unviable for large-scale toxicological testing. The scarcity of reported methods in prior art underscores the difficulty chemists have faced in accessing this molecule efficiently. For supply chain managers, relying on such inefficient processes translates to long lead times, high volatility in pricing, and a constant risk of supply disruption, which can jeopardize critical regulatory submission timelines.
The Novel Approach
In stark contrast to the arduous traditional pathways, the methodology disclosed in patent CN115894518A leverages the structural integrity of the parent herbicide, Pinoxaden, as the starting material. This strategy effectively shortens the synthetic timeline by utilizing the pre-formed heterocyclic core, requiring only specific functional group modifications to arrive at the target metabolite. The process initiates with a mild hydrolysis step to generate a key intermediate, followed by a highly selective nitration reaction, and concludes with a final hydrolysis to unveil the Metabolite M3 structure. This approach not only simplifies the operational complexity but also drastically reduces the number of unit operations required. By shifting the paradigm from building the molecule up to modifying an existing scaffold, the novel method achieves a total yield exceeding 90%, a figure that is exceptionally high for fine chemical synthesis of this nature. This efficiency gain directly addresses the pain points of both R&D teams needing rapid access to materials and procurement officers seeking cost-effective sourcing solutions.
Mechanistic Insights into the Hydrolysis-Nitration Cascade
The chemical elegance of this synthesis lies in its precise control over reaction conditions to ensure high selectivity and minimal byproduct formation. The first stage involves the hydrolysis of the ester moiety on the Pinoxaden molecule. As depicted in the reaction scheme, treating Pinoxaden with sodium hydroxide in a methanol-water solvent system at a controlled temperature range of 30-50°C facilitates the cleavage of the ester bond. This step is critical as it generates the free acid or enolic form necessary for subsequent functionalization. The choice of mild alkaline conditions prevents the degradation of the sensitive pyrazolone ring, a common pitfall in harsher hydrolysis protocols. Following isolation, the intermediate undergoes a nitration reaction using sodium nitrite and N-halosuccinimide (such as N-bromosuccinimide) in DMF. This specific combination of reagents allows for the introduction of the nitro group or related functionality under mild temperatures of 20-30°C, avoiding the explosive risks and non-selective oxidation often associated with traditional mixed-acid nitration methods.
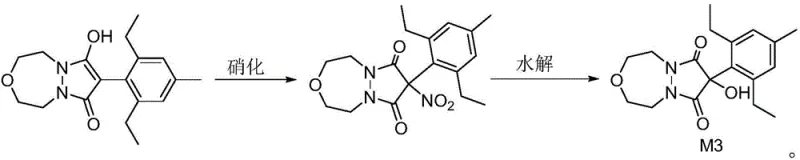
The final transformation involves a re-hydrolysis step where the nitrated intermediate is treated with ethyl acetate and water at elevated temperatures between 75-85°C. This step serves to finalize the structural arrangement of Metabolite M3, ensuring the correct positioning of hydroxyl and carbonyl groups as required for biological activity and detection. From an impurity control perspective, the use of crystallization techniques at each stage—first in methanol, then in ethyl acetate, and finally in ethanol—acts as a powerful purification engine. This iterative purification strategy ensures that the final product achieves a purity level of over 99%, which is essential for its use as a reference standard in analytical chemistry and toxicology. The mechanistic pathway avoids the use of transition metal catalysts, thereby eliminating the risk of heavy metal contamination, a crucial factor for meeting the stringent quality specifications demanded by global regulatory bodies for agrochemical residues.
How to Synthesize Pinoxaden Metabolite M3 Efficiently
Implementing this synthesis protocol requires careful attention to stoichiometry and temperature control to replicate the high yields reported in the patent data. The process is designed to be scalable, moving seamlessly from gram-scale laboratory preparation to multi-kilogram production without significant re-optimization. Operators should focus on maintaining the specific molar ratios, particularly the 1:1.5:1 ratio of the intermediate to sodium nitrite and N-halosuccinimide, to maximize conversion while minimizing side reactions. The detailed standardized operating procedures for each reaction stage, including work-up and purification, are critical for ensuring batch-to-batch consistency. For a comprehensive guide on the exact parameters and safety precautions required for execution, please refer to the technical breakdown below.
- Hydrolyze pinoxaden using sodium hydroxide in methanol/water at 30-50°C to obtain the primary intermediate.
- Perform nitration on the intermediate using sodium nitrite and N-halosuccinimide in DMF at 20-30°C.
- Conduct final hydrolysis of the nitrated product in ethyl acetate and water at 75-85°C to yield Metabolite M3.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain; by starting with Pinoxaden technical grade material, which is produced on a massive industrial scale, manufacturers can bypass the volatile supply chains associated with exotic starting materials. This reliance on a commoditized feedstock significantly enhances supply security and stabilizes pricing structures. Furthermore, the elimination of complex catalytic systems and the use of common solvents like methanol, DMF, and ethyl acetate reduce the dependency on specialized chemical vendors. The operational simplicity of the process, characterized by moderate temperatures and atmospheric pressure conditions, lowers the barrier for manufacturing partners, allowing for broader sourcing options and increased competition among suppliers, which ultimately drives down costs for the end buyer.
- Cost Reduction in Manufacturing: The economic impact of this new method is profound, primarily driven by the high overall yield and the avoidance of expensive reagents. Traditional routes often suffer from cumulative yield losses across multiple steps, whereas this three-step process maintains high efficiency throughout, significantly reducing the cost of goods sold (COGS). Additionally, the absence of precious metal catalysts removes the need for costly scavenging processes to meet residual metal limits, further trimming production expenses. The use of inexpensive, bulk-available reagents like sodium hydroxide and sodium nitrite ensures that raw material costs remain low and predictable. These factors combine to create a highly cost-competitive manufacturing profile, allowing for substantial savings that can be passed down the supply chain or reinvested into further R&D initiatives.
- Enhanced Supply Chain Reliability: In the context of global agrochemical markets, reliability is as valuable as price. This synthesis route mitigates supply risk by utilizing Pinoxaden, a mature product with established production capacity worldwide. Unlike niche intermediates that may be sourced from a single supplier, Pinoxaden is available from multiple manufacturers, providing a robust safety net against production outages. The simplified process flow also means shorter manufacturing cycles, enabling faster turnaround times from order to delivery. This agility is crucial for meeting the tight deadlines associated with regulatory filings and seasonal agricultural demands. By securing a supply of Metabolite M3 through this efficient route, companies can ensure uninterrupted progress in their toxicological testing programs, avoiding costly delays in product registration.
- Scalability and Environmental Compliance: Scalability is inherent in the design of this process, as it relies on unit operations that are standard in the fine chemical industry. The reaction conditions are mild enough to be safely scaled from pilot plants to full commercial production without requiring specialized high-pressure equipment. From an environmental standpoint, the process aligns well with green chemistry principles by minimizing waste generation through high atom economy and avoiding toxic heavy metals. The solvents used are readily recoverable and recyclable, reducing the environmental footprint of the manufacturing process. This compliance with environmental standards not only simplifies waste disposal logistics but also future-proofs the supply chain against tightening environmental regulations, ensuring long-term sustainability of the production capability.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential impact on their operations. The following questions address common inquiries regarding the process capabilities, quality metrics, and regulatory alignment. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a factual basis for decision-making. Whether you are assessing the feasibility of in-house production or evaluating external suppliers, these insights clarify the operational realities of generating high-purity Pinoxaden Metabolite M3.
Q: What is the total yield of the new Pinoxaden Metabolite M3 synthesis method?
A: According to patent CN115894518A, the optimized three-step process achieves a total yield exceeding 90%, with individual step yields often surpassing 94-97%.
Q: Why is Pinoxaden Metabolite M3 critical for agrochemical companies?
A: Metabolite M3 is essential for conducting mandatory toxicological experiments required for pesticide registration under new regulations, ensuring market access for herbicide products.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the process utilizes common reagents like sodium hydroxide, sodium nitrite, and N-halosuccinimide, eliminating the need for costly precious metal catalysts and simplifying purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pinoxaden Metabolite M3 Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of agrochemical products hinges on the availability of high-quality intermediates and reference standards. Our team of expert chemists has thoroughly analyzed the synthetic route described in CN115894518A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering Pinoxaden Metabolite M3 with stringent purity specifications, ensuring that every batch meets the rigorous demands of pesticide registration and quality control laboratories. Our state-of-the-art facilities and rigorous QC labs guarantee that our products are consistent, reliable, and fully compliant with international standards, providing you with the confidence needed to advance your regulatory submissions.
We invite you to collaborate with us to optimize your supply chain for this critical metabolite. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our efficient manufacturing processes can reduce your overall expenditure. We encourage you to contact us today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable source of high-purity agrochemical intermediates, empowering your R&D and regulatory teams to achieve their goals with speed and precision.
