Advanced Chiral Phosphoric Amide Catalysts For Scalable Pharmaceutical Manufacturing
The landscape of asymmetric synthesis is undergoing a transformative shift with the introduction of patent CN102309984B, which details a novel class of phosphoric acid amide bifunctional catalysts derived from cinchona alkaloids. This technology represents a significant leap forward for the production of high-value pharmaceutical intermediates, offering a robust alternative to traditional transition metal catalysis. By leveraging the inherent chirality of natural alkaloids and combining it with a tunable phosphoric acid amide moiety, this invention provides a powerful tool for constructing complex chiral molecules with exceptional precision. The strategic importance of this development lies in its ability to simplify synthetic routes while maintaining rigorous stereochemical control, a critical factor for R&D directors focused on impurity profiles and process feasibility. As the industry moves towards greener and more sustainable manufacturing practices, the adoption of such stable and efficient organocatalysts becomes not just a technical preference but a commercial imperative for maintaining competitiveness in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral intermediates has relied heavily on transition metal catalysts or multi-step enzymatic processes that introduce significant complexity and cost into the manufacturing workflow. Conventional methods often require stringent exclusion of moisture and oxygen, necessitating specialized equipment and increasing the operational expenditure for production facilities. Furthermore, the removal of trace metal residues from the final active pharmaceutical ingredient (API) is a costly and time-consuming purification step that can drastically impact overall yield and lead time. The reliance on precious metals also exposes supply chains to geopolitical volatility and price fluctuations, creating uncertainty for procurement managers tasked with budget forecasting. Additionally, many traditional chiral auxiliaries are stoichiometric rather than catalytic, generating substantial chemical waste and complicating environmental compliance efforts. These cumulative inefficiencies create bottlenecks that hinder the rapid scale-up of new drug candidates from the laboratory to commercial production volumes.
The Novel Approach
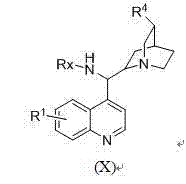
The innovative methodology described in the patent overcomes these hurdles by utilizing a one-step synthesis reaction between a cinchona alkaloid-derived primary amine and phosphoric acid acyl chloride. This streamlined approach eliminates the need for complex multi-step ligand synthesis, significantly reducing the raw material costs and processing time required to generate the catalyst. The resulting bifunctional catalyst possesses a unique structural architecture that combines a Lewis basic nitrogen center with a hydrogen-bond donating phosphoamide group, enabling dual activation of substrates. This dual activation mechanism allows for milder reaction conditions, often operating effectively at ambient or slightly reduced temperatures without the need for inert atmosphere protection. The simplicity of the synthesis protocol means that production can be scaled up rapidly using standard chemical processing equipment, thereby enhancing supply chain reliability. By shifting from metal-dependent to organocatalytic systems, manufacturers can achieve substantial cost savings while simultaneously improving the environmental footprint of their chemical processes.
Mechanistic Insights into Phosphoric Amide Bifunctional Catalysis
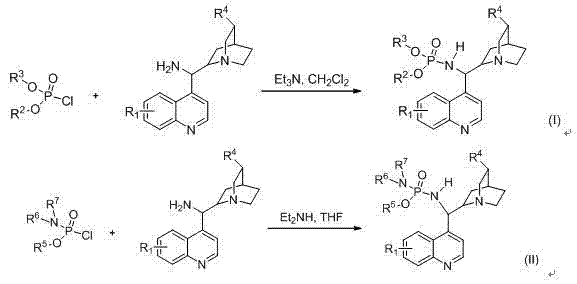
The core efficacy of this catalyst system lies in its sophisticated bifunctional activation mechanism, which orchestrates the spatial arrangement of reactants to favor a specific enantiomeric outcome. The cinchona alkaloid backbone provides a rigid chiral environment that dictates the facial selectivity of the nucleophilic attack, while the phosphoric acid amide moiety acts as a precise hydrogen-bond donor to activate the electrophile. This cooperative interaction lowers the activation energy of the transition state, facilitating reactions that might otherwise be sluggish or non-selective under standard conditions. The presence of the N-H bond in the phosphoamide group is particularly critical, as its acidity can be finely tuned by modifying the Rx substituent, allowing chemists to optimize the catalyst for specific substrate classes. This tunability is a powerful feature for R&D teams, as it enables the customization of catalytic properties without redesigning the entire molecular scaffold. The stability of the catalyst towards air and moisture further ensures that this mechanistic efficiency is maintained throughout the duration of the reaction, minimizing deactivation pathways that often plague sensitive catalytic systems.
Impurity control is inherently enhanced by the high selectivity of this catalytic system, which minimizes the formation of unwanted diastereomers and byproducts. The rigorous stereochemical control exerted by the catalyst ensures that the major diastereomer is produced with high diastereomeric ratios, often exceeding 20:1 in optimized conditions. This high level of selectivity simplifies downstream purification processes, as fewer chromatographic steps are required to isolate the desired product to pharmaceutical grade standards. The reduction in side reactions also means that raw material utilization is maximized, contributing to a more atom-economical process overall. For quality assurance teams, this translates to a more consistent impurity profile across different batches, reducing the risk of regulatory delays during drug filing. The ability to achieve greater than 90% ee values consistently demonstrates the robustness of the catalytic cycle, providing a reliable foundation for the synthesis of complex chiral building blocks required in modern medicine.
How to Synthesize Phosphoric Acid Amide Catalyst Efficiently
The synthesis protocol outlined in the patent is designed for operational simplicity, allowing for the rapid generation of the catalyst using widely available reagents and standard laboratory techniques. The process begins with the preparation of the chiral primary amine derivative, which serves as the foundational chiral pool material for the entire catalytic system. This precursor is then reacted with the appropriate phosphoric acid acyl chloride in the presence of a base such as triethylamine or sodium carbonate, facilitating the formation of the phosphoamide bond. The reaction proceeds smoothly over a broad temperature range, offering flexibility in process optimization depending on the specific reactivity of the substrates involved. Detailed standardized synthesis steps see the guide below.
- Prepare the chiral cinchona alkaloid derivative primary amine precursor through amination.
- React the amine with phosphoric acid acyl chloride in the presence of a base like triethylamine.
- Purify the resulting bifunctional catalyst using standard column chromatography techniques.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic technology offers profound advantages for procurement and supply chain management by fundamentally altering the cost structure of chiral synthesis. The reliance on cinchona alkaloids, which are naturally abundant and commercially available in large quantities, ensures a stable and predictable supply of raw materials that is not subject to the same volatility as precious metals. The one-step synthesis of the catalyst itself reduces the manufacturing lead time significantly, allowing suppliers to respond more agilely to fluctuating market demands. By eliminating the need for expensive transition metals and the associated removal processes, the overall cost of goods sold (COGS) for the final intermediate is drastically simplified. This cost efficiency can be passed down the supply chain, providing a competitive edge for pharmaceutical companies looking to optimize their production budgets. Furthermore, the robustness of the catalyst reduces the risk of batch failures due to sensitivity issues, ensuring a more consistent flow of materials to downstream processing units.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive scavenging resins and rigorous metal testing, leading to substantial cost savings in the purification phase. The use of inexpensive solvents and bases further contributes to a leaner manufacturing budget, while the high turnover number of the catalyst ensures that minimal amounts are needed to drive the reaction to completion. This efficiency translates directly into improved margins for the production of high-purity pharmaceutical intermediates, making the process economically viable even for lower-margin generic drug applications. The simplified workflow also reduces labor costs associated with complex handling procedures, allowing technical staff to focus on higher-value optimization tasks.
- Enhanced Supply Chain Reliability: The stability of the catalyst towards air and moisture means that storage and transportation requirements are less stringent, reducing the risk of degradation during logistics. This robustness allows for larger batch sizes to be produced and stored without the need for specialized inert atmosphere containment, facilitating better inventory management. The availability of raw materials from established natural product supply chains ensures that production can be sustained over long periods without interruption. This reliability is crucial for maintaining continuous manufacturing operations and meeting the strict delivery schedules demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: The synthetic method is inherently scalable, as it avoids exothermic hazards and complex pressure requirements often associated with metal-catalyzed hydrogenations or cross-couplings. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, minimizing the costs associated with waste disposal and treatment. The atom economy of the reaction is high, ensuring that the majority of input materials are incorporated into the final product rather than lost as byproducts. This green chemistry profile enhances the corporate sustainability metrics of the manufacturer, appealing to environmentally conscious stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology in industrial settings. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance. Understanding these details is essential for technical teams evaluating the feasibility of integrating this catalyst into existing production lines. The information covers stability, tunability, and performance metrics that are critical for process validation.
Q: What are the stability characteristics of this phosphoric acid amide catalyst?
A: The catalyst exhibits high stability towards both water and air, allowing for easier handling and storage compared to sensitive metal-based alternatives.
Q: Can the acidity of the catalyst be tuned for specific reactions?
A: Yes, the acidity of the N-H group in the phosphoamide moiety can be conveniently regulated by modifying the Rx substituent group.
Q: What level of enantioselectivity can be achieved with this system?
A: Applications in asymmetric reactions have demonstrated greater than 90% ee values with high diastereoselectivity ratios.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphoric Acid Amide Catalyst Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely qualified to adapt the synthetic routes described in patent CN102309984B to meet the specific needs of your drug development pipeline. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of catalyst meets the highest industry standards for performance and consistency. Our commitment to quality ensures that the chiral integrity of your intermediates is preserved throughout the manufacturing process, providing a solid foundation for your final API synthesis.
We invite you to engage with our technical procurement team to discuss how this advanced catalytic technology can optimize your current manufacturing processes. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this organocatalytic system. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Our goal is to be your strategic partner in achieving efficient, scalable, and cost-effective chemical synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
