Advanced Synthesis of Water-Soluble Nitrogenous Curcumin Analogues for Oncology Applications
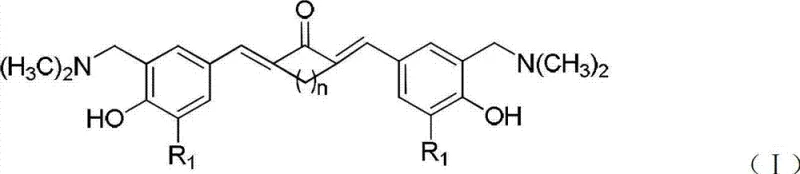
The pharmaceutical industry has long recognized the therapeutic potential of curcumin, yet its clinical translation has been severely hindered by intrinsic physicochemical limitations. Patent CN103012175A presents a groundbreaking solution by disclosing a novel class of nitrogen-containing substituent curcumin analogues that effectively resolve the issues of poor water solubility and instability associated with the natural product. By strategically modifying the central heptenedione bridge into an unsaturated monoketone chain and introducing dimethylaminomethyl groups onto the aromatic rings, this technology creates compounds with dramatically enhanced druggability. These structural alterations not only preserve the potent anti-tumor activity inherent to the curcumin scaffold but also enable the formation of pharmaceutically acceptable salts, thereby unlocking new possibilities for oncology drug development. For research and development teams seeking to advance anti-cancer pipelines, this patent offers a robust chemical platform that bridges the gap between natural product efficacy and modern pharmaceutical requirements.
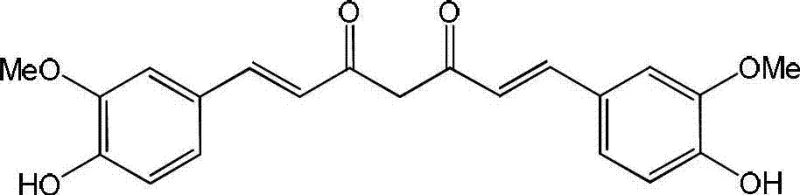
Traditional methods of utilizing curcumin rely heavily on direct extraction or simple derivatization, which often fail to address the fundamental stability issues of the beta-diketone moiety. Natural curcumin suffers from rapid degradation in physiological conditions and extremely low aqueous solubility, typically less than 1 mg/ml, which necessitates complex formulation strategies that increase manufacturing costs and complicate regulatory approval. Furthermore, previous attempts to modify the curcumin structure often focused solely on enhancing cytotoxicity without adequately addressing solubility, leading to candidates that were potent in vitro but failed in vivo due to poor pharmacokinetics. The conventional reliance on the unstable heptenedione bridge means that supply chains for high-quality curcumin derivatives are often plagued by batch-to-batch variability and short shelf-life, creating significant risks for long-term drug development projects.
In stark contrast, the novel approach detailed in this patent fundamentally re-engineers the molecular architecture to prioritize both stability and solubility without sacrificing biological activity. By replacing the labile heptenedione linker with a more robust unsaturated monoketone bridge, the chemical integrity of the molecule is significantly reinforced against hydrolytic degradation. Simultaneously, the introduction of basic amine functionalities via the Mannich reaction provides a handle for salt formation, a critical strategy for improving aqueous solubility. This dual-modification strategy results in compounds that demonstrate water solubility improvements of over 3000-fold compared to natural curcumin, as evidenced by solubility tests reaching concentrations above 300 mg/ml. This transformative shift allows for the creation of high-purity pharmaceutical intermediates that are far more manageable in standard manufacturing environments, reducing the need for exotic solvents and complex delivery systems.
Mechanistic Insights into Aldol Condensation and Mannich Reaction Sequence
The synthetic pathway relies on a precise two-stage functionalization process that begins with the construction of the core carbon skeleton through aldol condensation. In the first stage, substituted p-hydroxybenzaldehydes react with cyclic or acyclic ketones, such as acetone, cyclopentanone, or cyclohexanone, under acidic conditions to form the unsaturated monoketone intermediates. This Claisen-Schmidt type condensation is highly selective, favoring the formation of the trans-alkene geometry which is crucial for maintaining the planar conformation required for effective intercalation with biological targets. The reaction conditions are optimized to minimize side reactions such as polymerization or self-condensation of the aldehyde, ensuring that the yield of the desired enone intermediate remains high, typically ranging from 65% to 75% depending on the specific ketone used. This step establishes the rigid backbone that mimics the spatial arrangement of natural curcumin while eliminating the chemically vulnerable beta-diketone unit.
Following the skeleton construction, the second critical phase involves the introduction of the solubility-enhancing groups via a Mannich reaction. The intermediate enones are treated with dimethylamine and formaldehyde in a methanol solvent system at elevated temperatures, facilitating the electrophilic substitution at the ortho-position relative to the phenolic hydroxyl group. This reaction installs the dimethylaminomethyl moiety, which serves as a basic center capable of protonation. The mechanism proceeds through the formation of an iminium ion intermediate, which is then attacked by the electron-rich aromatic ring. Subsequent salt formation with acids like hydrochloric acid, sulfuric acid, or organic acids such as tartaric acid converts the free base into a stable crystalline salt. This final step is pivotal as it not only locks the molecule into a stable ionic lattice but also drastically alters the polarity of the compound, enabling rapid dissolution in aqueous media and enhancing its overall stability profile against oxidative degradation.
How to Synthesize Nitrogenous Curcumin Analogues Efficiently
The synthesis of these advanced anti-tumor intermediates follows a streamlined protocol designed for reproducibility and scalability, beginning with the preparation of the unsaturated ketone backbone. The process requires careful control of stoichiometry and temperature during the initial condensation to ensure high purity of the intermediate before proceeding to the amination step. Once the core structure is established, the Mannich reaction is performed under reflux conditions to drive the equilibrium towards the desired amine-substituted product. The final isolation involves a simple acid-base workup where the addition of a pharmaceutically acceptable acid induces precipitation of the target salt, allowing for easy filtration and drying. Detailed standardized synthesis steps are provided in the guide below to ensure consistent quality across production batches.
- Perform aldol condensation between substituted p-hydroxybenzaldehyde and cyclic or acyclic ketones to form the unsaturated monoketone bridge intermediate.
- Conduct a Mannich reaction on the intermediate using dimethylamine and formaldehyde in methanol to introduce the dimethylaminomethyl substituent.
- React the resulting base compound with pharmaceutically acceptable acids such as hydrochloric acid or sulfuric acid to precipitate the final stable salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits regarding cost efficiency and material availability. The starting materials, primarily substituted hydroxybenzaldehydes and common ketones like acetone or cyclohexanone, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This abundance of raw materials ensures a stable supply chain that is resilient to market fluctuations, unlike rare natural extracts which are subject to agricultural variability and seasonal harvest constraints. Furthermore, the synthetic process avoids the use of expensive transition metal catalysts or hazardous reagents, relying instead on straightforward acid-base chemistry that can be implemented in standard multipurpose reactors. This simplicity translates directly into lower operational expenditures and reduced waste treatment costs, making the commercial production of these intermediates economically attractive for large-scale manufacturing.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and expensive catalysts significantly lowers the overall cost of goods sold. Since the final product precipitates directly from the reaction mixture upon salt formation, the need for energy-intensive chromatographic separations is minimized, leading to substantial cost savings in downstream processing. Additionally, the high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted, further optimizing the financial footprint of the manufacturing process.
- Enhanced Supply Chain Reliability: By shifting from a natural extraction model to a fully synthetic route, manufacturers gain complete control over production schedules and output volumes. The synthetic pathway is not limited by the growth cycles of Curcuma longa plants, allowing for continuous year-round production to meet fluctuating demand. This reliability is crucial for pharmaceutical partners who require guaranteed continuity of supply to support clinical trials and eventual commercial launch without interruption.
- Scalability and Environmental Compliance: The reaction conditions utilize common organic solvents like methanol and dichloromethane, which are well-understood in terms of recovery and recycling, facilitating compliance with environmental regulations. The process is inherently scalable from gram-scale laboratory synthesis to multi-ton industrial production without requiring specialized high-pressure or high-temperature equipment. This ease of scale-up reduces the capital investment required for technology transfer and accelerates the time to market for new drug candidates based on this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, activity, and formulation potential of these nitrogenous curcumin analogues. Understanding these aspects is essential for evaluating their suitability for specific oncology applications and formulation strategies. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation.
Q: How does this patent address the low bioavailability of natural curcumin?
A: The patent overcomes natural curcumin's limitations by replacing the unstable heptenedione bridge with an unsaturated monoketone chain and introducing basic dimethylaminomethyl groups. This structural modification allows for the formation of pharmaceutically acceptable salts, increasing water solubility by over 3000 times compared to the parent compound.
Q: What represents the key innovation in the synthetic route described in CN103012175A?
A: The key innovation lies in the sequential application of Claisen-Schmidt condensation followed by a Mannich reaction. This approach efficiently constructs the core pharmacophore while simultaneously installing solubility-enhancing amine functionalities, streamlining the production of high-purity anti-tumor candidates.
Q: Are the resulting curcumin analogues stable in aqueous solutions?
A: Yes, stability tests indicate that these nitrogenous analogues exhibit significantly superior stability in aqueous buffers compared to natural curcumin. While curcumin degrades rapidly within hours, the patented analogues maintain their structural integrity and UV absorption profiles over extended periods, ensuring reliable performance in biological assays.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrogenous Curcumin Analogues Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our rigorous QC labs and stringent purity specifications guarantee that every batch of nitrogenous curcumin analogues meets the highest international standards for pharmaceutical intermediates. We understand the critical nature of oncology drug development and are committed to providing a supply chain that supports your regulatory filings with consistent, high-quality material. Our technical team is ready to collaborate on process optimization to further enhance yield and reduce impurities, aligning with your specific project goals.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. By engaging with us, you can access specific COA data and route feasibility assessments that demonstrate the viability of integrating these advanced intermediates into your pipeline. Let us help you leverage this innovative technology to accelerate the development of next-generation anti-tumor therapies while optimizing your manufacturing economics.
