Advanced Chiral Resolution Technology for Sitagliptin Intermediates: Scaling High-Purity Production
Advanced Chiral Resolution Technology for Sitagliptin Intermediates: Scaling High-Purity Production
The global demand for Type II diabetes treatments continues to surge, driving the need for efficient, scalable manufacturing of key active pharmaceutical ingredients (APIs) like Sitagliptin. Patent CN102363599A introduces a pivotal advancement in the production of the critical chiral intermediate, (R)-3-amino-4-(2,4,5-trifluorophenyl)benzyl butyrate. This technology addresses the longstanding challenges of achieving high optical purity without relying on prohibitively expensive chiral catalysts. By shifting the paradigm from asymmetric catalysis to a highly optimized diastereomeric salt resolution process, this method offers a robust pathway for industrial-scale production. The structural complexity of Sitagliptin, as illustrated below, necessitates precise stereochemical control to ensure therapeutic efficacy and safety.
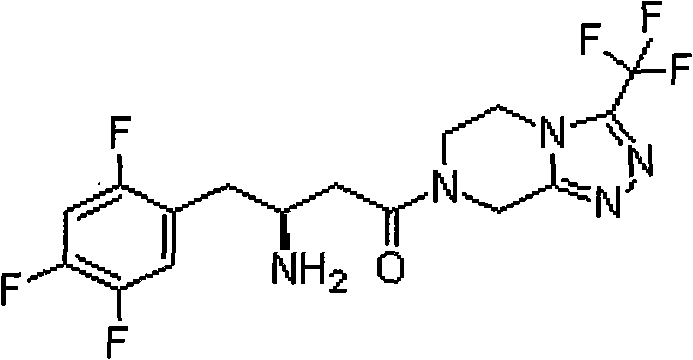
This patent specifically targets the preparation of the benzyl ester derivative, a crucial building block that overcomes the limitations of earlier methyl or ethyl ester variants. The innovation lies not just in the molecule itself, but in the holistic process design that integrates solvent recyclability and high-yield crystallization. For R&D directors and procurement strategists, understanding this shift from catalytic dependency to resolution efficiency is key to securing a stable supply chain for next-generation anti-diabetic medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-amino acid derivatives for Sitagliptin has relied heavily on direct chiral reduction or the resolution of simpler methyl and ethyl esters. However, these conventional pathways present significant bottlenecks for commercial manufacturing. Direct chiral reduction often requires precious metal catalysts or specialized ligands that drive up the cost of goods sold (COGS) dramatically, creating pressure on the final medicine price. Furthermore, prior art regarding methyl and ethyl esters, such as those reported in WO2010131025 and WO2010009630, indicates substantial difficulties in achieving effective separation. When using resolving agents like D-tartaric acid or camphorsulfonic acid with these smaller esters, the salification process is often inefficient, leading to poor crystal formation and low enantiomeric excess. The inability to effectively separate enantiomers at the ester stage forces manufacturers into costly recycling loops or results in significant yield loss, rendering these methods suboptimal for large-scale industrial application.
The Novel Approach
The methodology disclosed in CN102363599A represents a strategic pivot by utilizing the benzyl ester variant, designated as compound (2), as the substrate for resolution. This approach leverages the steric and electronic properties of the benzyl group to facilitate superior interaction with chiral resolving agents. Specifically, the process employs D-di-p-toluoyl tartrate in an acetone solvent system, a combination that was previously unreported for this specific benzyl ester configuration. Unlike the problematic methyl and ethyl analogs, the benzyl ester forms a stable, highly crystalline salt with the resolving agent, allowing for efficient precipitation and filtration. This novel route eliminates the need for expensive transition metal catalysts entirely, replacing them with readily available organic acids. The result is a process that is not only chemically robust but also economically superior, as it utilizes common solvents like acetone which can be easily recovered and reused, thereby minimizing waste and operational expenditure.
Mechanistic Insights into Diastereomeric Salt Resolution
The core of this technology rests on the principles of diastereomeric salt formation, a classical yet highly effective technique for chiral separation. The process begins with the synthesis of the racemic 3-amino-4-(2,4,5-trifluorophenyl)benzyl butyrate. As depicted in the synthetic scheme below, the racemate is constructed through a multi-step sequence involving the condensation of 2,4,5-trifluorophenylacetic acid with a Meldrum's acid derivative, followed by alcoholysis and reductive amination. This pathway ensures the availability of the racemic starting material in high quantities before the critical resolution step occurs.
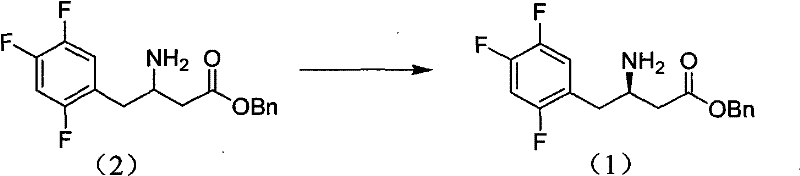
Once the racemic amine is obtained, the chiral discrimination occurs upon addition of the resolving agent, D-di-p-toluoyl tartrate. In the acetone medium, the (R)-enantiomer of the amine preferentially binds with the chiral acid to form a less soluble diastereomeric salt compared to the (S)-enantiomer salt. This difference in solubility is the driving force for the separation. The patent highlights that the molar ratio of the resolving agent is critical, typically ranging from 0.5 to 1.5 equivalents, with an optimal range of 0.8 to 1.2 equivalents to maximize yield without compromising purity. The choice of acetone as the solvent is mechanistically significant; it provides the perfect polarity balance to keep the unwanted (S)-enantiomer in solution while precipitating the desired (R)-salt. Following filtration, the pure salt is treated with a mild base, such as sodium bicarbonate or sodium carbonate, in a biphasic system (e.g., ethyl acetate and water). This neutralization step liberates the free amine base, yielding the target (R)-3-amino-4-(2,4,5-trifluorophenyl)benzyl butyrate with an enantiomeric excess (ee) exceeding 99% and chemical purity greater than 99%, ready for downstream coupling reactions.
How to Synthesize (R)-3-amino-4-(2,4,5-trifluorophenyl)benzyl butyrate Efficiently
The synthesis of this high-value intermediate requires strict adherence to the optimized parameters regarding solvent volume, temperature control, and stoichiometry to ensure consistent quality. The process is designed to be operationally simple, avoiding complex equipment or hazardous conditions, which facilitates easy technology transfer from the lab to the pilot plant. The following guide outlines the critical stages of the resolution process as validated by the patent examples, ensuring that the final product meets the rigorous specifications required for API synthesis.
- Synthesize the racemic 3-amino-4-(2,4,5-trifluorophenyl)benzyl butyrate via condensation of 2,4,5-trifluorophenylacetic acid followed by reduction.
- Perform chiral resolution by reacting the racemate with D-di-p-toluoyl tartrate in acetone to form the diastereomeric salt.
- Isolate the resolved salt through filtration and washing, then liberate the free base using sodium carbonate to obtain the pure (R)-enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resolution technology translates directly into enhanced operational stability and cost efficiency. By moving away from proprietary chiral catalysts to commodity chemicals like tartaric acid derivatives and acetone, the supply chain becomes significantly more resilient to market fluctuations. The elimination of heavy metal catalysts also simplifies the regulatory landscape, reducing the burden of residual metal testing and clearance procedures which often delay batch release. Furthermore, the ability to recycle the primary solvent, acetone, creates a closed-loop system that drastically reduces raw material consumption and waste disposal costs, aligning with modern green chemistry initiatives and environmental compliance standards.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the substitution of expensive chiral catalysts with cost-effective resolving agents. Traditional asymmetric hydrogenation or enzymatic routes often require catalysts that constitute a significant portion of the variable cost. By utilizing D-di-p-toluoyl tartrate, a bulk chemical, the direct material cost is significantly reduced. Additionally, the high purity achieved (>99%) minimizes the need for additional purification steps such as chromatography or repeated recrystallizations, which are both time-consuming and solvent-intensive. The process yield, while subject to the theoretical 50% limit of resolution, is optimized through the efficient recovery of the mother liquor or potential racemization of the unwanted isomer, ensuring that the overall process economics remain highly competitive compared to catalytic alternatives.
- Enhanced Supply Chain Reliability: Reliance on specialized chiral ligands often exposes manufacturers to single-source supplier risks and long lead times. In contrast, the reagents required for this resolution method—acetone, sodium carbonate, and tartaric acid derivatives—are globally available commodity chemicals with robust supply networks. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Moreover, the simplicity of the unit operations (dissolution, crystallization, filtration) means that the process can be executed in standard multipurpose reactors found in most fine chemical facilities, eliminating the need for specialized high-pressure hydrogenation equipment. This flexibility allows for rapid scale-up and the ability to shift production between different manufacturing sites if necessary, ensuring continuous supply continuity for downstream API customers.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its reliance on crystallization, a unit operation that scales linearly and predictably from grams to tons. The use of acetone, a Class 3 solvent with low toxicity, simplifies environmental, health, and safety (EHS) management compared to chlorinated solvents or high-boiling polar aprotic solvents. The patent explicitly notes the recyclability of the solvent, which not only lowers costs but also reduces the facility's environmental footprint by minimizing volatile organic compound (VOC) emissions. The absence of heavy metals in the process stream further simplifies wastewater treatment and reduces the generation of hazardous solid waste. These factors collectively make the technology highly attractive for manufacturers aiming to meet stringent international environmental regulations while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution technology. These insights are derived directly from the experimental data and process descriptions within the patent, providing clarity on the feasibility and advantages of this specific manufacturing route for Sitagliptin intermediates.
Q: Why is the benzyl ester preferred over methyl or ethyl esters for this resolution?
A: According to the patent background, methyl and ethyl esters exhibit poor resolution efficiency with common resolving agents like tartaric acid derivatives. The benzyl ester structure provides superior crystallization properties and higher enantiomeric excess when paired with D-di-p-toluoyl tartrate.
Q: What are the advantages of using acetone as the resolution solvent?
A: Acetone is preferred over acetonitrile-water mixtures because it allows for easy solvent recovery and recycling, significantly reducing raw material costs. Additionally, acetone facilitates the formation of high-purity salts with excellent filtration characteristics.
Q: What level of optical purity can be achieved with this method?
A: The described process consistently achieves an enantiomeric excess (ee) value of greater than 99% and chemical purity exceeding 99%, making it suitable for direct use in subsequent API synthesis steps without further purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
The technological breakthroughs detailed in CN102363599A underscore the potential for producing high-quality Sitagliptin intermediates with exceptional purity and efficiency. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical pharmaceutical building blocks. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels that meet or exceed global pharmacopoeia standards. We understand that in the competitive landscape of diabetes care, the reliability of your supply chain is paramount, and we are committed to being a partner that delivers both technical excellence and commercial stability.
We invite potential partners to engage with our technical procurement team to discuss how this optimized resolution process can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your API manufacturing operations.
