Optimizing Barbiturate Intermediate Production: A Technical Analysis of the Novel Methoxide Cyclization Route
The pharmaceutical industry's demand for high-quality sedative and anticonvulsant intermediates necessitates robust, scalable, and economically viable synthetic routes. Patent CN111606862A introduces a significant technological advancement in the preparation and purification of 5-ethyl-5-isoamylmalonyl urea, a critical precursor in the manufacture of barbiturate drugs. This innovation addresses longstanding inefficiencies in the classical condensation of diethyl ethylisoamylmalonate with urea, offering a pathway that is not only chemically superior but also commercially advantageous for large-scale manufacturing. By shifting the reaction medium and optimizing the base catalyst system, the disclosed method achieves a stable process that eliminates the need for stringent concentration controls typically associated with alkoxide reagents. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this patented methodology is essential for securing a supply chain that balances cost-efficiency with rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-ethyl-5-isoamylmalonyl urea has relied heavily on the use of sodium ethoxide dissolved in ethanol, a method fraught with operational complexities and yield limitations. In these conventional processes, the reaction concentration of sodium ethoxide must be maintained within a very narrow and specific range to prevent side reactions, making the operation cumbersome and highly sensitive to minor deviations in reagent quality. A critical failure point in the traditional route is the inability of the reaction to proceed to full completion, often leaving behind significant quantities of unreacted starting materials that manifest as difficult-to-handle oily residues. This incomplete conversion necessitates arduous post-processing steps to separate the oily intermediates from the desired product, leading to poor product appearance and necessitating the use of large amounts of activated carbon for decolorization. Consequently, the traditional method generates substantial three-waste emissions, incurs high energy consumption due to prolonged reaction times, and ultimately results in a low overall yield that negatively impacts the cost structure of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast to the legacy ethanol-based systems, the novel approach detailed in the patent utilizes a sodium methoxide methanol solution, fundamentally altering the reaction kinetics and thermodynamics to favor product formation. This method introduces a strategic pre-treatment step where ethyl acetate is added to the methoxide solution and heated to reflux, effectively scavenging free bases that could otherwise catalyze unwanted degradation pathways. The reaction is ingeniously coupled with a distillation process where the temperature is gradually raised from 80°C to 100°C over a period of 2 to 3 hours, continuously removing the generated alcohol to drive the equilibrium towards the cyclized product according to Le Chatelier's principle. This dynamic removal of byproducts ensures that the reaction proceeds to near-completion, avoiding the formation of stubborn oily intermediates and facilitating the direct crystallization of the sodium salt. The result is a process that is operationally simple, requires shorter reaction times, and produces a crude product with significantly improved physical properties, thereby streamlining the downstream purification workflow.
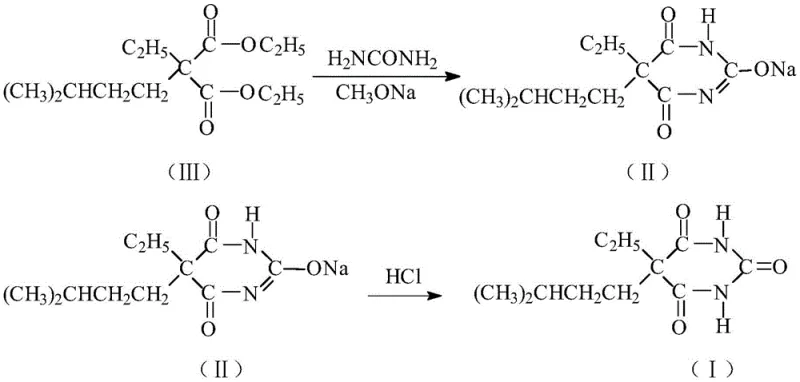
Mechanistic Insights into Sodium Methoxide-Mediated Cyclization
The core chemical transformation in this synthesis involves the nucleophilic attack of urea on the ester groups of diethyl ethylisoamylmalonate, facilitated by the strong basicity of the sodium methoxide catalyst. In the presence of the methoxide ion, the urea molecule is deprotonated to form a nucleophilic species that attacks the carbonyl carbon of the ester, initiating the ring-closing condensation. The choice of methanol as the solvent, rather than ethanol, appears to offer kinetic advantages, potentially due to differences in solvation effects on the transition state or the volatility of the byproduct alcohol which aids in its removal during the distillation phase. The specific molar ratio of the diester to sodium methoxide and urea, optimized at approximately 1:2.0-2.5:1.5-2.0, ensures that there is sufficient base to drive the double condensation required to form the six-membered pyrimidine ring without causing excessive hydrolysis of the ester groups. This precise stoichiometric balance is critical for minimizing the formation of mono-condensed intermediates or ring-opened byproducts that would complicate the purification process.
Furthermore, the mechanism of impurity control is deeply embedded in the thermal profile and workup conditions of the new process. By conducting the reaction under distillation conditions at 80-100°C, the system effectively removes methanol and ethanol azeotropes, preventing the reverse reaction and locking the product into the cyclic ureide structure. Upon completion, the reaction mass is cooled to 20-30°C and dissolved in ice-cold water (0-5°C), a step that exploits the differential solubility of the sodium salt intermediate versus organic impurities. The subsequent acidification to pH 3-4 triggers the precipitation of the free acid form of the barbiturate, while the low temperature of 10-15°C during centrifugation ensures that the crystal lattice forms tightly, excluding soluble impurities from the solid phase. This meticulous control over pH and temperature during the isolation phase is what allows the process to effectively reduce impurity content and improve the overall quality of the target product without resorting to excessive chromatographic purification.
How to Synthesize 5-Ethyl-5-Isoamylmalonyl Urea Efficiently
The implementation of this synthesis route requires careful attention to the sequence of reagent addition and thermal management to replicate the high yields reported in the patent literature. Operators must first ensure the neutralization of free bases in the sodium methoxide solution using ethyl acetate before introducing the urea and diester substrates, as this preconditioning step is vital for preventing discoloration and side reactions. The distillation phase must be monitored closely to maintain the internal temperature ramp from 80°C to 100°C over the designated 2 to 3-hour window, ensuring that the alcohol byproducts are removed at a rate that matches their formation. Following the reaction, the quenching and acidification steps must be performed rapidly and at low temperatures to maximize the recovery of the crystalline product. For a comprehensive breakdown of the specific equipment setup and safety protocols required for this transformation, please refer to the standardized synthesis guide below.
- React alpha-ethyl-alpha-isoamyl diethyl malonate with urea in a sodium methoxide methanol solution, utilizing ethyl acetate to scavenge free bases, while distilling off alcohol at 80-100°C to drive the cyclization.
- Cool the reaction mass, dissolve the sodium salt intermediate in ice-cold water (0-5°C), and acidify to pH 3-4 to precipitate the crude 5-ethyl-5-isoamylmalonyl urea.
- Purify the wet crude product via recrystallization using a 40-50% aqueous ethanol solution, employing activated carbon decolorization at 78°C followed by cooling crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this novel methoxide-based process offers substantial benefits that directly address the pain points of cost volatility and supply reliability in the pharmaceutical intermediate market. The ability to use industrial-grade sodium methoxide without the need for precise concentration adjustments significantly reduces the raw material qualification burden and allows for the procurement of cheaper, bulk-grade reagents. This flexibility translates into a more resilient supply chain, as manufacturers are not dependent on specialized, high-purity reagent batches that may have longer lead times or higher price points. Moreover, the simplification of the post-reaction workup, characterized by the elimination of oily residue separation and reduced reliance on activated carbon, lowers the operational expenditure associated with waste disposal and consumable materials. These cumulative efficiencies create a cost structure that is inherently more competitive, allowing suppliers to offer better pricing stability to their downstream partners in the API manufacturing sector.
- Cost Reduction in Manufacturing: The elimination of complex separation steps for unreacted oily intermediates drastically reduces the labor and energy costs associated with the purification phase. By avoiding the need for extensive decolorization treatments and minimizing the loss of product during difficult filtrations, the overall material efficiency of the process is significantly enhanced. This improvement in yield, combined with the reduced consumption of auxiliary materials like activated carbon and solvents, leads to a substantial decrease in the cost of goods sold, providing a clear economic advantage over the traditional ethoxide-based methodology.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate variations in reagent concentration and utilize common industrial solvents like methanol and ethyl acetate, ensures consistent batch-to-batch performance. This reliability minimizes the risk of production delays caused by failed batches or out-of-specification intermediates, thereby securing a steady flow of material for downstream API synthesis. For supply chain heads, this means reduced safety stock requirements and a lower risk of disruption, as the process is less sensitive to minor fluctuations in raw material quality that might otherwise halt production.
- Scalability and Environmental Compliance: The process generates significantly less three-waste emission compared to conventional methods, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The simplified workflow, involving standard unit operations such as distillation and crystallization, is inherently easier to scale from pilot plant to commercial production volumes without encountering the mixing or heat transfer issues often seen with viscous oily intermediates. This ease of scale-up ensures that the technology can meet growing market demand without requiring disproportionate increases in capital investment or waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 5-ethyl-5-isoamylmalonyl urea, derived directly from the specific advancements detailed in the patent documentation. These insights are intended to clarify the operational benefits and quality assurances provided by this modernized synthetic route. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing manufacturing pipelines.
Q: Why is the sodium methoxide route superior to the traditional sodium ethoxide method for this synthesis?
A: The traditional sodium ethoxide method often suffers from incomplete reactions leading to oily intermediates that are difficult to separate, requiring extensive purification. The novel sodium methoxide route described in patent CN111606862A utilizes a specific distillation protocol that drives the reaction to completion more effectively, resulting in a solid crude product that is easier to handle and purify, significantly reducing processing time and waste.
Q: How does this process impact the impurity profile of the final API intermediate?
A: By incorporating an initial step where ethyl acetate is added to the sodium methoxide solution to eliminate free bases prior to the main reaction, the process minimizes side reactions caused by alkaline impurities. Furthermore, the controlled acidification and specific recrystallization temperatures (0-5°C) ensure that organic impurities remain in the mother liquor, yielding a product with superior外观 (appearance) and reduced impurity content compared to conventional methods.
Q: Is this synthesis method scalable for industrial manufacturing?
A: Yes, the process is explicitly designed for industrial scalability. It tolerates industrial-grade sodium methoxide concentrations (29-31%) without requiring precise laboratory-grade adjustments. The operation involves standard unit operations such as distillation, filtration, and crystallization, which are easily transferable to large-scale reactors, ensuring consistent quality and supply continuity for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Ethyl-5-Isoamylmalonyl Urea Supplier
As the global pharmaceutical landscape evolves, the need for intermediates produced via efficient and compliant technologies has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies like the one described in CN111606862A to deliver superior value to our partners. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 5-ethyl-5-isoamylmalonyl urea meets the exacting standards required for hypnotic and anticonvulsant drug manufacturing.
We invite you to engage with our technical procurement team to discuss how our optimized production capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our supply chain. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and profitability of your final pharmaceutical products.
