Advanced Stereodivergent Synthesis of Polysubstituted Tetrahydro-γ-Carboline Derivatives for Pharmaceutical Applications
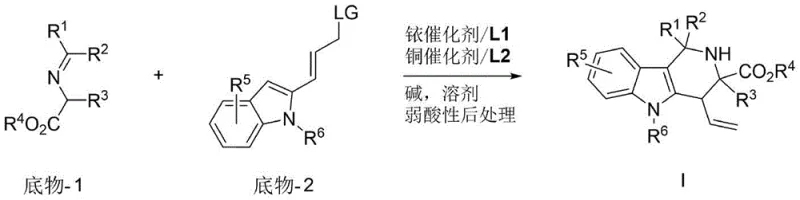
The pharmaceutical industry continuously seeks robust and versatile synthetic methodologies to access complex heterocyclic scaffolds, particularly those exhibiting significant biological activity in the central nervous system. Patent CN110669046A discloses a groundbreaking approach for the synthesis of polysubstituted tetrahydro-γ-carboline derivatives featuring multiple chiral centers through a novel copper and iridium synergistic catalyzed allylation and iso-Pictet–Spengler reaction. This technology represents a significant leap forward in medicinal chemistry, offering a streamlined pathway to construct highly functionalized indole alkaloids that are pivotal in the development of therapeutics for depression and Alzheimer's disease. The core innovation lies in the ability to generate structural complexity and stereochemical diversity simultaneously from readily available starting materials, specifically indole-2-derived allyl carbonates and amino acid-derived imines, under remarkably mild reaction conditions.
The strategic importance of this invention cannot be overstated given the pharmacological relevance of the tetrahydro-γ-carboline core. Traditional synthetic routes often struggle with controlling multiple stereocenters efficiently or require harsh conditions that limit functional group tolerance. In contrast, the methodology described in CN110669046A utilizes a dual catalytic system where an iridium complex and a copper complex operate in concert to facilitate bond formation with exceptional precision. This process not only simplifies the synthetic sequence but also enhances the overall efficiency of producing high-purity intermediates, making it an attractive option for reliable pharmaceutical intermediate supplier networks aiming to scale up production of neuroactive compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polychiral tetrahydro-γ-carboline frameworks has been fraught with challenges related to stereocontrol and reaction efficiency. Conventional Pictet-Spengler reactions often suffer from poor diastereoselectivity, necessitating tedious chromatographic separations to isolate the desired isomer, which drastically reduces overall material throughput and increases waste generation. Furthermore, many existing protocols rely on stoichiometric amounts of chiral auxiliaries or require cryogenic temperatures to achieve acceptable enantiomeric excess, imposing significant energy costs and operational complexities on the manufacturing process. The inability to easily access different stereoisomers from a single set of precursors also hampers the drug discovery phase, where evaluating the biological activity of all possible stereoisomers is critical for identifying the most potent and safe drug candidate.
The Novel Approach
The novel approach detailed in the patent overcomes these hurdles by employing a sophisticated Cu/Ir co-catalytic system that operates effectively at room temperature, typically between 15 and 35 degrees Celsius. This method leverages the unique reactivity of methyleneamine ylides and indole-derived allyl carbonates to forge carbon-carbon and carbon-nitrogen bonds in a tandem fashion. By merely switching the chirality of the ligands associated with the metal centers, chemists can access diverse stereoisomers with high fidelity, achieving diastereomeric ratios greater than 20:1 and enantiomeric excess values exceeding 99 percent. This level of control transforms the synthesis from a bottleneck into a flexible platform, enabling rapid exploration of chemical space while maintaining the rigorous purity standards required for clinical applications.
Mechanistic Insights into Cu/Ir Synergistic Catalyzed Allylation
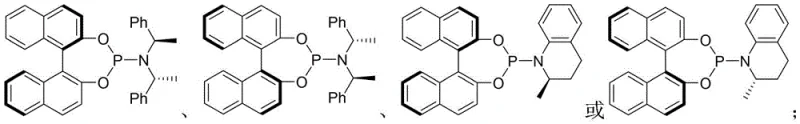
The mechanistic elegance of this transformation relies on the precise orchestration of two distinct catalytic cycles that converge to form the final product. The iridium catalyst, coordinated with a chiral phosphoramidite ligand such as those depicted in the structural formulas of L1, activates the allyl carbonate substrate through oxidative addition to generate a reactive pi-allyl iridium intermediate. This species serves as a soft electrophile that is poised for nucleophilic attack. Simultaneously, the copper catalyst, complexed with a chiral ferrocene-based P,N-ligand like L2, activates the imine substrate, potentially forming a copper-enolate or facilitating the generation of the methyleneamine ylide species. The synergy arises when the nucleophilic species generated by the copper cycle attacks the electrophilic allyl-iridium complex, establishing the first stereocenter with high fidelity dictated by the chiral environment of the iridium ligand.
Following the initial allylation, an intramolecular iso-Pictet-Spengler cyclization occurs, closing the ring to form the tetrahydro-γ-carboline core and establishing the second and third stereocenters. The choice of ligands is paramount in this process; for instance, using specific enantiomers of the BINOL-derived phosphoramidite ligands shown in the patent data ensures that the transition states are energetically differentiated enough to favor one stereoisomer overwhelmingly over others. This dual activation strategy minimizes background non-catalyzed reactions that could lead to racemic byproducts, thereby ensuring that the impurity profile remains clean and manageable. The compatibility of various bases, such as diisopropylethylamine or cesium carbonate, further fine-tunes the reaction kinetics, allowing for optimization based on the specific electronic nature of the substrates involved.
How to Synthesize Polysubstituted Tetrahydro-γ-Carboline Efficiently
Executing this synthesis requires careful attention to the preparation of the catalytic species and the maintenance of an inert atmosphere to prevent catalyst deactivation. The process begins with the in situ or pre-formation of the iridium and copper complexes by mixing the respective metal salts, such as [Ir(COD)Cl]2 and Cu(MeCN)4BF4, with their corresponding chiral ligands in dry, degassed solvents like tetrahydrofuran or dichloromethane. Once the active catalysts are ready, the substrates are combined in the reaction vessel along with a suitable base, and the mixture is stirred at ambient temperature for a period ranging from 12 to 24 hours. The reaction progress is typically monitored via thin-layer chromatography or HPLC, and upon completion, the mixture is quenched with a weak acid solution to neutralize the base and decompose any remaining reactive intermediates.
- Prepare the iridium and copper catalyst complexes by mixing the respective metal salts with chiral ligands L1 and L2 in organic solvents under inert atmosphere.
- Combine the indole-derived allyl carbonate and amino acid-derived imine substrates in a solvent such as dichloromethane with a base like DIPEA or Cs2CO3.
- Add the prepared catalyst systems to the reaction mixture and stir at room temperature (15-35°C) for 12-24 hours, followed by acidic quenching and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers substantial benefits by streamlining the manufacturing workflow and reducing dependency on specialized equipment. The ability to conduct the reaction at room temperature eliminates the need for expensive heating or cooling infrastructure, leading to significant energy savings and a lower carbon footprint for the production facility. Moreover, the high yields and selectivity reported in the patent data mean that less raw material is wasted, and the downstream purification burden is greatly reduced, which translates directly into lower cost of goods sold. The use of commercially available starting materials and standard solvents further enhances supply chain reliability, ensuring that production schedules can be met without the risk of delays associated with sourcing exotic reagents.
- Cost Reduction in Manufacturing: The elimination of extreme temperature requirements and the use of low catalyst loadings significantly reduce operational expenditures associated with energy consumption and precious metal recovery. The high stereoselectivity minimizes the need for costly chiral separation techniques like preparative chiral HPLC, which are often the most expensive step in the production of chiral pharmaceutical intermediates. Additionally, the robustness of the reaction across a wide range of substrates allows for a standardized manufacturing protocol that can be applied to multiple products, maximizing asset utilization and reducing validation costs.
- Enhanced Supply Chain Reliability: The reliance on stable and readily accessible reagents such as indole derivatives and amino acid esters ensures a consistent supply of raw materials, mitigating the risk of production stoppages due to material shortages. The mild reaction conditions also improve safety profiles by reducing the potential for thermal runaways or hazardous exotherms, facilitating easier regulatory approval and insurance underwriting. This stability allows for more predictable lead times and inventory management, enabling procurement teams to negotiate better terms with suppliers and maintain optimal stock levels.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving standard aqueous quenching and extraction, makes the process highly amenable to scale-up from gram to multi-kilogram quantities without significant re-engineering. The reduced generation of hazardous waste due to high atom economy and selectivity aligns with increasingly stringent environmental regulations, lowering disposal costs and enhancing the sustainability credentials of the manufacturing site. This scalability ensures that the technology can support both early-stage clinical trial material needs and eventual commercial production volumes seamlessly.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthetic methodology, providing clarity for R&D and technical procurement teams evaluating this technology for their pipelines. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to ensure accuracy and relevance to practical application scenarios.
Q: What are the key advantages of the Cu/Ir synergistic catalysis system?
A: The dual catalytic system allows for the simultaneous activation of both electrophilic and nucleophilic partners under mild conditions, achieving high stereoselectivity (ee ≥ 99%, dr > 20:1) and excellent yields (75-99%) without requiring extreme temperatures.
Q: Can this method produce multiple stereoisomers of the same compound?
A: Yes, by simply adjusting the combination of chiral ligands (L1 and L2) used in the catalytic system, this method enables the stereodivergent synthesis of various stereoisomers from the same set of starting materials, which is crucial for drug screening.
Q: What types of substituents are tolerated in this reaction?
A: The reaction exhibits broad substrate scope, tolerating various substituents on the aryl rings including halogens (Cl, Br), alkyl groups (Me, Et), alkoxy groups (OMe), and heterocycles, as well as different ester groups on the imine substrate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydro-γ-Carboline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is smooth and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and potency.
We invite you to collaborate with us to leverage this cutting-edge Cu/Ir catalytic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain resilience and drive down your overall development costs.
