Advanced Synthesis of 5,6,7-Trimethoxybaicalein Derivatives for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking robust synthetic routes for bioactive flavonoids that offer improved pharmacokinetic profiles over their natural counterparts. Patent CN102060832A discloses a highly efficient method for synthesizing novel 5,6,7-trimethoxybaicalein derivatives, specifically targeting modifications on the B-ring skeleton to enhance solubility and anticancer activity. Unlike traditional extraction methods which suffer from seasonal variability and low purity, this chemical synthesis approach utilizes readily available commodity chemicals such as substituted benzaldehydes and 3,4,5-trimethoxyphenol. The process is designed to overcome the inherent limitations of natural baicalein, particularly its poor solubility, by introducing specific substituents like alkyl, alkoxy, or nitro groups at the 4-prime position. This technological breakthrough provides a reliable foundation for developing next-generation therapeutic agents with optimized bioavailability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of baicalein and its analogues has relied heavily on extraction from the roots of Scutellaria baicalensis, a process fraught with significant supply chain and quality inconsistencies. Natural extraction yields are inherently low and subject to agricultural variables such as climate, soil composition, and harvest timing, leading to fluctuating market prices and unreliable delivery schedules for pharmaceutical manufacturers. Furthermore, isolating high-purity baicalein from the complex matrix of plant extracts requires extensive downstream processing, often involving multiple chromatography steps that generate substantial solvent waste and increase the overall cost of goods sold. From a medicinal chemistry perspective, natural baicalein exhibits limited water solubility, which severely restricts its formulation options and oral bioavailability, thereby limiting its clinical utility despite its promising in vitro pharmacological profile.
The Novel Approach
The methodology presented in the patent data introduces a streamlined four-step synthetic pathway that bypasses the unpredictability of natural sourcing entirely. By constructing the flavonoid skeleton from simple aromatic precursors, this approach allows for precise structural engineering of the B-ring, enabling the creation of derivatives with tailored physicochemical properties. The route employs a logical sequence of Perkin condensation, chlorination, acylation, and finally, an oxidative cyclization, each step optimized for high conversion rates and ease of purification. This synthetic flexibility not only ensures a consistent supply of high-purity intermediates but also opens the door to generating diverse libraries of analogues for structure-activity relationship studies. The ability to introduce various functional groups systematically makes this method superior for developing specialized pharmaceutical intermediates with enhanced therapeutic indices.
Mechanistic Insights into Iodine-Catalyzed Oxidative Cyclization
The cornerstone of this synthesis is the final cyclization step, where a chalcone intermediate is converted into the flavone core structure using molecular iodine in dimethyl sulfoxide. This transformation is mechanistically fascinating as it involves an oxidative intramolecular coupling that closes the heterocyclic C-ring. The iodine acts as a mild yet effective oxidant, facilitating the dehydrogenation required to establish the aromatic character of the pyrone ring without degrading sensitive functional groups on the B-ring. The use of DMSO as a solvent is critical, as it stabilizes the transition state and solubilizes the polar intermediates, ensuring a homogeneous reaction environment that promotes high yields. This specific catalytic system avoids the use of harsh heavy metal oxidants, aligning with modern green chemistry principles while maintaining the rigorous purity standards required for active pharmaceutical ingredients.
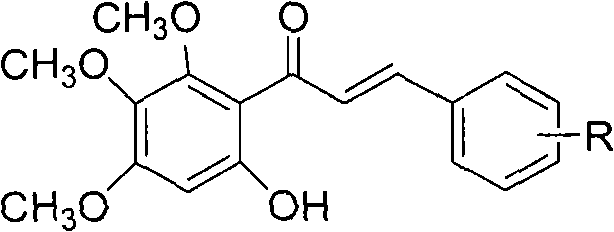
Preceding the cyclization, the formation of the chalcone scaffold via Lewis acid-catalyzed acylation is another critical control point for impurity management. The use of boron trifluoride diethyl etherate ensures a highly regioselective acylation of the 3,4,5-trimethoxyphenol, directing the incoming cinnamoyl group to the desired position to set up the subsequent cyclization correctly. Strict temperature control during the preceding chlorination step, typically maintained between zero and five degrees Celsius, prevents the formation of poly-chlorinated byproducts that could carry through to the final step. By optimizing the stoichiometry of the halogenating agent and the reaction time, the process minimizes the generation of difficult-to-remove impurities, resulting in a crude product that requires minimal purification effort. This level of mechanistic control is essential for R&D teams aiming to scale this process while meeting stringent regulatory requirements for impurity profiles.
How to Synthesize 5,6,7-Trimethoxybaicalein Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable intermediates, starting from the condensation of benzaldehyde derivatives with acetic anhydride. The process is designed to be operationally simple, utilizing standard laboratory glassware and common reagents that are easily sourced from global chemical suppliers. Detailed procedural parameters, including specific reflux times and solvent volumes, are optimized to maximize throughput while maintaining safety standards. For a comprehensive understanding of the exact operational parameters and safety precautions required for each stage of this synthesis, please refer to the standardized guide below.
- Perform Perkin condensation between substituted benzaldehyde and acetic anhydride using a basic catalyst to generate substituted cinnamic acid.
- Convert the cinnamic acid derivative into cinnamoyl chloride using a halogenating agent like thionyl chloride under inert atmosphere.
- Execute Friedel-Crafts acylation with 3,4,5-trimethoxyphenol using BF3-Et2O to form the chalcone intermediate.
- Complete the synthesis via iodine-catalyzed oxidative cyclization in DMSO to yield the final 5,6,7-trimethoxybaicalein derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from extraction-based sourcing to this defined synthetic route represents a strategic opportunity to stabilize costs and secure long-term supply continuity. The reliance on bulk commodity chemicals like benzaldehyde and acetic anhydride insulates the production process from the volatility associated with agricultural commodities, ensuring that raw material costs remain predictable and manageable. Furthermore, the high yields reported in the patent examples indicate a material-efficient process that reduces the overall consumption of starting materials per kilogram of finished product. This efficiency translates directly into a lower cost basis, allowing pharmaceutical companies to allocate resources more effectively towards clinical development and marketing initiatives without compromising on the quality of the active ingredient.
- Cost Reduction in Manufacturing: The elimination of expensive and variable natural extraction processes significantly lowers the operational expenditure associated with producing these intermediates. By utilizing a synthetic route with high atom economy and avoiding the need for complex chromatographic purifications typical of natural products, manufacturers can achieve substantial cost savings. The use of recyclable solvents and the avoidance of precious metal catalysts further contribute to a leaner cost structure, making the final API more economically viable for mass-market pharmaceutical applications.
- Enhanced Supply Chain Reliability: Synthetic manufacturing decouples production from seasonal harvest cycles, enabling year-round production schedules that can respond dynamically to market demand. The raw materials required for this synthesis are produced on a massive global scale for various industries, ensuring that supply disruptions are rare and easily mitigated through multi-vendor sourcing strategies. This reliability is crucial for maintaining uninterrupted drug manufacturing lines and meeting the just-in-time delivery expectations of modern pharmaceutical supply chains.
- Scalability and Environmental Compliance: The reaction conditions described, such as atmospheric pressure reflux and standard crystallization techniques, are inherently scalable from pilot plant to commercial tonnage without requiring specialized high-pressure reactors. Additionally, the process generates less hazardous waste compared to traditional extraction methods which often involve large volumes of organic solvents to process biomass. The simplified waste stream facilitates easier compliance with environmental regulations, reducing the administrative and financial burden associated with waste disposal and environmental permitting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these baicalein derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is vital for making informed decisions regarding process adoption and supplier selection.
Q: What are the primary advantages of this synthetic route over natural extraction?
A: This synthetic method offers superior control over impurity profiles and significantly higher yields compared to extracting baicalein from Scutellaria baicalensis. It allows for precise B-ring modifications that enhance solubility and biological activity, which are difficult to achieve through natural sourcing alone.
Q: Is the iodine-catalyzed cyclization step scalable for industrial production?
A: Yes, the use of molecular iodine in DMSO is a robust and scalable oxidative cyclization method. The reaction conditions described in the patent utilize standard reflux and crystallization techniques that are easily adaptable to large-scale reactors without requiring exotic high-pressure equipment.
Q: How does the B-ring substitution affect the pharmacological properties?
A: Modifying the B-ring skeleton, as demonstrated in this patent, significantly improves the water and lipid solubility of the molecule. This structural enhancement leads to better bioavailability and potentially stronger anticancer and antiviral activities compared to the parent baicalein compound.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,6,7-Trimethoxybaicalein Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for complex pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market launch is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5,6,7-trimethoxybaicalein derivatives meets the highest international standards for safety and efficacy. Our commitment to quality assurance means that you can trust our materials for even the most demanding clinical trial applications.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and budget requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall project costs. Please contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to be your long-term strategic partner in the development of advanced anticancer therapeutics.
