Revolutionizing 2-Thiopheneethamine Production: A Safer Route for Cardiovascular Drug Manufacturing
Revolutionizing 2-Thiopheneethamine Production: A Safer Route for Cardiovascular Drug Manufacturing
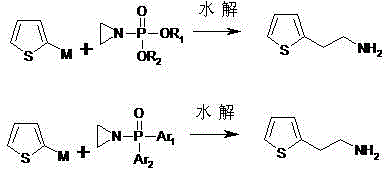
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for critical intermediates, particularly for high-volume cardiovascular medications. Patent CN102336738B introduces a groundbreaking preparation method for 2-thiopheneethamine, a pivotal building block in the synthesis of life-saving drugs like clopidogrel bisulfate. This technology represents a significant paradigm shift away from hazardous traditional chemistries, utilizing a novel addition-hydrolysis sequence that starts with 2-activated thiophene and N-(phosphate)-aziridine. By fundamentally altering the synthetic logic, this approach addresses long-standing concerns regarding toxicity, cost, and operational safety that have plagued the manufacturing of this key fine-chemical intermediate for decades.
For R&D directors and process chemists, the elegance of this invention lies in its simplicity and robustness. The method bypasses the need for harsh reducing agents or toxic cyanide salts, instead relying on the controlled ring-opening of an aziridine derivative. This not only streamlines the purification process but also drastically improves the impurity profile of the final API intermediate. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic advantages is crucial for ensuring a continuous, high-quality supply chain for downstream drug manufacturers who demand stringent purity specifications for their cardiovascular portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-thiopheneethamine has relied on several established but problematic routes, each carrying significant baggage in terms of safety and cost. The nitromethane condensation method, for instance, involves the reduction of 2-nitrothiophene ethylene, a process that necessitates the use of large quantities of reducing agents which are often expensive, volatile, or highly toxic. Furthermore, the isopropyl chloroacetate route suffers from harsh reaction conditions and low yields, making it economically unviable for large-scale operations. Perhaps most concerning is the 2-thiophene acetonitrile method, which requires the handling of sodium cyanide, a substance posing severe environmental and occupational health risks that modern green chemistry initiatives strive to eliminate entirely from the supply chain.
Another common approach involves the use of ethylene oxide, a carcinogenic and highly explosive gas that presents unacceptable safety hazards in an industrial setting. The epoxide route also entails high raw material costs and complex safety protocols for storage and handling. Additionally, existing catalytic shortening methods often employ expensive catalysts that cannot be recycled, leading to inflated production costs and increased heavy metal waste. These cumulative disadvantages have created a pressing need for a manufacturing process that is not only cost-effective but also aligns with modern environmental, health, and safety (EHS) standards without compromising on yield or throughput.
The Novel Approach
The patented methodology offers a transformative solution by employing a direct addition reaction between a 2-activated thiophene species and an N-(phosphate)-aziridine. This strategy elegantly constructs the carbon-nitrogen bond in a single step, followed by a straightforward hydrolysis to reveal the primary amine. The reaction conditions are remarkably mild, operating effectively across a broad temperature range from -50°C to 100°C, which provides process engineers with significant flexibility in reactor management. By avoiding the use of any reducing agents, the process inherently eliminates the generation of associated reduction by-products, simplifying the downstream workup and purification stages significantly.
This novel route is particularly advantageous for cost reduction in pharmaceutical intermediate manufacturing because it utilizes readily available starting materials and avoids the procurement of specialized, high-cost reagents like hydrazine or high-pressure hydrogen. The use of N-(phosphate)-aziridine as an electrophile ensures high regioselectivity during the ring-opening event, minimizing the formation of isomeric impurities that are difficult to separate. Consequently, this method facilitates a cleaner reaction profile, higher overall yields, and a much smaller environmental footprint, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates required for global markets.
Mechanistic Insights into Nucleophilic Aziridine Ring-Opening
At the heart of this synthesis is a classic yet highly effective nucleophilic substitution mechanism driven by ring strain. The process begins with the generation of a 2-activated thiophene species, which acts as a potent nucleophile. As illustrated in the structural data, this activated species can take various forms depending on the metal counterion employed, such as a Grignard reagent (magnesium bromide/chloride), a lithium salt, or even a sodium derivative. The choice of metal allows for fine-tuning the reactivity of the thiophene ring, ensuring that the nucleophilic attack on the aziridine nitrogen occurs efficiently without causing unwanted side reactions on the thiophene ring itself.
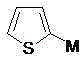
The electrophilic partner, N-(phosphate)-aziridine, is engineered to be highly reactive due to the significant angle strain inherent in the three-membered aziridine ring. The presence of the phosphate group on the nitrogen atom serves a dual purpose: it protects the amine during the addition step and acts as a leaving group precursor for the subsequent hydrolysis. When the 2-activated thiophene attacks the less hindered carbon of the aziridine ring, the ring opens to form a stable N-(phosphate)-2-thiopheneethylamine intermediate. This intermediate is then subjected to hydrolysis under either acidic or alkaline conditions, cleaving the phosphate ester to release the free amine. This two-step sequence ensures high chemoselectivity and prevents the polymerization issues often seen with unprotected aziridines.
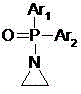
From an impurity control perspective, this mechanism is superior because it avoids the radical pathways associated with catalytic hydrogenation. The absence of metal catalysts like palladium or nickel means there is no risk of heavy metal contamination in the final product, a critical quality attribute for API intermediates. Furthermore, the hydrolysis step can be precisely controlled by adjusting the pH and temperature, allowing manufacturers to optimize the conversion rate while minimizing the degradation of the sensitive thiophene moiety. This level of control is essential for meeting the rigorous purity standards demanded by regulatory bodies for cardiovascular drug substances.
How to Synthesize 2-Thiopheneethamine Efficiently
Implementing this synthesis route requires careful attention to solvent selection and temperature control to maximize the efficiency of the organometallic addition. The patent outlines a versatile protocol where the 2-activated thiophene and the N-(phosphate)-aziridine are reacted in anhydrous aprotic solvents such as toluene, tetrahydrofuran (THF), or ether. The molar ratio of reactants can be adjusted between 0.5:1 and 5:1, providing flexibility to drive the reaction to completion based on the specific reactivity of the metal reagent used. Following the addition, the reaction is quenched with water, and the intermediate is extracted, setting the stage for the final deprotection step.
- Prepare 2-activated thiophene (Grignard, Lithium, or Sodium reagent) and react with N-(phosphate)-aziridine in anhydrous aprotic solvent at -50 to 100°C.
- Quench the addition reaction with water and extract the intermediate N-(phosphate)-2-thiopheneethylamine using organic solvents.
- Hydrolyze the intermediate under acidic or alkaline conditions (0-150°C), adjust pH > 12, and extract the final 2-thiopheneethamine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond simple chemistry. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for hazardous gases like ethylene oxide or toxic salts like sodium cyanide, companies can significantly reduce their regulatory compliance burden and insurance costs. The supply chain becomes more resilient as the reliance on specialized, high-risk chemicals is replaced by stable, commodity-grade organometallic reagents and phosphates that are easier to source and store in bulk quantities without special permits.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the complete removal of expensive reducing agents and precious metal catalysts. Traditional hydrogenation steps require high-pressure equipment and costly catalysts that degrade over time, whereas this addition-hydrolysis route operates at ambient or moderate pressures with inexpensive reagents. The elimination of catalyst recovery and heavy metal scavenging steps further reduces processing time and consumable costs, leading to substantial overall cost savings per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: Operational safety is directly correlated with supply continuity. By removing highly toxic and volatile reagents from the production line, the risk of unplanned shutdowns due to safety incidents or regulatory inspections is minimized. The robust nature of the reaction conditions, which tolerate a wide range of temperatures and solvent mixtures, ensures that production can continue reliably even if minor fluctuations in utility supplies occur, thereby guaranteeing consistent delivery schedules to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The environmental profile of this method is exceptionally clean, generating significantly less hazardous waste compared to nitro-reduction or cyanide-based routes. The aqueous waste streams from the hydrolysis step are easier to treat, and the absence of heavy metals simplifies effluent management. This green chemistry approach facilitates easier permitting for capacity expansion, allowing manufacturers to scale up from pilot plants to multi-ton commercial production facilities with fewer environmental hurdles and lower waste disposal costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and strategic value of this novel route for industry stakeholders evaluating potential technology transfers or sourcing partnerships.
Q: Why is this new synthesis method safer than traditional cyanide routes?
A: Traditional methods often utilize highly toxic sodium cyanide (NaCN) or hazardous ethylene oxide. This patented route replaces those dangerous reagents with stable N-(phosphate)-aziridines and standard organometallic thiophene reagents, significantly reducing operator risk and environmental toxicity.
Q: Does this process require expensive hydrogenation catalysts?
A: No. Unlike nitro-reduction routes that require costly palladium or Raney nickel catalysts and high-pressure hydrogen, this addition-hydrolysis pathway operates under mild thermal conditions without the need for any reducing agents, lowering both CAPEX and OPEX.
Q: What is the scalability potential of this aziridine opening reaction?
A: The reaction utilizes robust organometallic chemistry compatible with standard industrial reactors. The wide temperature tolerance (-50°C to 100°C) and flexible solvent systems (toluene, THF, ether) allow for easy scale-up from kilogram to multi-ton production without complex cryogenic infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Thiopheneethamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of cardiovascular medications. Our technical team has extensively analyzed this patented route and possesses the expertise to implement it effectively within our GMP-compliant facilities. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 2-thiopheneethamine meets the exacting standards required for API synthesis, minimizing the risk of downstream processing failures.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation and safety can drive value and efficiency in your supply chain.
