Advanced Iridium-Catalyzed Synthesis of Chiral Beta-Hydroxy Amino Acid Esters for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust synthetic pathways for complex chiral intermediates, particularly those serving as backbones for critical antibiotic classes. Patent CN113354554B introduces a groundbreaking methodology for the preparation of (2R,3S)-β′-hydroxy-β-amino acid ester derivatives, which are pivotal precursors in the synthesis of carbapenem antibiotics such as imipenem and meropenem. This technology leverages a sophisticated Iridium-catalyzed asymmetric hydrogenation strategy that addresses long-standing challenges in stereoselectivity and reaction efficiency. By utilizing specialized chiral P,N-ligands in conjunction with Iridium complexes, the process achieves conversion rates and optical purity levels that were previously difficult to attain on a commercial scale. The significance of this innovation lies not only in its chemical elegance but also in its potential to streamline the supply chain for high-value pharmaceutical intermediates, ensuring consistent quality and availability for downstream drug manufacturing.
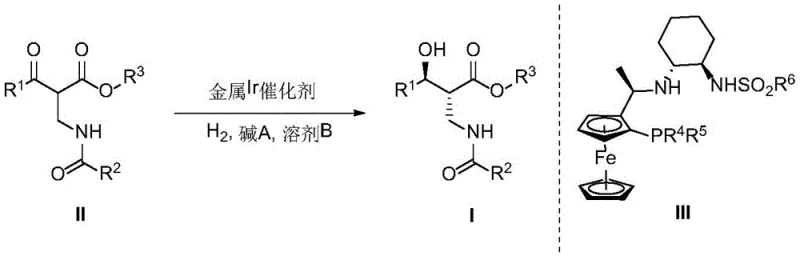
Historically, the synthesis of β′-hydroxy-β-amino acid esters has relied on methods that often compromise either on time or selectivity. Prior art, such as the work by Rimoldi et al., utilized asymmetric catalytic hydrogenation but suffered from excessively long reaction times exceeding 60 hours, which is a significant bottleneck for industrial throughput. Other approaches, including asymmetric hydrogen transfer reactions reported by Zhang et al., achieved moderate enantioselectivity around 95.4% ee but lacked the necessary diastereocontrol, with dr values hovering near 97.8%. Furthermore, biocatalytic methods, while green, often struggled with diastereoselectivity, sometimes yielding values as low as 70% de. These limitations necessitated extensive purification steps, increased solvent consumption, and ultimately drove up the cost of goods. The conventional reliance on these suboptimal routes created vulnerabilities in the supply chain for critical antibiotic intermediates, where batch-to-batch consistency is paramount for regulatory compliance.
The novel approach detailed in the patent fundamentally shifts the paradigm by employing a highly active Iridium catalyst system tailored for this specific transformation. By optimizing the ligand environment around the metal center, the new method drastically reduces reaction times while simultaneously boosting stereoselectivity to near-perfect levels. For instance, specific embodiments demonstrate that under optimized conditions, the reaction can reach completion with yields up to 95%, enantioselectivity up to 99% ee, and diastereoselectivity up to 99:1 dr. This leap in performance is attributed to the precise spatial arrangement provided by the chiral ligands, which effectively differentiate the prochiral faces of the ketone substrate during the hydrogenation event. The result is a process that is not only chemically superior but also economically viable, as it minimizes waste and maximizes the output of the desired (2R,3S) isomer without the need for cumbersome resolution steps.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the intricate interplay between the Iridium metal center and the chiral P,N-ligands, specifically those derived from ferrocene and cyclohexyl backbones as illustrated in the patent documentation. The catalyst formation typically involves mixing an Iridium precursor, such as [Ir(COD)Cl]2, with the chiral ligand in a solvent like ethyl acetate or toluene under an inert atmosphere. This generates an active cationic or neutral Iridium species capable of activating molecular hydrogen. The unique structure of ligands like L4, which features bulky substituents on the phosphine and amine moieties, creates a rigid chiral pocket. This pocket dictates the approach of the substrate, ensuring that hydride transfer occurs exclusively to one face of the carbonyl group. The presence of a base, such as sodium acetate or sodium carbonate, is also critical, likely facilitating the heterolytic cleavage of hydrogen or stabilizing the transition state through coordination.
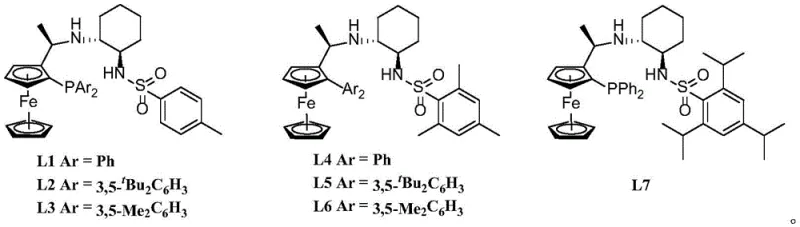
Controlling impurities in this synthesis is largely a function of the catalyst's inherent selectivity and the reaction conditions. The high diastereoselectivity observed (up to 99:1) implies that the formation of the undesired (2S,3S) or (2R,3R) isomers is kinetically suppressed. Mechanistically, this suggests that the transition state leading to the minor diastereomers is significantly higher in energy due to steric clashes between the substrate's protecting groups (like the benzamide moiety) and the ligand's bulky aryl or alkyl groups. Furthermore, the choice of solvent plays a non-trivial role; data indicates that non-polar solvents like toluene often yield superior selectivity compared to polar aprotic solvents like THF, likely due to differences in ion pairing and catalyst solubility. This deep understanding of the mechanistic drivers allows for fine-tuning the process to handle diverse substrates, ensuring that even with variations in the R-groups of the starting material, the impurity profile remains tightly controlled within acceptable limits for pharmaceutical applications.
How to Synthesize (2R,3S)-Beta-Hydroxy-Beta-Amino Acid Esters Efficiently
The practical implementation of this synthesis involves a straightforward two-stage protocol that is amenable to standard chemical manufacturing equipment. The first stage focuses on the in-situ generation of the active catalyst, which requires careful control of the atmosphere to prevent oxidation of the sensitive Iridium complex. The second stage involves the actual hydrogenation in a pressurized reactor, where parameters like temperature and hydrogen pressure are adjusted based on the specific substrate reactivity. This modular approach allows manufacturers to decouple catalyst preparation from the main reaction, providing flexibility in scheduling and quality control. For a comprehensive breakdown of the specific molar ratios, solvent choices, and workup procedures validated in the patent, please refer to the standardized guide below.
- Prepare the Iridium catalyst solution by mixing an Ir complex like [Ir(COD)Cl]2 with a chiral P,N-ligand (e.g., L4) in a solvent such as ethyl acetate or toluene under argon at 0-60°C.
- Combine the beta-carbonyl-beta-amino acid ester substrate, the prepared catalyst solution, a base like sodium acetate, and solvent in an autoclave.
- Conduct the hydrogenation reaction under 1.0-10.0 MPa H2 pressure at 10-100°C for 2-24 hours, then isolate the product via concentration and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this Iridium-catalyzed hydrogenation technology offers substantial strategic benefits over legacy synthesis routes. The primary advantage stems from the drastic improvement in process efficiency, which directly translates to cost reduction in pharmaceutical intermediate manufacturing. By achieving near-quantitative conversion and exceptional selectivity in a single step, the need for downstream purification processes such as recrystallization or chiral chromatography is significantly minimized. This reduction in processing steps lowers the consumption of solvents and consumables, thereby reducing the overall variable cost per kilogram of the produced intermediate. Additionally, the high atom economy of the hydrogenation reaction ensures that the majority of the starting material mass is incorporated into the final product, further enhancing the economic viability of the process for large-scale production runs.
- Cost Reduction in Manufacturing: The elimination of lengthy reaction times and inefficient resolution steps leads to a streamlined production workflow. Unlike previous methods that required over 60 hours to achieve moderate selectivity, this new protocol operates efficiently within a 2 to 24-hour window depending on the substrate. This acceleration in throughput allows for better utilization of reactor capacity, effectively increasing the annual production volume without the need for capital expenditure on new equipment. Furthermore, the high selectivity reduces the loss of valuable chiral materials to unwanted isomers, ensuring that the yield of the commercially relevant (2R,3S) isomer is maximized, which is a critical factor in maintaining healthy profit margins in the competitive API intermediate market.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system contributes to a more reliable supply chain for critical antibiotic precursors. The method tolerates a wide range of substrates with various electronic and steric properties, meaning that a single catalytic platform can be used to produce a diverse portfolio of intermediates. This versatility reduces the complexity of inventory management and mitigates the risk of supply disruptions caused by the failure of specific, narrow-scope synthetic routes. Moreover, the use of commercially available Iridium precursors and ligands ensures that the raw material supply is stable and not subject to the volatility often associated with exotic or proprietary reagents, providing procurement managers with greater confidence in long-term sourcing strategies.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard hydrogenation equipment and common organic solvents like toluene and ethyl acetate. The patent highlights the method's characteristics of little green pollution, which aligns with increasingly stringent environmental regulations governing chemical manufacturing. The high efficiency of the reaction means less waste generation per unit of product, simplifying waste treatment protocols and reducing the environmental footprint of the facility. This compliance advantage is crucial for maintaining operational licenses and meeting the sustainability goals of major pharmaceutical clients who prioritize green chemistry principles in their supply chain audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process feasibility. Understanding these details is essential for technical teams assessing the integration of this method into existing manufacturing lines.
Q: What are the stereoselectivity metrics achieved with this Iridium catalytic system?
A: The patented method achieves exceptional stereocontrol, with enantioselectivity (ee) values reaching up to 99% and diastereoselectivity (dr) ratios as high as 99:1, significantly outperforming previous biocatalytic or transfer hydrogenation methods.
Q: Which substrates are compatible with this asymmetric hydrogenation protocol?
A: The process demonstrates broad substrate scope, successfully converting various beta-carbonyl-beta-amino acid esters containing substituted aryl groups, heterocycles, and different ester moieties into the corresponding hydroxy derivatives with high efficiency.
Q: How does this method improve upon conventional synthesis routes for carbapenem precursors?
A: Unlike older methods that required over 60 hours or suffered from low diastereoselectivity, this Iridium-catalyzed approach offers faster reaction times, higher atomic economy, and reduced environmental impact, making it more suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Hydroxy-Beta-Amino Acid Ester Supplier
The technological potential of this Iridium-catalyzed synthesis represents a significant opportunity for pharmaceutical companies looking to optimize their carbapenem antibiotic supply chains. NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry to deliver high-quality intermediates that meet the rigorous demands of modern drug development. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from pilot studies to full-scale manufacturing. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards, guaranteeing the consistency and reliability required for regulatory filings.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic impact of switching to this high-efficiency protocol. We encourage you to request specific COA data and route feasibility assessments to validate the performance metrics in your own context. Let us collaborate to secure a sustainable and cost-effective supply of these critical chiral building blocks for your next generation of therapeutic agents.
