Advanced Synthesis of Optically Active Epoxy Alcohol Intermediates for Antifungal APIs
Introduction to Patent CN1819998B Technology
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex chiral intermediates, particularly for high-value antifungal agents. Patent CN1819998B introduces a groundbreaking methodology for producing optically active epoxy alcohol derivatives, which serve as critical precursors for triazole-based antifungal drugs. This technology represents a significant paradigm shift from traditional synthesis routes that rely on hazardous and costly reagents. By leveraging a novel sequence involving optically active halohydrin derivatives, the invention enables the production of key intermediates such as 2-aryl-3-methyl-2-[(1H-1,2,4-triazol-1-yl)methyl]epoxides with superior stereocontrol. For R&D directors and procurement specialists, understanding this patent is crucial as it offers a viable alternative to legacy processes that have long been burdened by safety concerns and supply chain bottlenecks associated with heavy metal catalysts.
The core innovation lies in the utilization of readily available chiral pool materials, specifically optically active lactic acid esters, to construct the carbon backbone. This approach not only simplifies the synthetic logic but also ensures a high degree of optical purity from the outset. The process circumvents the need for late-stage resolution or complex asymmetric catalysis that often plagues the manufacture of fine chemicals. Instead, it employs a robust Grignard addition strategy followed by a base-mediated epoxidation. This sequence is particularly attractive for commercial scale-up because it relies on standard unit operations and avoids the extreme sensitivity to moisture and air found in many transition-metal-catalyzed reactions. Consequently, this technology positions itself as a reliable solution for reliable pharmaceutical intermediate supplier networks aiming to diversify their sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-aryl-3-methyl-2-[(1H-1,2,4-triazol-1-yl)methyl]epoxy compounds has been fraught with significant technical and economic challenges. Conventional routes, such as those described in earlier patents like JP-A-2-191262, often necessitate the use of osmium tetroxide (OsO4) for stereoselective oxidation. Osmium tetroxide is not only prohibitively expensive but also extremely toxic, posing severe health risks to operators and requiring specialized waste disposal protocols that drastically inflate operational expenditures. Furthermore, these traditional methods frequently involve lengthy synthetic sequences, sometimes exceeding nine to fifteen steps from commercially available starting materials. Each additional step introduces potential yield losses and increases the accumulation of impurities, necessitating rigorous and costly purification procedures such as column chromatography, which is notoriously difficult to implement on a multi-ton scale.
Another critical drawback of legacy technologies is the reliance on peracids, such as tert-butyl hydroperoxide, for asymmetric oxidation reactions like the Sharpless epoxidation. While effective on a laboratory scale, the handling of large quantities of peracids presents substantial safety hazards regarding explosion and fire risks in an industrial setting. Additionally, these methods often suffer from limited diastereoselectivity, producing mixtures of threo and erythro isomers that require separation. The inability to directly access the desired threo isomer often forces manufacturers to include additional conversion steps, further eroding overall process efficiency. These cumulative factors result in high production costs, extended lead times, and a fragile supply chain that is vulnerable to regulatory changes regarding hazardous chemical usage.
The Novel Approach
In stark contrast, the methodology disclosed in CN1819998B offers a streamlined and industrially safe alternative that addresses these pain points directly. The novel approach initiates with the reaction of an optically active substituted propionic acid ester with a haloacetic acid derivative in the presence of a base. This key transformation generates an optically active haloketone derivative, which serves as a versatile platform for subsequent functionalization. By avoiding the use of osmium catalysts entirely, the process eliminates the associated toxicity and cost burdens, aligning perfectly with modern green chemistry principles. The route is significantly shorter, reducing the number of unit operations and minimizing the time required to reach the final intermediate. This reduction in step count translates directly into improved throughput and lower capital expenditure for manufacturing facilities.
Furthermore, the new method achieves high stereoselectivity through the strategic manipulation of substituents at the alpha-position of the haloketone. This allows for the preferential formation of the desired diastereomer without the need for complex chiral auxiliaries or enzymatic resolutions. The subsequent conversion to the epoxy alcohol is achieved via a simple base treatment, a reaction that is robust, scalable, and easy to control. By replacing hazardous peracids with standard inorganic or organic bases, the process enhances workplace safety and simplifies regulatory compliance. This technological leap provides a compelling value proposition for cost reduction in API manufacturing, enabling producers to deliver high-purity intermediates with greater consistency and reliability.
Mechanistic Insights into Grignard Addition and Base Epoxidation
The chemical elegance of this process is rooted in the precise control of stereochemistry during the Grignard addition step. The synthesis begins with the formation of an enolate from a haloacetic acid derivative, which then reacts with an optically active propionic acid ester to yield a haloketone intermediate. 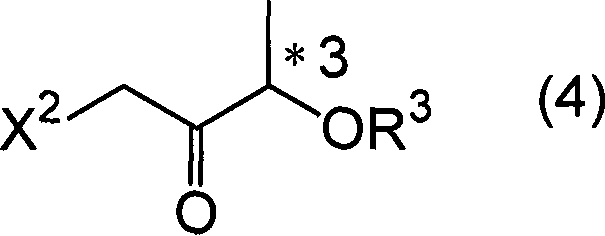 . This haloketone possesses a chiral center derived from the lactic acid precursor, which exerts a strong directing effect on the incoming nucleophile. When an aryl metal compound, such as a Grignard reagent (Ar2M2), is introduced, it attacks the carbonyl carbon. The stereochemical outcome of this addition—whether it forms the threo or erythro configuration—is critically dependent on the nature of the protecting group (R3) at the adjacent position. By selecting bulky silyl groups like tert-butyldimethylsilyl or specific acyl groups like trimethylacetyl, chemists can sterically hinder one face of the carbonyl, thereby guiding the Grignard reagent to attack from the opposite side with high fidelity.
. This haloketone possesses a chiral center derived from the lactic acid precursor, which exerts a strong directing effect on the incoming nucleophile. When an aryl metal compound, such as a Grignard reagent (Ar2M2), is introduced, it attacks the carbonyl carbon. The stereochemical outcome of this addition—whether it forms the threo or erythro configuration—is critically dependent on the nature of the protecting group (R3) at the adjacent position. By selecting bulky silyl groups like tert-butyldimethylsilyl or specific acyl groups like trimethylacetyl, chemists can sterically hinder one face of the carbonyl, thereby guiding the Grignard reagent to attack from the opposite side with high fidelity.
Following the formation of the optically active halohydrin derivative, the pathway proceeds to the construction of the epoxide ring. This transformation is achieved through an intramolecular nucleophilic substitution reaction facilitated by a base. 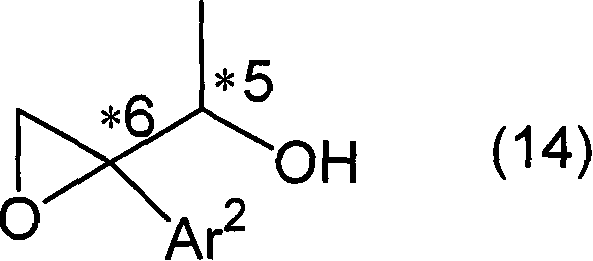 . If the hydroxyl protecting group (R3) is removable under acidic or fluoridic conditions, it is first deprotected to reveal a free diol. Subsequent treatment with a base, such as sodium hydroxide or sodium methoxide, deprotonates the secondary hydroxyl group. The resulting alkoxide ion then performs a backside attack on the carbon bearing the halogen atom, displacing the halide and closing the three-membered epoxide ring. This mechanism ensures that the stereochemical information established in the previous steps is retained and transferred efficiently to the final epoxy alcohol structure. The ability to tune the R3 group allows for flexibility in managing the timing of the epoxidation, offering process chemists valuable options for optimizing yield and purity.
. If the hydroxyl protecting group (R3) is removable under acidic or fluoridic conditions, it is first deprotected to reveal a free diol. Subsequent treatment with a base, such as sodium hydroxide or sodium methoxide, deprotonates the secondary hydroxyl group. The resulting alkoxide ion then performs a backside attack on the carbon bearing the halogen atom, displacing the halide and closing the three-membered epoxide ring. This mechanism ensures that the stereochemical information established in the previous steps is retained and transferred efficiently to the final epoxy alcohol structure. The ability to tune the R3 group allows for flexibility in managing the timing of the epoxidation, offering process chemists valuable options for optimizing yield and purity.
How to Synthesize Optically Active Halohydrin Derivatives Efficiently
Implementing this synthesis route requires careful attention to reaction conditions, particularly temperature control and reagent stoichiometry, to maximize the diastereomeric ratio. The initial formation of the haloketone typically involves reacting a lactic acid ester with a haloacetate in the presence of a strong base like tert-butylmagnesium chloride at low temperatures, often ranging from -20°C to 0°C, to prevent side reactions. Once the haloketone is isolated, the Grignard addition is performed under inert atmosphere conditions to maintain the integrity of the organometallic reagent. The detailed standardized synthesis steps see the guide below.
- React optically active substituted propionic acid ester with a haloacetic acid derivative in the presence of a base to form an optically active haloketone derivative.
- Perform a highly stereoselective addition of an aryl metal compound (Grignard reagent) to the haloketone to obtain the optically active halohydrin derivative.
- Treat the resulting halohydrin with a base to induce intramolecular epoxidation, yielding the final optically active epoxy alcohol derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1819998B offers transformative benefits that extend beyond mere chemical novelty. The most immediate impact is seen in the realm of cost structure optimization. By eliminating the requirement for osmium tetroxide, a precious metal catalyst with volatile pricing and strict handling regulations, manufacturers can achieve substantial cost savings in raw material procurement. Additionally, the removal of column chromatography from the purification workflow significantly reduces the consumption of silica gel and organic solvents, which are major cost drivers in fine chemical production. This simplification of the downstream processing allows for higher batch volumes and faster turnaround times, directly contributing to cost reduction in pharmaceutical intermediate manufacturing without compromising on quality standards.
Supply chain reliability is another critical area where this technology excels. The reliance on commodity chemicals such as lactic acid esters, chloroacetic acid, and common Grignard reagents ensures a stable and diversified supply base. Unlike specialized chiral catalysts that may be sourced from a single vendor, these starting materials are available from multiple global suppliers, mitigating the risk of supply disruptions. Furthermore, the enhanced safety profile of the process, characterized by the absence of explosive peracids and highly toxic heavy metals, facilitates smoother regulatory approvals and reduces insurance premiums. This robustness makes the process highly scalable, supporting the commercial scale-up of complex pharmaceutical intermediates from pilot plant quantities to multi-ton annual production capacities with minimal technical risk.
- Cost Reduction in Manufacturing: The elimination of expensive osmium catalysts and the reduction of synthetic steps lead to a leaner cost structure. By avoiding complex purification techniques like column chromatography and utilizing standard extraction and crystallization methods, the process minimizes solvent usage and waste generation. This operational efficiency translates into lower unit costs, allowing buyers to negotiate more competitive pricing for high-purity intermediates while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: The use of widely available starting materials such as lactic acid derivatives and common halogens ensures a resilient supply chain. This reduces dependency on niche reagents that are prone to shortages. The simplified process flow also means shorter manufacturing cycles, which enhances the ability to respond quickly to market demand fluctuations. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API production schedules are met consistently.
- Scalability and Environmental Compliance: The process is inherently designed for large-scale operation, utilizing robust reaction conditions that are easily managed in standard stainless steel reactors. The avoidance of hazardous waste streams associated with heavy metals and peracids simplifies environmental compliance and waste treatment. This sustainability aspect not only lowers disposal costs but also aligns with the increasing corporate social responsibility goals of major pharmaceutical companies, making the supplier a more attractive partner for long-term contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of the technology for potential partners and licensees.
Q: What are the primary advantages of this synthesis route over conventional methods?
A: This method eliminates the need for expensive and highly toxic osmium oxide catalysts and peracid oxidants used in traditional Sharpless or dihydroxylation routes. It utilizes readily available raw materials like lactic acid esters and reduces the total number of synthetic steps significantly, improving overall process safety and cost-efficiency.
Q: How is stereoselectivity controlled in the production of halohydrin derivatives?
A: Stereoselectivity is precisely controlled by modifying the substituent at the alpha-position of the haloketone derivative. By selecting specific protecting groups such as silyl, acyl, or heterocyclic groups, manufacturers can dictate the formation of specific diastereomers (threo vs. erythro) required for the target antifungal agent.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for industrial scalability. It avoids hazardous reagents like tert-butyl hydroperoxide in large quantities and eliminates complex purification steps like column chromatography, relying instead on standard extraction and crystallization techniques that are robust for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Halohydrin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting advanced synthetic methodologies to stay competitive in the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the technology outlined in CN1819998B and possesses the technical capability to implement this route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of optically active halohydrin derivatives meets the highest international standards for chirality and chemical purity.
We invite you to collaborate with us to leverage this innovative technology for your antifungal drug development programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and secure a reliable source of high-quality intermediates that drive your success in the competitive landscape of generic and proprietary pharmaceuticals.
