Scalable Synthesis of 3-Quaternary Carbon Indoline-2-Ones via Copper-Catalyzed 1,4-Ester Migration
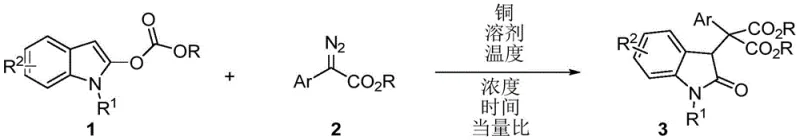
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds, particularly those containing quaternary carbon centers which are prevalent in bioactive molecules. Patent CN115636778A introduces a groundbreaking synthetic strategy for producing 3-quaternary carbon substituted indoline-2-one compounds, a core skeleton found in numerous drugs exhibiting antitumor, antimicrobial, and antiviral activities. This innovation leverages a copper-catalyzed 1,4-ester group migration reaction between indole-2-carbonate derivatives and diazoacetates. Unlike traditional methods that rely on expensive noble metals or harsh oxidative conditions, this protocol utilizes inexpensive copper salts to achieve high efficiency and excellent atom economy. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-functionalized indolin-2-ones has been fraught with significant challenges that hinder efficient commercial production. Traditional approaches often involve the oxidation of indoles, a method severely limited by substrate applicability and primarily restricted to the synthesis of spirocyclic variants at the 3-position. Another common pathway involves Lewis acid-catalyzed intramolecular cyclization of aryl amidoalkenyl esters; however, this route suffers from complex raw material synthesis and is generally incapable of constructing the coveted 3-quaternary carbon center, yielding only 3-tertiary carbon products. Furthermore, direct nucleophilic substitution is problematic due to the dual nucleophilicity of the indolin-2-one at the 3-position carbon and 2-position oxygen, leading to numerous by-products and elimination reactions when electrophiles with quaternary centers are employed. These limitations result in low yields, difficult purification processes, and inflated production costs, making them unsuitable for reliable large-scale supply chains.
The Novel Approach
The methodology disclosed in CN115636778A overcomes these historical bottlenecks by employing a novel 1,4-acyl rearrangement strategy catalyzed by affordable copper salts. By reacting readily available indole-2-carbonate derivatives with diazoacetates, the process efficiently constructs the challenging 3-quaternary carbon center through a migration mechanism. This approach eliminates the need for expensive rhodium catalysts, which were previously reported for similar rearrangements but often led to undesired C-H insertion by-products. The new copper-catalyzed route operates under mild conditions, typically between 25°C and 70°C, and utilizes common organic solvents like dichloromethane. This not only simplifies the operational requirements but also drastically reduces the environmental footprint and raw material costs, positioning it as a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Copper-Catalyzed 1,4-Ester Migration
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the copper catalyst. Upon interaction with the diazoacetate, the copper salt generates a reactive copper carbene intermediate. This highly electrophilic species undergoes a migratory insertion into the indole-2-carbonate framework. Crucially, the reaction proceeds through a 1,4-ester group migration rather than the typical C-H insertion observed with other metal catalysts like rhodium. This specific migration pathway is essential for forming the sterically congested quaternary carbon center at the 3-position of the indoline ring. The precise control over this migration ensures high regioselectivity and minimizes the formation of structural isomers, which is a critical quality attribute for R&D teams focused on impurity profiling and regulatory compliance.
Furthermore, the mechanism inherently suppresses side reactions that plague conventional nucleophilic substitutions. The dual nucleophilicity of the indoline scaffold, which usually leads to O-alkylation or elimination by-products, is effectively managed by the concerted nature of the carbene insertion and subsequent rearrangement. The use of copper thiophene-2-carboxylate (CuTc) as the optimal catalyst, typically at a loading of 5 mol%, provides the right balance of reactivity and selectivity. This mechanistic understanding allows process chemists to fine-tune reaction parameters, such as temperature and stoichiometry, to maximize yield and purity. The ability to consistently produce high-purity 3-quaternary carbon indoline-2-ones with minimal impurity burden significantly downstream processing costs and accelerates the timeline for clinical candidate selection.
How to Synthesize 3-Quaternary Carbon Indoline-2-One Efficiently
Implementing this synthesis in a laboratory or pilot plant setting is straightforward and relies on standard organic synthesis techniques. The process begins with the preparation of the indole-2-carbonate derivative, which can be easily synthesized from commercially available indolin-2-ones. The key transformation involves mixing the copper catalyst with the carbonate derivative under an inert argon atmosphere to prevent catalyst deactivation. Subsequently, the diazoacetate component is introduced, and the mixture is stirred at moderate temperatures. The reaction progress can be monitored via TLC, and upon completion, the workup involves simple solvent evaporation followed by silica gel column chromatography. This operational simplicity makes it an ideal candidate for technology transfer and scale-up.
- Prepare the reaction mixture by adding the copper catalyst (e.g., CuTc, 5 mol%) and indole-2-carbonate derivative to a reaction vessel under argon protection.
- Add the appropriate organic solvent (e.g., dichloromethane) and introduce the diazoacetate compound (1-2 equivalents) while maintaining an inert atmosphere.
- Stir the reaction at 25-70°C for 0.2-12 hours, then remove the solvent under reduced pressure and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology offers tangible strategic advantages beyond mere technical feasibility. The most significant benefit is the drastic reduction in catalyst costs. By replacing precious rhodium complexes with abundant copper salts, the direct material cost of the catalytic system is lowered substantially. Additionally, the reaction conditions are mild, avoiding the need for extreme cryogenic temperatures or high-pressure equipment, which translates to lower energy consumption and reduced capital expenditure on specialized reactor infrastructure. The use of common solvents like dichloromethane further simplifies solvent recovery and waste management protocols, aligning with modern green chemistry initiatives and reducing disposal costs.
- Cost Reduction in Manufacturing: The substitution of expensive rhodium catalysts with inexpensive copper salts like CuTc results in a profound decrease in raw material expenses. Since the catalyst loading is low (1-10 mol%) and the metal is non-precious, the overall cost of goods sold (COGS) for the final API intermediate is significantly optimized. Moreover, the high atom economy of the 1,4-migration reaction ensures that a greater proportion of the starting mass is incorporated into the final product, minimizing waste generation and maximizing material efficiency. This economic efficiency is critical for maintaining competitive pricing in the global pharmaceutical market.
- Enhanced Supply Chain Reliability: The starting materials, indole-2-carbonate derivatives and diazoacetates, are either commercially available or easily synthesized from bulk chemicals, ensuring a stable and resilient supply chain. Unlike specialized reagents that may have long lead times or single-source dependencies, the precursors for this reaction are widely accessible. The robustness of the reaction across a wide range of substrates, including those with electron-withdrawing and electron-donating groups, means that supply chain disruptions for specific analogs can be mitigated by the flexibility of the synthetic route. This reliability is paramount for ensuring continuous production schedules and meeting delivery commitments to downstream partners.
- Scalability and Environmental Compliance: The patent data explicitly demonstrates the scalability of this process through successful gram-scale reactions yielding over 1 gram of product with consistent efficiency. The mild reaction temperatures (25-70°C) and ambient pressure conditions simplify the engineering requirements for scaling up to multi-kilogram or tonne levels. Furthermore, the avoidance of heavy metal contaminants associated with other catalytic systems simplifies the purification process, reducing the burden on wastewater treatment facilities. The simplified workup procedure, involving standard column chromatography or crystallization, facilitates faster batch turnover times, thereby enhancing overall production throughput and environmental compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for potential integration into their manufacturing portfolios.
Q: Why is copper catalysis preferred over rhodium for this transformation?
A: Copper salts like CuTc are significantly more cost-effective and commercially abundant compared to expensive rhodium catalysts. Furthermore, the patent data indicates that rhodium catalysts tend to favor C-H insertion by-products, whereas the copper system efficiently drives the desired 1,4-ester migration to form the quaternary center.
Q: What is the substrate scope for the diazo component in this reaction?
A: The method demonstrates broad adaptability, successfully accommodating diazoacetates with various aryl substituents including electron-donating groups (methyl, methoxy, tert-butyl) and electron-withdrawing groups (chloro, bromo, trifluoromethyl), as well as naphthyl and hydrogen substituents.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly describes a gram-scale reaction (Example 1-1) yielding 1.03g of product with a 73% yield, demonstrating that the mild conditions and simple workup procedures are amenable to commercial scale-up without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quaternary Carbon Indoline-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed 1,4-ester migration technology for producing high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art rigorous QC labs and adherence to stringent purity specifications guarantee that every batch of 3-quaternary carbon indoline-2-one meets the highest industry standards, supporting your drug development timelines with unwavering quality and consistency.
We invite you to collaborate with us to leverage this cost-effective and scalable synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this methodology can optimize your budget. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a reliable supply of these critical building blocks for your next-generation therapeutics.
