Advanced Tetradentate Palladium Complexes for High-Efficiency Deep Blue OLED Manufacturing
The rapid evolution of the organic light-emitting diode (OLED) industry has created an urgent demand for high-performance blue phosphorescent materials that can match the efficiency and stability of their red and green counterparts. Patent CN108424425B addresses this critical technological gap by disclosing a novel class of tetradentate ring metal palladium complexes based on 4-aryl-3,5-disubstituted pyrazole ligands. These materials represent a significant breakthrough in the field of optoelectronics, offering a pathway to achieve deep blue emission with high internal quantum efficiency. Unlike traditional fluorescent emitters that are limited by spin statistics, these phosphorescent complexes can theoretically utilize both singlet and triplet excitons, potentially reaching 100% internal quantum efficiency. The structural innovation lies in the specific coordination environment around the palladium center, which stabilizes the excited states necessary for efficient light emission.
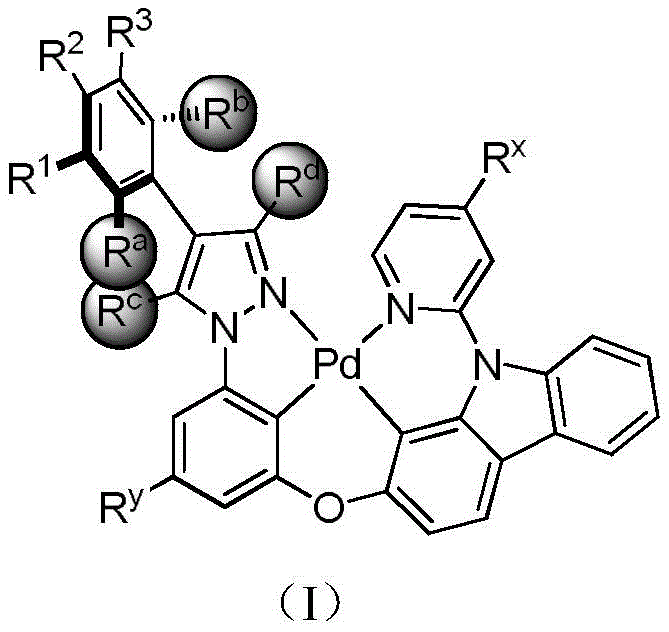
For R&D directors and material scientists, the significance of this patent extends beyond mere emission color. The disclosed compounds are engineered to overcome the notorious instability associated with blue OLED devices. By incorporating bulky substituents at the 3 and 5 positions of the pyrazole ring and specific aryl groups at the 4 position, the molecular architecture achieves a balance between electronic properties and physical robustness. This design strategy effectively modulates the HOMO and LUMO energy levels, allowing for precise tuning of the emission spectrum into the deep blue region (430-450 nm). Furthermore, the tetradentate nature of the ligand system ensures a rigid coordination geometry, which minimizes non-radiative decay pathways caused by molecular vibration, thereby enhancing the overall luminous intensity and operational lifetime of the device.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of blue phosphorescent materials has been hindered by several intrinsic challenges that have slowed the commercialization of full-color OLED displays. Conventional blue emitters often suffer from poor thermal stability, which becomes a critical bottleneck during the vacuum thermal evaporation process used in device manufacturing. Many existing materials decompose at temperatures close to or below the required evaporation temperature of approximately 300°C, leading to impurity formation and inconsistent film quality. Additionally, achieving deep blue emission usually requires high triplet energy levels, which places stringent demands on the host materials and often results in inefficient energy transfer. Traditional bidentate or tridentate complexes may lack the structural rigidity needed to suppress vibrational relaxation, resulting in broader emission spectra and lower color purity. These limitations collectively contribute to shorter device lifespans and higher production costs due to yield losses and the need for frequent equipment maintenance.
The Novel Approach
The approach detailed in patent CN108424425B offers a robust solution to these longstanding issues through the strategic design of tetradentate palladium complexes. By utilizing a 4-aryl-3,5-disubstituted pyrazole core, the inventors have created a ligand system that provides superior thermal stability, with decomposition temperatures consistently exceeding 340°C. This thermal margin ensures that the materials remain intact during the high-temperature deposition processes, guaranteeing high purity in the final emissive layer. The introduction of sterically hindered substituents effectively isolates the pyrazole ring from the aryl group, reducing unwanted conjugation that could redshift the emission. This structural modification allows the material to maintain a high minimum triplet energy level, which is essential for deep blue phosphorescence. Moreover, the rigid tetradentate coordination significantly restricts molecular motion, reducing energy loss through non-radiative transitions and resulting in narrower emission spectra with improved color coordinates (CIE y < 0.1).
Mechanistic Insights into Tetradentate Palladium Cyclometalation
The synthesis and function of these complexes rely on sophisticated organometallic mechanisms that ensure precise control over the final molecular structure. The formation of the tetradentate ligand involves a sequence of coupling reactions that build the complex aromatic framework necessary for effective light emission. Initially, a copper-catalyzed coupling reaction links the pyrazole core with substituted phenyl rings, establishing the foundational skeleton. This is followed by a palladium-catalyzed Suzuki cross-coupling to introduce further aryl diversity at the 4-position of the pyrazole. The final and most critical step is the cyclometalation process, where the palladium center coordinates with the nitrogen atoms of the pyrazole and the carbazole-pyridine auxiliary ligand. This cyclometalation creates a stable, planar coordination geometry that locks the molecule into a conformation favorable for phosphorescence. The strong spin-orbit coupling induced by the heavy palladium atom facilitates efficient intersystem crossing from singlet to triplet states, enabling the harvesting of triplet excitons for light emission.
From an impurity control perspective, the synthetic route is designed to minimize side reactions that could generate non-emissive byproducts. The use of specific catalysts such as CuI with L-proline and Pd2(dba)3 with S-Phos ensures high selectivity during the bond-forming steps. The purification process typically involves silica gel column chromatography, which effectively removes residual catalysts and unreacted starting materials. This high level of chemical purity is paramount for OLED applications, as trace impurities can act as quenching sites that drastically reduce device efficiency. The structural rigidity imparted by the tetradentate ligand also contributes to chemical stability, preventing degradation under electrical stress. By carefully controlling the substituents on the pyridine and carbazole moieties, manufacturers can fine-tune the electron transport properties of the emitter, ensuring balanced charge injection within the OLED stack. This mechanistic understanding allows for the rational design of derivatives with even greater performance metrics.
How to Synthesize Tetradentate Palladium Complex Efficiently
The preparation of these high-value OLED materials follows a well-defined multi-step synthetic protocol that balances yield with purity. The process begins with the construction of the functionalized pyrazole intermediate, followed by the attachment of the auxiliary carbazole-pyridine ligand, and concludes with the metallation step. Each stage requires careful control of reaction conditions, including temperature, atmosphere, and stoichiometry, to ensure optimal conversion. The use of inert atmospheres and anhydrous solvents is critical to prevent catalyst deactivation and side reactions. Detailed standard operating procedures for each transformation are essential for scaling this chemistry from the laboratory to commercial production volumes.
- Perform copper-catalyzed coupling of 3,5-dimethyl-4-bromopyrazole with iodoanisole using CuI and L-proline in DMSO at 120°C.
- Execute Suzuki cross-coupling with arylboronic acids using Pd2(dba)3 and S-Phos catalyst in toluene at 110°C to form the pyrazole intermediate.
- Complete the synthesis via cyclometalation with Pd(OAc)2 in acetic acid at 110°C to form the final tetradentate palladium complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology offers tangible benefits that extend beyond technical performance metrics. The enhanced thermal stability of these palladium complexes directly translates to improved process reliability in manufacturing facilities. Because the materials can withstand higher temperatures without decomposition, there is less risk of clogging evaporation sources or contaminating vacuum chambers, which reduces downtime and maintenance costs. This reliability ensures a more consistent supply of high-quality emissive layers, minimizing waste and maximizing yield in the production of OLED panels. Furthermore, the modular nature of the synthetic route allows for the use of readily available starting materials, which mitigates supply chain risks associated with exotic or scarce reagents. The ability to tune the emission properties through simple structural modifications means that a single platform technology can serve multiple product lines, simplifying inventory management and reducing the complexity of the supply chain.
- Cost Reduction in Manufacturing: The robust thermal profile of these complexes eliminates the need for specialized low-temperature deposition equipment, allowing manufacturers to utilize existing infrastructure more efficiently. By reducing the frequency of source cleaning and replacement due to material decomposition, operational expenditures are significantly lowered. Additionally, the high quantum efficiency of the emitters means that less material is required to achieve the desired brightness, leading to direct savings on raw material costs per unit. The streamlined synthesis pathway, which avoids overly complex protection-deprotection sequences, further contributes to a lower cost of goods sold, making high-performance blue OLEDs more economically viable for mass-market applications.
- Enhanced Supply Chain Reliability: The synthetic methodology relies on established coupling reactions that are well-understood and scalable, reducing the risk of production bottlenecks. The use of common catalysts and solvents ensures that sourcing remains stable even during market fluctuations. The high purity achievable through standard purification techniques reduces the dependency on specialized refining services, shortening lead times for finished goods. This reliability is crucial for meeting the tight delivery schedules of consumer electronics manufacturers, ensuring that display production lines remain operational without interruption. The versatility of the chemical platform also allows for rapid adaptation to changing market demands, providing a strategic advantage in a fast-paced industry.
- Scalability and Environmental Compliance: The synthesis is amenable to scale-up from gram to kilogram quantities without significant changes in reaction parameters, facilitating a smooth transition from R&D to commercial production. The processes described utilize solvents and reagents that can be managed within standard environmental health and safety frameworks, simplifying regulatory compliance. The high efficiency of the reactions minimizes waste generation, aligning with modern sustainability goals and reducing the environmental footprint of the manufacturing process. This scalability ensures that the supply can grow in tandem with the increasing demand for blue OLED displays in smartphones, televisions, and lighting applications.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial viability of these advanced palladium complexes. Understanding these details is essential for stakeholders evaluating the integration of this technology into their product roadmaps. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for decision-making purposes.
Q: What is the thermal stability of these palladium complexes?
A: The complexes exhibit exceptional thermal stability with decomposition temperatures exceeding 340°C, which is significantly higher than the typical thermal evaporation temperature of 300°C used in OLED device fabrication.
Q: What is the emission color range of these materials?
A: These complexes are designed for deep blue emission, with maximum emission wavelengths typically falling between 430 nm and 450 nm, making them ideal for high-quality display applications.
Q: How does the ligand structure affect performance?
A: The 4-aryl-3,5-disubstituted pyrazole ligand introduces steric hindrance that reduces conjugation and enhances molecular rigidity, leading to higher triplet energy levels and improved quantum efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetradentate Palladium Complex Supplier
As the global demand for high-resolution and energy-efficient displays continues to surge, securing a reliable source of advanced emissive materials is paramount for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in organometallic chemistry to deliver premium OLED materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of major display manufacturers. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of tetradentate palladium complex meets the exacting standards required for high-performance OLED devices. Our commitment to quality assurance ensures that your production lines run smoothly with minimal disruption.
We invite you to collaborate with us to explore how these innovative materials can enhance your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific manufacturing needs. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate the practical benefits of integrating these deep blue emitters into your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to cutting-edge technology backed by a proven track record of excellence in fine chemical manufacturing.
