Advanced Synthesis of 4-(2,2,2-Trichloroethyl)-Beta-Lactam Derivatives for Commercial Scale-Up
Introduction to Patent CN112552285A Technology
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing privileged scaffolds, particularly the beta-lactam core which serves as the backbone for numerous antibiotics and bioactive natural products. Patent CN112552285A discloses a groundbreaking synthesis method for 4-(2,2,2-trichloroethyl)-beta-lactam derivatives, addressing critical bottlenecks in existing manufacturing protocols. This innovation leverages a copper-catalyzed radical cyclization strategy that transforms substituted N-quinoline-3-butenamide derivatives into valuable heterocyclic structures with exceptional efficiency. By utilizing chloroform not merely as a solvent but as a key reactant source for the trichloromethyl group, the process achieves high atom economy and operational simplicity. For R&D directors and procurement specialists, this technology represents a significant leap forward in accessing complex nitrogen-containing heterocycles, offering a pathway that balances high purity with economic viability in a competitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of beta-lactam rings has relied on classical methods such as the Staudinger synthesis or various oxidative cyclization protocols that often suffer from significant drawbacks in a commercial setting. Traditional approaches frequently necessitate the use of expensive, moisture-sensitive reagents or harsh reaction conditions that can compromise the integrity of sensitive functional groups on the substrate. Furthermore, many prior art methods involve multi-step sequences with low overall yields, generating substantial chemical waste and requiring rigorous purification to remove toxic metal residues or byproducts. These inefficiencies translate directly into higher production costs and extended lead times, creating friction in the supply chain for critical pharmaceutical intermediates. The reliance on specialized starting materials that are not readily available on a bulk scale further exacerbates the challenge, limiting the ability of manufacturers to respond quickly to market demand fluctuations.
The Novel Approach
In stark contrast, the methodology described in patent CN112552285A introduces a streamlined, single-pot radical addition cyclization that fundamentally simplifies the synthetic landscape. This novel approach employs a copper salt catalyst in conjunction with an organic peroxide oxidant to generate trichloromethyl radicals in situ from chloroform, which then add across the alkene moiety of the substrate to trigger ring closure. 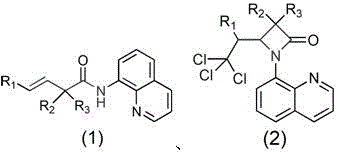 The reaction proceeds under relatively mild thermal conditions, typically between 100°C and 120°C, eliminating the need for cryogenic temperatures or extreme pressures. This shift allows for the direct functionalization of simple, commercially available alkenyl amides, drastically reducing the number of synthetic steps and the associated material costs. The versatility of this system is evidenced by its tolerance for a wide range of substituents, including alkyl, aryl, and ester groups, making it a universal platform for diverse derivative synthesis.
The reaction proceeds under relatively mild thermal conditions, typically between 100°C and 120°C, eliminating the need for cryogenic temperatures or extreme pressures. This shift allows for the direct functionalization of simple, commercially available alkenyl amides, drastically reducing the number of synthetic steps and the associated material costs. The versatility of this system is evidenced by its tolerance for a wide range of substituents, including alkyl, aryl, and ester groups, making it a universal platform for diverse derivative synthesis.
Mechanistic Insights into Copper-Catalyzed Radical Cyclization
The core of this technological advancement lies in the precise orchestration of a free-radical chain mechanism mediated by the copper catalyst and the peroxide oxidant. The reaction initiates with the homolytic cleavage of the oxidant, di-tert-butyl peroxide (DTBP), generating tert-butoxy radicals which abstract a hydrogen atom or interact with the copper center to facilitate the formation of the trichloromethyl radical from the chloroform solvent. This highly reactive radical species then undergoes a regioselective addition to the terminal carbon of the alkene double bond in the N-quinoline-3-butenamide substrate. The resulting carbon-centered radical intermediate is stabilized by the adjacent quinoline directing group, which plays a crucial role in guiding the subsequent intramolecular cyclization onto the amide carbonyl or nitrogen center to form the four-membered beta-lactam ring. Understanding this mechanistic nuance is vital for process chemists, as it explains the high regioselectivity and the minimal formation of oligomeric byproducts that often plague radical reactions.
From an impurity control perspective, the choice of catalyst and reaction parameters is paramount to ensuring the high purity required for pharmaceutical applications. The patent data highlights that copper tetraacetonitrile hexafluorophosphate (Cu(CH3CN)4PF6) outperforms other copper salts, likely due to its specific redox potential and solubility profile which optimizes the radical generation rate.  By maintaining the reaction temperature at an optimal 110°C, the process minimizes thermal decomposition of the sensitive beta-lactam product while ensuring complete conversion of the starting material. The use of chloroform as both solvent and reactant simplifies the post-reaction workup, as the removal of excess solvent also removes unreacted trichloromethane, thereby reducing the burden on downstream purification units. This mechanistic clarity allows for tight process control, ensuring that the final API intermediate meets stringent quality specifications with a consistent impurity profile.
By maintaining the reaction temperature at an optimal 110°C, the process minimizes thermal decomposition of the sensitive beta-lactam product while ensuring complete conversion of the starting material. The use of chloroform as both solvent and reactant simplifies the post-reaction workup, as the removal of excess solvent also removes unreacted trichloromethane, thereby reducing the burden on downstream purification units. This mechanistic clarity allows for tight process control, ensuring that the final API intermediate meets stringent quality specifications with a consistent impurity profile.
How to Synthesize 4-(2,2,2-Trichloroethyl)-Beta-Lactam Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The process begins with the dissolution of the substituted N-quinoline-3-butenamide derivative and the copper catalyst in chloroform, followed by the careful addition of the oxidant. The detailed standardized synthesis steps below outline the precise stoichiometry and conditions validated in the patent examples, serving as a reliable guide for technical teams aiming to replicate these results. Following these protocols ensures that the radical cyclization proceeds smoothly, avoiding common pitfalls such as incomplete conversion or catalyst deactivation.
- Dissolve the substituted N-quinoline-3-butenamide derivative and the copper salt catalyst (e.g., Cu(CH3CN)4PF6) in chloroform.
- Add the oxidizing agent di-tert-butyl peroxide (DTBP) to the mixture and heat the solution to 110°C until the reaction is complete.
- Purify the crude product using silica gel column chromatography with a petroleum ether and ethyl acetate solvent system to isolate the final beta-lactam derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible strategic benefits that extend beyond mere chemical yield. The transition to a process that utilizes commodity chemicals like chloroform and widely available copper salts significantly de-risks the supply chain by reducing dependence on exotic or single-source reagents. This shift enhances supply continuity and mitigates the volatility associated with sourcing specialized precursors, allowing for more predictable production planning and inventory management. Furthermore, the simplified operational workflow reduces the demand for specialized equipment and extensive operator training, contributing to overall operational excellence.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive transition metal catalysts and the reduction of synthetic steps, which collectively lower the cost of goods sold. By using chloroform as a dual-purpose solvent and reactant, the process minimizes raw material consumption and waste disposal costs, leading to substantial savings in variable production expenses. The high isolated yields reported, such as 92% for the model substrate, mean that less starting material is required to produce the same amount of product, directly improving the material efficiency of the manufacturing line.
- Enhanced Supply Chain Reliability: The reliance on easily obtainable starting materials ensures that production schedules are not disrupted by raw material shortages, a common issue in the fine chemical sector. The robustness of the reaction conditions allows for flexible manufacturing across different facilities without the need for highly specialized infrastructure, thereby diversifying the supply base and increasing resilience against regional disruptions. This reliability is critical for maintaining uninterrupted supply to downstream pharmaceutical customers who depend on consistent availability of key intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and simple workup procedures make this process inherently scalable from gram to ton quantities without significant re-engineering. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, reducing the compliance burden and associated costs for waste treatment. This green chemistry approach not only improves the corporate sustainability profile but also future-proofs the manufacturing process against tightening regulatory standards regarding solvent use and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-lactam synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production portfolios.
Q: What are the primary advantages of this copper-catalyzed method over traditional beta-lactam synthesis?
A: This method utilizes readily available chloroform as both solvent and reactant, operates under mild thermal conditions (110°C), and avoids the use of expensive or toxic reagents often required in conventional ring-closure strategies, resulting in a more environmentally friendly and cost-effective process.
Q: Which catalyst system provides the optimal yield for this transformation?
A: Experimental data indicates that copper tetraacetonitrile hexafluorophosphate (Cu(CH3CN)4PF6) combined with di-tert-butyl peroxide (DTBP) as the oxidant delivers superior yields, such as 92% for the model substrate, compared to other copper salts like CuBr2 or Cu(OAc)2.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process is highly suitable for scale-up due to its simple operation, the use of commodity chemicals like chloroform, and the absence of complex purification steps, which facilitates efficient commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(2,2,2-Trichloroethyl)-Beta-Lactam Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in patent CN112552285A for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 4-(2,2,2-trichloroethyl)-beta-lactam derivative delivered meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with our technical team to explore how this efficient synthesis route can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits specific to your volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, taking the first step towards a more efficient and reliable supply partnership for your critical beta-lactam intermediates.
