Advanced One-Pot Synthesis of 2,3,5,6-Tetrafluoro Terephthalyl Alcohol for Commercial Scale-Up
The global demand for high-performance fluorinated agrochemicals continues to surge, driving the need for efficient and scalable synthetic routes for key intermediates such as 2,3,5,6-tetrafluoro terephthalyl alcohol. This critical building block is essential for the manufacture of advanced pyrethroid insecticides like tefluthrin, which offer high efficacy and low toxicity profiles. A groundbreaking approach detailed in patent CN103304377A introduces a robust methodology that addresses longstanding challenges in fluorine chemistry. By leveraging a strategic protection-lithiation sequence, this invention transforms the production landscape, offering a pathway that is not only chemically elegant but also commercially viable for reliable agrochemical intermediate supplier networks seeking to optimize their portfolios.
The significance of this technology lies in its ability to bypass the limitations of conventional reduction and substitution chemistries. Traditional methods often suffer from harsh reaction conditions, difficult-to-source starting materials, or complex purification requirements that inflate costs and extend lead times. In contrast, the disclosed method utilizes a sequential one-pot protocol that maximizes atom economy and operational simplicity. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and controllable manufacturing processes, ensuring consistent quality for high-purity OLED material and agrochemical applications alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3,5,6-tetrafluoro terephthalyl alcohol has relied heavily on the reduction of tetrafluoro terephthalic acid or its derivatives. While this route is industrially established, it is plagued by significant logistical and technical hurdles. The primary starting material, tetrafluoro terephthalic acid, is often difficult to procure in bulk quantities with consistent quality, creating supply chain bottlenecks. Furthermore, the reduction step typically requires rigorous conditions, including high pressures and temperatures, which demand specialized equipment and pose safety risks. As illustrated in the reaction scheme below, the direct reduction pathway lacks flexibility and often results in variable yields due to the stability issues of the fluorinated aromatic ring under aggressive reducing environments.
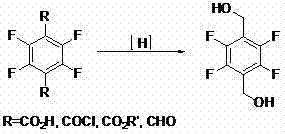
Alternative historical routes, such as the chloromethylation of 1,2,4,5-tetrafluorobenzene, present even greater environmental and operational challenges. These processes typically involve the use of concentrated sulfuric acid and chlorosulfonic acid, generating substantial volumes of hazardous acidic wastewater that require expensive treatment protocols. Additionally, the hydrolysis step in these sequences is prone to side reactions, leading to impurities that are difficult to remove. The combination of harsh acidic conditions, poor atom economy, and complex waste management makes these legacy methods increasingly unattractive for modern cost reduction in agrochemical intermediate manufacturing initiatives.
The Novel Approach
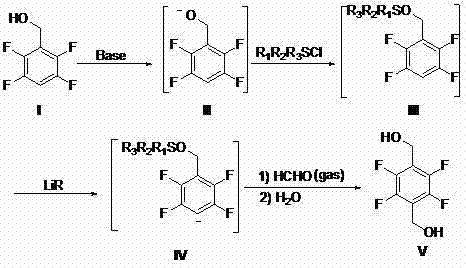
The innovative strategy presented in the patent data fundamentally reimagines the synthetic logic by starting from 2,3,5,6-tetrafluorobenzyl alcohol, a more accessible and manageable precursor. The core of this novelty lies in a four-step cascade that cleverly employs a silyl protecting group to mask the benzylic hydroxyl functionality. This protection is crucial as it prevents the highly reactive organolithium reagents from attacking the benzylic position, thereby directing the lithiation exclusively to the aromatic ring. Following lithiation, the introduction of formaldehyde gas allows for the precise installation of the second hydroxymethyl group. The entire sequence can be performed in a single reactor vessel without isolating intermediates, drastically reducing solvent usage and processing time.
This streamlined approach offers distinct advantages for commercial scale-up of complex fluorinated intermediates. By eliminating the need for intermediate workups, the process minimizes material loss and exposure to atmospheric moisture, which is critical for maintaining the integrity of organometallic species. The use of formaldehyde gas, generated in situ or introduced directly, ensures a clean reaction profile with minimal byproduct formation. Consequently, this method delivers superior product purity and yield compared to traditional routes, positioning it as a preferred choice for manufacturers aiming to enhance their supply chain reliability and reduce overall production costs through process intensification.
Mechanistic Insights into Silyl-Protection Directed Ortho-Lithiation
The success of this synthetic route hinges on the precise control of reactivity afforded by the silyl protection strategy. In the initial step, the reaction of 2,3,5,6-tetrafluorobenzyl alcohol with a strong base, such as n-butyllithium or sodium hydride, generates a benzyloxy anion. This anionic species is then immediately trapped with an alkyl chlorosilane, such as trimethylchlorosilane or tert-butyldimethylchlorosilane, to form a stable silyl ether. This transformation is mechanistically vital because the resulting silyl group acts as an electron-donating substituent that stabilizes the adjacent aromatic system while simultaneously blocking the benzylic carbon from nucleophilic attack. Without this protection, subsequent treatment with organolithium reagents would likely result in decomposition or non-selective metalation at the benzylic position.
Following protection, the introduction of a second equivalent of organolithium reagent at low temperatures (typically between -80°C and -30°C) effects the halogen-metal exchange or directed lithiation on the fluorinated aromatic ring. The presence of multiple fluorine atoms activates the ring towards nucleophilic attack but also requires careful temperature control to prevent defluorination. The resulting aryl lithium intermediate is highly reactive and is immediately quenched with formaldehyde gas. This electrophilic trapping step extends the carbon skeleton by one unit, installing the second hydroxymethyl group precisely at the para-position relative to the protected group. The final hydrolysis step removes the silyl protecting group under mild aqueous conditions, revealing the target diol structure with high fidelity and minimal impurity generation.
How to Synthesize 2,3,5,6-Tetrafluoro Terephthalyl Alcohol Efficiently
The execution of this synthesis requires strict adherence to anhydrous conditions and precise temperature management to ensure the stability of the organolithium intermediates. The process begins with the dissolution of the starting benzyl alcohol in an inert solvent such as tetrahydrofuran or ether, followed by cooling to cryogenic temperatures. The sequential addition of base, silyl chloride, and lithiating agent must be monitored carefully to maintain the reaction within the optimal thermal window. Detailed standard operating procedures regarding reagent stoichiometry, addition rates, and quenching protocols are essential for reproducing the high yields reported in the patent literature. For a comprehensive guide on the specific operational parameters and safety considerations, please refer to the standardized synthesis steps outlined below.
- React 2,3,5,6-tetrafluorobenzyl alcohol with a strong base in an inert solvent to form the benzyloxy anion.
- Treat the anion with alkyl chlorosilane to generate the protected 2,3,5,6-tetrafluoro benzyloxy silane intermediate.
- Perform lithiation using an organolithium reagent at low temperature to create the aryl lithium species.
- Introduce formaldehyde gas followed by hydrolysis to yield the final 2,3,5,6-tetrafluoro terephthalyl alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers transformative benefits that directly impact the bottom line. The shift away from scarce tetrafluoro terephthalic acid derivatives to more abundant benzyl alcohol precursors mitigates the risk of raw material shortages and price volatility. This change in feedstock strategy enhances supply security, allowing manufacturers to maintain consistent production schedules even during market fluctuations. Furthermore, the simplification of the process flow reduces the dependency on specialized high-pressure equipment, lowering capital expenditure requirements for new production lines and facilitating faster technology transfer between sites.
- Cost Reduction in Manufacturing: The implementation of a one-pot synthesis protocol significantly lowers operational expenditures by eliminating multiple isolation and purification steps. By avoiding the use of expensive transition metal catalysts often required for hydrogenation routes, the process reduces both material costs and the complexity of downstream metal removal. The qualitative improvement in process efficiency translates to substantial cost savings, as less energy is consumed for heating and cooling cycles, and solvent recovery becomes more straightforward due to the reduced number of unit operations involved in the workflow.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as 2,3,5,6-tetrafluorobenzyl alcohol ensures a more resilient supply chain compared to routes dependent on niche nitrile or acid derivatives. The robustness of the reaction conditions, which do not require extreme pressures, reduces the likelihood of unplanned downtime due to equipment failure. This reliability is crucial for meeting the stringent delivery timelines demanded by global agrochemical companies, ensuring that reducing lead time for high-purity intermediates becomes a tangible reality rather than just a goal.
- Scalability and Environmental Compliance: The avoidance of harsh chloromethylation reagents and the subsequent generation of acidic wastewater aligns perfectly with modern environmental regulations. This green chemistry advantage simplifies waste treatment protocols and reduces the environmental footprint of the manufacturing site. The process is inherently scalable, as the exothermic nature of the lithiation and formaldehyde addition steps can be managed effectively in larger reactors through controlled dosing, facilitating the commercial scale-up of complex fluorinated intermediates without compromising safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2,3,5,6-tetrafluoro terephthalyl alcohol using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical rationale behind the process. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing frameworks.
Q: What are the primary advantages of this new synthetic route over traditional reduction methods?
A: The novel route utilizes readily available 2,3,5,6-tetrafluorobenzyl alcohol as a starting material, avoiding the scarcity and high cost associated with tetrafluoro terephthalic acid derivatives. Furthermore, the one-pot procedure eliminates the need for intermediate isolation and purification, significantly streamlining the manufacturing process.
Q: How does the silyl protection strategy improve product purity?
A: By converting the hydroxyl group into a silyl ether prior to lithiation, the method prevents unwanted side reactions at the benzylic position. This selective protection ensures that the organolithium reagent reacts exclusively at the aromatic ring, leading to higher regioselectivity and improved final product purity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It operates under relatively mild temperature conditions compared to high-pressure hydrogenation routes and avoids the generation of large volumes of acidic wastewater typical of chloromethylation methods, making it environmentally compliant and operationally simpler for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5,6-Tetrafluoro Terephthalyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation agrochemicals and pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the most stringent purity specifications. We leverage state-of-the-art rigorous QC labs to verify every batch, guaranteeing that our 2,3,5,6-tetrafluoro terephthalyl alcohol is free from detrimental impurities that could affect downstream synthesis. Our commitment to technical excellence allows us to adapt quickly to evolving market demands while maintaining unwavering quality standards.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthetic route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this efficient manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive global market.
