Scalable Synthesis of 4-Halo Pyridine Bis-Imidazole Ligands for Advanced Optoelectronic Applications
Scalable Synthesis of 4-Halo Pyridine Bis-Imidazole Ligands for Advanced Optoelectronic Applications
The rapid advancement of optoelectronic materials and molecular catalysis relies heavily on the availability of sophisticated coordination ligands with precise structural tunability. A significant breakthrough in this domain is documented in Chinese Patent CN115403562A, which discloses a robust and efficient preparation method for 2',6'-di-2-(1-hydroimidazolyl)-4-halopyridine compounds. These tridentate ligands are critical building blocks for constructing supramolecular assemblies with transition metals, offering enhanced optical, electrical, and magnetic properties essential for next-generation display technologies and catalytic systems. The patent addresses a long-standing industry bottleneck by providing a pathway to introduce reactive halogen handles at the 4-position of the pyridine ring, a feature previously difficult to achieve without cumbersome synthetic routes. This innovation not only streamlines the production of high-purity intermediates but also opens new avenues for the derivatization of complex functional materials.
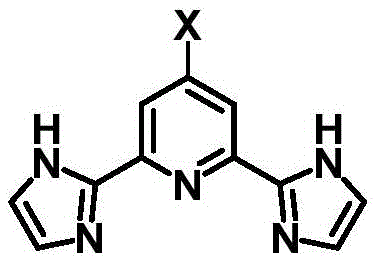
For R&D directors and procurement specialists seeking a reliable optoelectronic material supplier, understanding the mechanistic advantages of this new route is paramount. The traditional reliance on 4-oxo-1,4-dihydro-2,6-pyridinedicarboxylic acid as a starting material often necessitates harsh reagents like phosphorus pentabromide and involves multiple steps that complicate isolation. In contrast, the methodology outlined in CN115403562A utilizes 2,6-dibromo-4-aminopyridine, a commercially accessible feedstock, to achieve the target architecture through a logical sequence of cyanation, halogenation, and cyclization. This shift in synthetic strategy represents a paradigm change in how high-value pyridine bis-imidazole ligands are manufactured, promising substantial reductions in both operational complexity and environmental footprint for fine chemical producers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing N-H containing pyridine bis-imidazole ligands have been plagued by significant inefficiencies that hinder commercial viability. Prior art, such as methods described in Tetrahedron (2008), typically yields ligands with hydrogen atoms at the 4-position of the pyridine ring, which severely limits their utility for further functionalization. To overcome this, alternative routes utilizing 4-oxo-1,4-dihydro-2,6-pyridinedicarboxylic acid were developed, but these introduce severe processing challenges. The requirement for phosphorus pentabromide in methanol creates hazardous waste streams and demands rigorous safety protocols, while the subsequent multistep transformations often result in low overall yields. Furthermore, the separation and purification of intermediates in these conventional pathways frequently require complex column chromatography, a technique that is notoriously difficult to scale and economically prohibitive for bulk manufacturing. These factors collectively restrict the widespread adoption of such ligands in cost-sensitive industries like large-scale display manufacturing.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this landscape by leveraging a streamlined three-step sequence that prioritizes operational simplicity and scalability. By initiating the synthesis with 2,6-dibromo-4-aminopyridine, the process bypasses the need for dangerous phosphorylating agents entirely. The key innovation lies in the strategic installation of cyano groups followed by a versatile halogenation step, which preserves the reactivity of the 4-position for downstream cross-coupling reactions. Crucially, the patent emphasizes that neither the intermediate products nor the final target compound require complex column separation; instead, high-purity materials are obtained through simple filtration and washing procedures. This drastic simplification of the workup process not only accelerates production timelines but also significantly lowers the cost of goods sold (COGS), making high-performance coordination ligands accessible for broader industrial applications including molecular catalysis and self-assembly systems.
Mechanistic Insights into Nickel-Catalyzed Cyanation and Cyclization
The core of this synthetic breakthrough rests on a sophisticated yet practical catalytic system employed during the initial cyanation step. The conversion of 2,6-dibromo-4-aminopyridine to 2,6-dicyano-4-aminopyridine is achieved through a nucleophilic substitution reaction mediated by a synergistic catalyst mixture comprising nickel chloride hexahydrate, ferrocene, zinc powder, and N,N-dimethylaminopyridine (DMAP). This specific combination facilitates the Rosenmund-von Braun type reaction under relatively mild conditions of 80-90°C, ensuring high conversion rates while minimizing side reactions. The presence of ferrocene and zinc powder likely serves to maintain the active oxidation state of the nickel catalyst and scavenge halide byproducts, thereby sustaining catalytic turnover. Following this, the amino group is transformed into a halogen (bromine or iodine) via a diazotization-like mechanism using copper bromide or an iodine/potassium iodide system, respectively. This step is critical as it installs the reactive handle necessary for future derivatization without compromising the integrity of the newly formed nitrile groups.
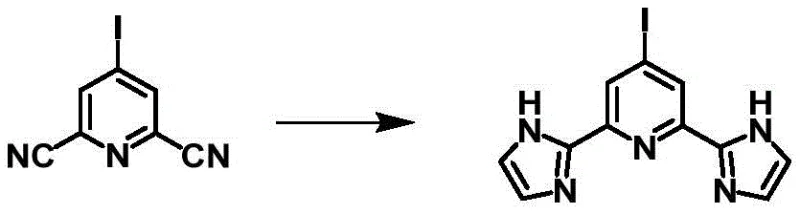
The final transformation involves a delicate balance of nucleophilic addition and acid-mediated cyclization to construct the imidazole rings. The 2,6-dicyano-4-halopyridine intermediate undergoes nucleophilic addition with sodium methoxide in methanol, generating an imidate species that subsequently reacts with 2,2-dimethoxyethylamine. This nucleophilic substitution is carefully controlled at temperatures between 45-55°C to prevent decomposition. The subsequent cyclization, driven by hydrochloric acid at 75-85°C, closes the imidazole rings to yield the final tridentate ligand. This mechanism ensures that the N-H protons on the imidazole rings remain intact, a feature essential for the ligand's ability to dissociate under weakly alkaline conditions and coordinate with transition metals to form negatively charged complexes. Such mechanistic precision guarantees the production of ligands with consistent electronic properties, vital for reproducible performance in optoelectronic devices.
How to Synthesize 2',6'-di-2-(1-hydroimidazolyl)-4-halopyridine Efficiently
Implementing this synthesis route requires strict adherence to the molar ratios and temperature profiles specified in the patent to ensure optimal yield and purity. The process begins with the cyanation step, where precise stoichiometry of the nickel and zinc additives is crucial for driving the reaction to completion without excessive metal contamination. Following the isolation of the dicyano intermediate, the halogenation step must be monitored closely, typically via TLC, to ensure full conversion of the amine before proceeding to the final cyclization. The final step involves a sequential addition of reagents where the timing of acid addition is critical for successful ring closure. For detailed operational parameters and safety guidelines regarding the handling of cyanide sources and strong acids, please refer to the standardized protocol below.
- Perform nucleophilic substitution of 2,6-dibromo-4-aminopyridine with zinc cyanide using a Ni/Fe/Zn catalyst system at 80-90°C to yield 2,6-dicyano-4-aminopyridine.
- Convert the amino group to a halogen (Br or I) via diazotization using copper bromide or iodine/potassium iodide at 70-80°C to obtain 2,6-dicyano-4-halopyridine.
- Execute nucleophilic addition with sodium methoxide followed by substitution with 2,2-dimethoxyethylamine and final cyclization in hydrochloric acid at 75-85°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this patented methodology offers transformative benefits that directly address the pain points of sourcing complex organic intermediates. The elimination of column chromatography is perhaps the most significant advantage, as it removes a major bottleneck in production throughput and drastically reduces the consumption of high-purity solvents. This simplification translates into a more robust supply chain capable of meeting tight delivery schedules without the variability associated with manual purification techniques. Furthermore, the use of commodity chemicals like 2,6-dibromo-4-aminopyridine and zinc cyanide ensures that raw material availability remains stable, mitigating the risk of supply disruptions common with exotic reagents. For procurement managers, this means a more predictable cost structure and the ability to negotiate better terms based on the efficiency of the manufacturing process.
- Cost Reduction in Manufacturing: The streamlined workflow significantly lowers operational expenditures by removing the need for expensive silica gel and large volumes of chromatographic solvents. The ability to isolate products via simple filtration reduces labor hours and energy consumption associated with solvent recovery systems. Additionally, the high atom economy of the cyanation and cyclization steps minimizes waste generation, leading to lower disposal costs and a smaller environmental footprint. These cumulative efficiencies allow for a substantial reduction in the unit cost of the final ligand, making it a viable option for cost-sensitive applications in the display and lighting sectors.
- Enhanced Supply Chain Reliability: By relying on a synthetic route that avoids specialized, hard-to-source reagents, manufacturers can maintain consistent production schedules even during market fluctuations. The robustness of the nickel-catalyzed system ensures high reproducibility batch-to-batch, which is critical for maintaining quality standards in downstream electronics manufacturing. The simplified purification process also reduces the risk of batch failure due to purification errors, thereby enhancing the overall reliability of the supply chain. This stability is essential for long-term partnerships where continuity of supply is a primary contractual obligation.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, as the reaction conditions (temperatures of 70-90°C) are easily manageable in standard stainless steel reactors. The absence of phosphorus pentabromide eliminates the generation of corrosive phosphoric acid waste, simplifying effluent treatment and ensuring compliance with stringent environmental regulations. The use of aqueous workups and common organic solvents like methanol and ethyl acetate further facilitates waste management and solvent recycling. This alignment with green chemistry principles not only future-proofs the manufacturing process against tightening regulations but also appeals to end-users prioritizing sustainable sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized pyridine bis-imidazole ligands. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating these materials into your specific product development pipelines.
Q: How does this synthesis method improve purification compared to traditional routes?
A: Unlike conventional methods requiring complex column chromatography, this patented route allows intermediates and final products to be isolated via simple filtration and washing, significantly reducing solvent waste and processing time.
Q: What is the reactivity of the 4-position halogen in the final ligand?
A: The 4-position bromine or iodine exhibits high cross-coupling activity, enabling facile derivatization through Suzuki or Sonogashira reactions to create complex multi-armed ligands for specific optoelectronic functions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of readily available starting materials like 2,6-dibromo-4-aminopyridine and the elimination of tedious purification steps make this method highly amenable to commercial scale-up and cost-effective manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2',6'-di-2-(1-hydroimidazolyl)-4-halopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance ligands play in the advancement of optoelectronic and catalytic technologies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market deployment is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2',6'-di-2-(1-hydroimidazolyl)-4-halopyridine meets the exacting standards required for sensitive electronic applications. We are committed to delivering not just a chemical product, but a reliable supply solution that supports your innovation goals.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this streamlined manufacturing method. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume needs. Let us collaborate to drive down costs and accelerate the development of next-generation functional materials together.
