Revolutionizing Gemifloxacin Side Chain Production: A Low-Pressure Catalytic Breakthrough for Commercial Scale
Revolutionizing Gemifloxacin Side Chain Production: A Low-Pressure Catalytic Breakthrough for Commercial Scale
The pharmaceutical landscape for fourth-generation fluoroquinolone antibiotics is constantly evolving, driven by the need for more efficient and cost-effective synthetic routes for critical intermediates. Patent CN113773240B introduces a transformative preparation method for the gemifloxacin side chain compound, specifically 4-aminomethyl-3-methoxyiminopyrrolidine, which serves as a pivotal building block for the potent antibacterial agent Gemifloxacin. This innovation addresses long-standing challenges in the industry by replacing hazardous, high-energy processes with a mild, catalytic hydrogenation strategy that operates under significantly reduced pressure. The strategic implementation of this technology allows manufacturers to achieve superior purity profiles while minimizing operational risks associated with high-pressure reactors and toxic reagents.
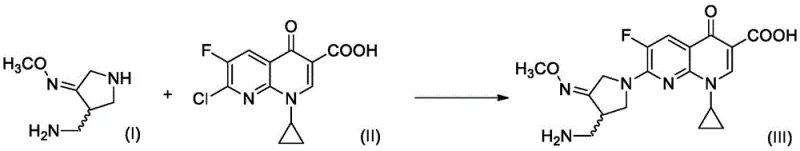
Gemifloxacin, known commercially as Factive, represents a significant advancement in treating community-acquired pneumonia and acute bacterial exacerbations of chronic bronchitis. Its efficacy is largely attributed to the unique lipophilicity and structural configuration of its 7-position side chain, which enhances activity against Gram-positive bacteria including multidrug-resistant strains. The synthesis of this specific side chain has historically been a bottleneck due to complex multi-step sequences and difficult purification requirements. The new methodology detailed in this patent offers a streamlined alternative that not only improves yield but also aligns with modern green chemistry principles by reducing waste and energy consumption. For R&D directors and process chemists, this represents a viable pathway to optimize the overall supply chain for this critical antibiotic.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art technologies for synthesizing the gemifloxacin side chain have been plagued by significant operational inefficiencies and safety hazards that hinder large-scale production. Existing routes, such as those disclosed in European patent EP0688771A1, typically rely on strong reducing agents like lithium aluminum hydride and oxidizing reagents such as Jones reagent. These chemicals are not only expensive and hazardous to handle but also generate substantial amounts of toxic waste, complicating environmental compliance and disposal protocols. Furthermore, these traditional pathways often necessitate rigorous purification steps, including column chromatography, which are notoriously difficult to scale up and result in significant product loss and extended production cycles.
Another prevalent issue in conventional synthesis is the requirement for extreme reaction conditions, particularly regarding hydrogenation pressure. Some established methods, like those protected under patent CN1149192C, demand hydrogen pressures exceeding 4.0 MPa to achieve the necessary reduction of nitrile groups. Operating at such high pressures requires specialized, costly equipment and poses inherent safety risks in a manufacturing environment. Additionally, the use of Raney nickel in conjunction with palladium carbon in these high-pressure scenarios often leads to inconsistent selectivity, resulting in complex impurity profiles that require further downstream processing. These factors collectively drive up the cost of goods sold and extend lead times, creating vulnerabilities in the supply chain for essential antibiotic intermediates.
The Novel Approach
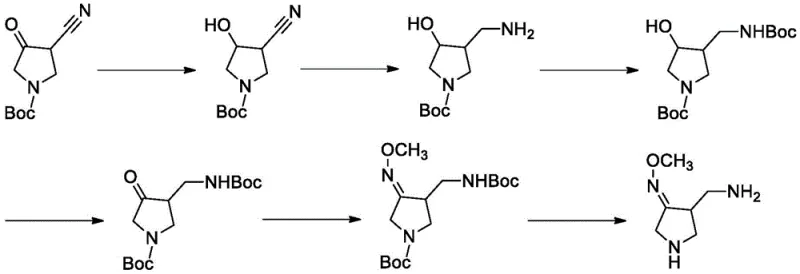
The innovative process described in patent CN113773240B fundamentally reengineers the synthesis workflow by introducing a mild, low-pressure catalytic system that eliminates the need for hazardous stoichiometric reagents. The core of this advancement lies in a three-step sequence starting from 1-N-tert-butyloxycarbonyl-4-cyano-3-pyrrolidone, proceeding through a controlled oximation and a sophisticated hydrogenation-protection cascade. By utilizing palladium carbon as a heterogeneous catalyst under a hydrogen atmosphere of merely 0.1 to 0.5 MPa, the process achieves high conversion rates at near-atmospheric conditions. This drastic reduction in pressure not only enhances operational safety but also allows the reaction to be performed in standard stainless steel reactors, significantly lowering capital expenditure requirements for manufacturing facilities.
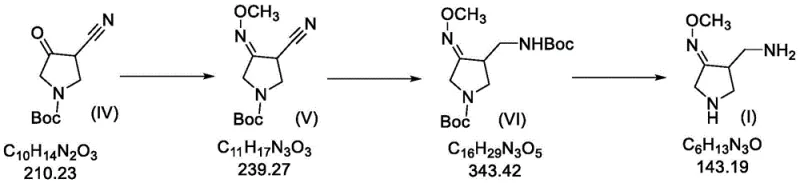
Moreover, this novel approach integrates an in-situ protection strategy that effectively manages reaction selectivity and minimizes byproduct formation. Instead of isolating unstable intermediates, the process involves the sequential addition of Boc anhydride during the hydrogenation phase. This clever manipulation of reaction kinetics ensures that the newly formed amine groups are immediately protected, preventing unwanted side reactions such as dimerization or over-reduction. The result is a crude product of such high purity that it can often proceed to the next step without intermediate purification, thereby collapsing the overall process timeline. This efficiency translates directly into reduced solvent usage, lower energy consumption, and a markedly smaller environmental footprint compared to legacy methods.
Mechanistic Insights into Pd/C Catalyzed Stepwise Hydrogenation
The success of this synthesis hinges on a precise understanding of the catalytic hydrogenation mechanism and the timing of reagent addition. In the critical second step, the nitrile group of the methoxime intermediate is reduced to an amine via an imine intermediate. Under standard conditions, this highly reactive imine species is prone to various degradation pathways. However, the patented method introduces Boc anhydride into the reaction mixture after the initial reduction phase but before the reaction is complete. This creates a dynamic equilibrium where the transient imine or primary amine is rapidly capped with the tert-butoxycarbonyl group. This mechanistic intervention stabilizes the molecule and directs the reaction pathway exclusively toward the desired protected aminomethyl product.
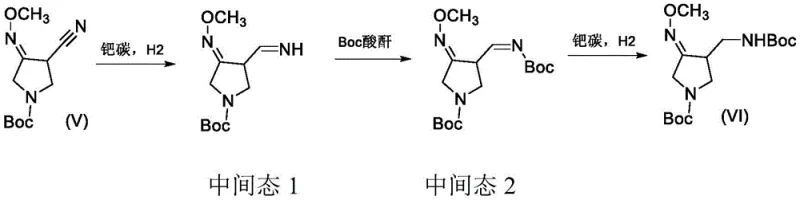
Furthermore, the choice of palladium carbon as the catalyst plays a pivotal role in controlling the stereochemistry and impurity profile of the final product. The patent data indicates that optimizing the loading of the catalyst and the specific pressure range (preferably 0.1-0.2 MPa) is crucial for maximizing yield while minimizing the formation of over-reduced byproducts. The neutral reaction environment maintained during this step prevents the hydrolysis of the sensitive methoxime functionality, which is a common failure point in acidic or basic hydrogenation conditions. By maintaining these mild parameters, the process ensures that the chiral integrity of the pyrrolidine ring is preserved, which is essential for the biological activity of the final gemifloxacin API. This level of mechanistic control demonstrates a deep optimization of reaction engineering tailored for robust industrial application.
How to Synthesize 4-Aminomethyl-3-Methoxyiminopyrrolidine Efficiently
The synthesis of this high-value pharmaceutical intermediate is achieved through a logical sequence of oximation, controlled hydrogenation, and deprotection. The process begins with the reaction of a cyano-pyrrolidone precursor with methoxyamine hydrochloride to install the critical methoxime motif. Following this, the core transformation occurs via a palladium-catalyzed hydrogenation where Boc anhydride is introduced in a specific temporal window to trap the reactive amine. Finally, global deprotection yields the target free base or salt. This streamlined workflow eliminates the need for isolation of unstable intermediates and avoids the use of column chromatography, making it ideal for kilogram-to-ton scale production. Detailed standardized synthesis steps are provided in the guide below.
- React 1-N-tert-butyloxycarbonyl-4-cyano-3-pyrrolidone with methoxyamine hydrochloride under pyridine catalysis to form the methoxime intermediate.
- Perform a stepwise hydrogenation using palladium carbon at low pressure (0.1-0.5 MPa), introducing Boc anhydride mid-reaction to protect the amine and prevent side reactions.
- Remove the protecting groups using methanesulfonic acid to isolate the final gemifloxacin side chain compound with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this novel synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift from high-pressure hydrogenation (>4.0 MPa) to low-pressure conditions (0.1-0.5 MPa) fundamentally alters the infrastructure requirements for production. Facilities no longer need to invest in or maintain specialized high-pressure autoclaves, which are expensive to certify and operate. This reduction in technical barriers allows for greater flexibility in manufacturing site selection and significantly lowers the barrier to entry for secondary suppliers, thereby enhancing supply chain resilience and reducing the risk of single-source bottlenecks.
- Cost Reduction in Manufacturing: The elimination of hazardous stoichiometric reagents like lithium aluminum hydride results in substantial raw material cost savings and reduces the expense associated with hazardous waste disposal. Furthermore, the ability to bypass column chromatography purification steps drastically cuts down on solvent consumption and silica gel costs, which are major drivers of variable costs in fine chemical manufacturing. The simplified work-up procedure, often involving direct crystallization or filtration, reduces labor hours and equipment occupancy time, leading to a more favorable cost of goods sold structure for the final intermediate.
- Enhanced Supply Chain Reliability: By utilizing widely available and stable starting materials such as 1-N-tert-butyloxycarbonyl-4-cyano-3-pyrrolidone and methoxyamine hydrochloride, the process mitigates the risk of raw material shortages. The robustness of the catalytic system, which tolerates mild conditions and neutral pH, ensures consistent batch-to-batch quality, reducing the likelihood of production failures or off-spec material that could disrupt downstream API synthesis schedules. This reliability is critical for maintaining continuous supply lines for essential antibiotics, especially in the face of global demand fluctuations.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on heterogeneous catalysis and standard solvent systems like methanol. The absence of heavy metal contaminants from stoichiometric reductants simplifies the purification of the final API, aiding in meeting stringent regulatory limits for residual metals. Additionally, the reduced energy footprint from operating at lower temperatures and pressures aligns with corporate sustainability goals and increasingly strict environmental regulations, future-proofing the manufacturing process against potential carbon taxes or emission caps.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a clear picture of the technology's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer or licensing agreements.
Q: How does this new synthesis route improve upon traditional methods for gemifloxacin intermediates?
A: Traditional methods often require harsh reducing agents like lithium aluminum hydride or high-pressure hydrogenation (>4 MPa) with complex purification steps like column chromatography. This patented route utilizes mild catalytic hydrogenation at significantly lower pressures (0.1-0.5 MPa) and eliminates the need for column separation, drastically simplifying industrial operations.
Q: What is the key mechanism for controlling impurities during the hydrogenation step?
A: The process employs a unique stepwise feeding strategy where Boc anhydride is added after the initial reduction of the nitrile group to an imine. This immediate in-situ protection prevents over-reduction or polymerization side reactions, ensuring high conversion rates and superior product purity without extensive downstream processing.
Q: Is this synthesis method scalable for commercial API production?
A: Yes, the method is specifically designed for industrial scalability. By operating at near-atmospheric pressure and using common solvents like methanol, it avoids the need for specialized high-pressure reactors. The simplified work-up procedures, such as direct crystallization instead of chromatography, make it highly suitable for large-scale manufacturing of fluoroquinolone antibiotics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aminomethyl-3-Methoxyiminopyrrolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the broader context of antibiotic manufacturing. Our team of expert process chemists has thoroughly analyzed this patented route and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. We are equipped with state-of-the-art hydrogenation reactors capable of operating safely at the specified low pressures and rigorous QC labs to ensure stringent purity specifications are met for every batch. Our commitment to technical excellence ensures that we can deliver high-quality gemifloxacin side chain intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for fluoroquinolone antibiotics. By leveraging our expertise in catalytic hydrogenation and process optimization, we can help you achieve significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term production goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
