Advanced Manufacturing of Tafluprost Intermediates: Overcoming Fluorination Challenges for Commercial Scale-up
Advanced Manufacturing of Tafluprost Intermediates: Overcoming Fluorination Challenges for Commercial Scale-up
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex active pharmaceutical ingredients (APIs), particularly for high-value ophthalmic treatments like Tafluprost. A significant breakthrough in this domain is detailed in Chinese Patent CN112457277B, which outlines a novel preparation method that fundamentally reorders the synthetic sequence to maximize yield and purity. Unlike traditional approaches that struggle with side reactions during the fluorination of unsaturated intermediates, this innovative protocol introduces fluorine atoms at an earlier stage of the synthesis. By utilizing bis(2-methoxyethyl)aminosulfur trifluoride (BAST) to fluorinate a saturated ketone precursor prior to double bond formation, the method effectively circumvents the notorious issue of double bond addition side reactions. This strategic adjustment not only streamlines the purification process but also ensures the production of high-purity intermediates essential for regulatory compliance in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tafluprost and its analogs has relied on routes where the fluorinated side chain is constructed or modified after the formation of the critical carbon-carbon double bond. As illustrated in prior art such as the work by Shirasawa et al., the fluorination step often employs reagents like trifluoro-sulfuryl morpholine or DAST on a substrate that already contains an alkene functionality. 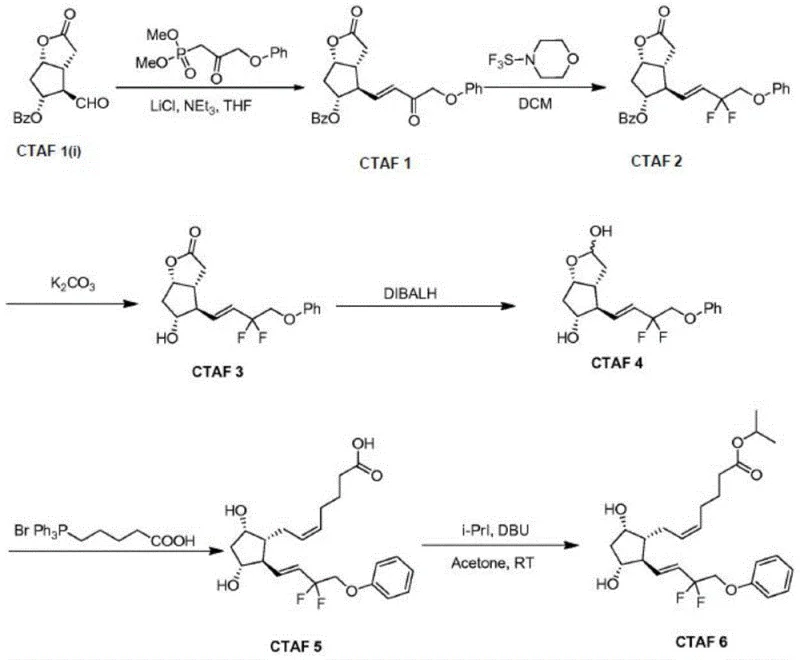 This sequence presents a significant chemical vulnerability: the electrophilic nature of fluorinating agents can lead to unwanted addition reactions across the electron-rich double bond. Consequently, manufacturers face the formation of omega-side chain double bond addition by-products, which are structurally similar to the target molecule and notoriously difficult to separate. This results in compromised conversion rates, lower overall yields, and a complex impurity profile that necessitates expensive and time-consuming chromatographic purification, thereby inflating the cost of goods sold (COGS) and extending lead times for a reliable pharmaceutical intermediate supplier.
This sequence presents a significant chemical vulnerability: the electrophilic nature of fluorinating agents can lead to unwanted addition reactions across the electron-rich double bond. Consequently, manufacturers face the formation of omega-side chain double bond addition by-products, which are structurally similar to the target molecule and notoriously difficult to separate. This results in compromised conversion rates, lower overall yields, and a complex impurity profile that necessitates expensive and time-consuming chromatographic purification, thereby inflating the cost of goods sold (COGS) and extending lead times for a reliable pharmaceutical intermediate supplier.
The Novel Approach
The methodology disclosed in CN112457277B represents a paradigm shift by decoupling the fluorination event from the presence of the alkene. The core innovation involves treating a bromo-ketone precursor, specifically 1-bromo-3-phenoxypropan-2-one derivatives, with BAST to install the difluoro motif while the carbon backbone remains saturated. 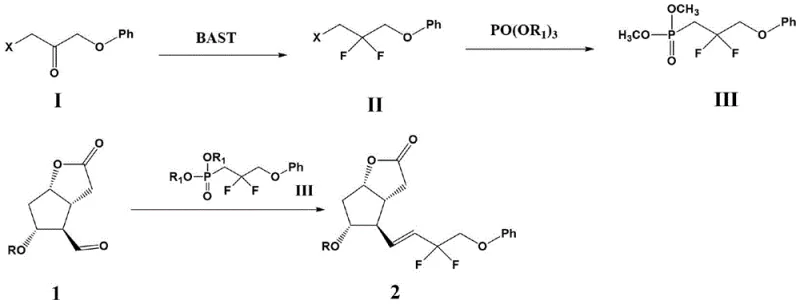 Once the stable difluoro-bromide is secured, it is converted into a phosphonate ester via reaction with trialkyl phosphate. This phosphonate then serves as the nucleophile in a Horner-Wadsworth-Emmons (HWE) olefination with the protected Corey lactone aldehyde. By delaying the formation of the double bond until after the harsh fluorination conditions are complete, the process completely eliminates the risk of fluorine adding across the alkene. This logical reordering of synthetic steps ensures that the resulting Tafluprost intermediate is obtained with exceptional purity, often exceeding 99%, and in high yields, providing a robust foundation for downstream processing.
Once the stable difluoro-bromide is secured, it is converted into a phosphonate ester via reaction with trialkyl phosphate. This phosphonate then serves as the nucleophile in a Horner-Wadsworth-Emmons (HWE) olefination with the protected Corey lactone aldehyde. By delaying the formation of the double bond until after the harsh fluorination conditions are complete, the process completely eliminates the risk of fluorine adding across the alkene. This logical reordering of synthetic steps ensures that the resulting Tafluprost intermediate is obtained with exceptional purity, often exceeding 99%, and in high yields, providing a robust foundation for downstream processing.
Mechanistic Insights into BAST-Mediated Fluorination and HWE Olefination
The success of this route hinges on the precise control of two critical transformations: the selective deoxofluorination and the subsequent stereoselective olefination. In the first stage, BAST acts as a mild yet effective fluorinating agent, replacing the carbonyl oxygen of the ketone precursor with two fluorine atoms. The reaction is typically conducted in solvents like dichloromethane or acetonitrile, starting at low temperatures (0-5°C) to manage exotherms, followed by heating to 40-60°C to drive the reaction to completion. The use of BAST is particularly advantageous as it offers a balance of reactivity and stability superior to older reagents, minimizing decomposition pathways. Following fluorination, the resulting gem-difluoro bromide undergoes an Arbuzov-type reaction with trialkyl phosphites (such as trimethyl or triethyl phosphate), often catalyzed by potassium iodide (KI), to generate the corresponding phosphonate. This phosphonate is then activated by a strong base, typically sodium hydride (NaH), to form a carbanion that attacks the aldehyde of the Corey lactone. 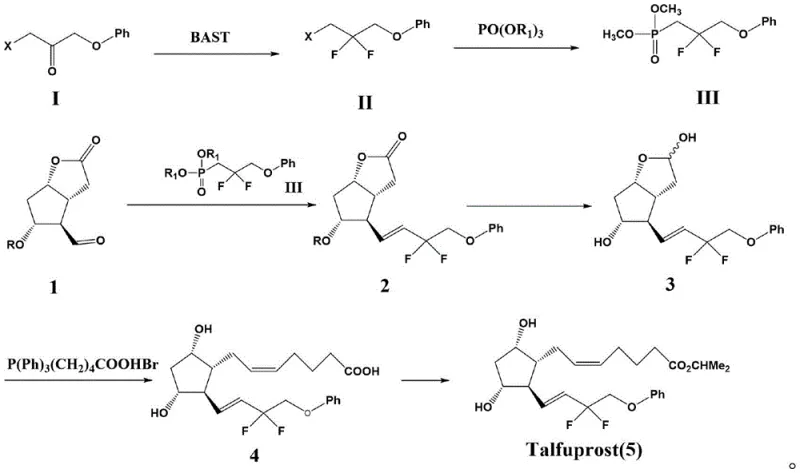 The HWE reaction conditions are meticulously optimized, with temperatures maintained between 35-50°C to ensure the formation of the desired E-isomer (trans-alkene) while suppressing Z-isomer formation. This mechanistic precision is vital for maintaining the biological activity of the final prostaglandin analog.
The HWE reaction conditions are meticulously optimized, with temperatures maintained between 35-50°C to ensure the formation of the desired E-isomer (trans-alkene) while suppressing Z-isomer formation. This mechanistic precision is vital for maintaining the biological activity of the final prostaglandin analog.
From an impurity control perspective, this mechanism offers distinct advantages. In conventional routes, the coexistence of fluorinating agents and alkenes generates a spectrum of halogenated by-products that share similar polarity and retention times with the target, complicating isolation. In contrast, the intermediates generated in this novel pathway—specifically the saturated difluoro-ketone and the phosphonate—are chemically distinct from the final alkene product. Any unreacted starting materials or minor side products from the fluorination step are easily removed before the HWE reaction even begins. Furthermore, the HWE reaction itself is highly selective; by controlling the base strength and temperature, the formation of the thermodynamically stable trans-alkene is favored. This inherent selectivity reduces the burden on downstream purification units, allowing for simpler work-up procedures involving standard aqueous washes and crystallization rather than preparative HPLC, which is a crucial factor for cost reduction in API manufacturing.
How to Synthesize Tafluprost Intermediate Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding stoichiometry, temperature, and solvent selection to ensure reproducibility at scale. The process begins with the dissolution of the bromo-ketone in a chlorinated solvent, followed by the controlled addition of BAST. After the fluorination is complete and the phosphonate is generated in situ or isolated, the critical coupling step involves the slow addition of the phosphonate solution to a suspension of sodium hydride in tetrahydrofuran (THF). The detailed standardized operating procedures, including specific molar ratios and quenching protocols necessary to achieve the reported >90% yields, are outlined below for technical reference.
- Fluorinate 1-bromo-3-phenoxypropan-2-one using BAST at 0-5°C followed by heating to 40-60°C to obtain the difluoro-bromide intermediate with high purity.
- React the difluoro-bromide with trialkyl phosphate (e.g., trimethyl phosphate) in the presence of KI to generate the phosphonate ester reagent.
- Perform the Horner-Wadsworth-Emmons (HWE) reaction between the phosphonate ester and protected Corey lactone aldehyde using NaH in THF to form the key alkene intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible operational efficiencies and risk mitigation. The primary value driver is the significant simplification of the purification train. By eliminating the formation of difficult-to-separate double bond addition by-products, the process reduces the reliance on resource-intensive chromatographic separations. This directly correlates to a reduction in solvent consumption, silica gel usage, and processing time, all of which contribute to substantial cost savings in the manufacturing of high-purity prostaglandin analogs. Furthermore, the use of BAST, while a specialized reagent, allows for a more predictable reaction profile compared to the thermal instability often associated with DAST, thereby enhancing process safety and reducing the risk of batch failures due to reagent decomposition.
- Cost Reduction in Manufacturing: The elimination of side reactions means that a higher percentage of raw materials are converted into the desired product, drastically improving atom economy. Since the impurities generated are easier to remove, the need for multiple recrystallizations or complex column chromatography is minimized. This streamlined workflow lowers the variable costs per kilogram, making the production of Tafluprost intermediates more economically viable and allowing for competitive pricing strategies in the generic ophthalmic market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which operate effectively at moderate temperatures (0-60°C) and utilize common organic solvents like THF and acetone, ensures high batch-to-batch consistency. This reliability is critical for maintaining continuous supply lines to downstream API manufacturers. Additionally, the high purity of the intermediate (>99%) reduces the likelihood of downstream processing issues, ensuring that the final API meets stringent regulatory specifications without unexpected delays, thus securing the supply chain for critical glaucoma medications.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to pilot plant and commercial production without requiring exotic equipment or extreme cryogenic conditions. The reduction in hazardous waste generation, stemming from higher yields and fewer purification steps, aligns with modern green chemistry principles. This facilitates easier environmental permitting and waste disposal management, reducing the regulatory burden and potential liabilities associated with large-scale chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industry standards.
Q: Why is early-stage fluorination superior for Tafluprost synthesis?
A: Traditional methods fluorinate the molecule after the double bond is formed, leading to unwanted addition side reactions across the alkene. By fluorinating the saturated ketone precursor first, this patent eliminates those side reactions, significantly improving yield and purity.
Q: What are the safety advantages of using BAST over DAST in this process?
A: While DAST (Diethylaminosulfur trifluoride) is effective, it can be thermally unstable. BAST (Bis(2-methoxyethyl)aminosulfur trifluoride) offers a safer profile with comparable reactivity, allowing for robust temperature control between 0°C and 60°C without compromising reaction efficiency.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process utilizes standard reagents like NaH and common solvents (THF, Acetone, DCM) and avoids cryogenic conditions or exotic catalysts. The high yields (>90% in key steps) and simplified purification make it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tafluprost Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has thoroughly analyzed the route disclosed in CN112457277B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are fully equipped to implement this high-efficiency fluorination strategy, leveraging our state-of-the-art facilities and rigorous QC labs to ensure that every batch of Tafluprost intermediate meets stringent purity specifications and regulatory requirements. Our commitment to technical excellence ensures that we can deliver complex organic intermediates with the consistency and quality demanded by top-tier pharmaceutical companies.
We invite you to collaborate with us to optimize your supply chain for ophthalmic APIs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of scientific rigor and commercial viability.
