Advanced Synthetic Route for 3-Amino-2-Oxazolone: Enhancing Purity and Scalability for Global Supply Chains
The global pharmaceutical and agrochemical sectors face increasing regulatory pressure to monitor residual contaminants, particularly metabolites of banned substances like furazolidone. Patent CN111303064B, published in August 2022, introduces a robust and efficient synthetic methodology for producing 3-amino-2-oxazolone (AOZ), the critical protein-bound metabolite used as a reference standard in residue analysis. This technical breakthrough addresses long-standing industry pain points regarding low yields and purification difficulties associated with traditional synthesis routes. By leveraging a strategic Boc-protection sequence, the disclosed method ensures high structural integrity and exceptional purity, which are paramount for analytical accuracy in food safety testing. For procurement leaders and R&D directors, understanding this pathway is essential for securing a reliable supply of high-quality intermediates that meet stringent international compliance standards.
The significance of this patent extends beyond mere academic interest; it represents a viable commercial pathway for manufacturing complex heterocyclic intermediates. The process transforms simple, readily available feedstocks into a high-value target molecule through a controlled four-step sequence. This approach not only mitigates the risks associated with handling unstable hydrazine derivatives but also streamlines the downstream processing requirements. As we delve deeper into the technical specifics, it becomes evident that this methodology offers a superior alternative for reliable pharma intermediate supplier networks aiming to enhance their portfolio of veterinary drug metabolite standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-amino-2-oxazolone has been plagued by significant technical hurdles that impede large-scale production and cost-effectiveness. Conventional routes often involve direct cyclization strategies that lack selectivity, leading to the formation of numerous side products and polymeric impurities that are notoriously difficult to separate. These impurities compromise the purity of the final product, rendering it unsuitable for use as a certified reference material in high-performance liquid chromatography (HPLC) or mass spectrometry applications. Furthermore, traditional methods frequently rely on harsh reaction conditions or expensive reagents that drive up the operational expenditure, making cost reduction in pharmaceutical intermediates manufacturing a challenging objective. The instability of free hydrazine intermediates in older protocols also poses safety risks and storage limitations, further complicating the supply chain logistics for manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN111303064B employs a sophisticated protection-deprotection strategy that fundamentally alters the reaction landscape. By introducing a tert-butyloxycarbonyl (Boc) group early in the synthesis, the process stabilizes the reactive hydrazine moiety, preventing unwanted side reactions and decomposition. This strategic modification allows for milder reaction conditions and significantly cleaner reaction profiles, which translates directly to easier workup procedures and higher isolated yields. The use of commodity chemicals such as ethylene glycol and diethyl carbonate ensures that the raw material costs remain low, facilitating substantial economic advantages. This novel approach effectively resolves the purification bottlenecks of the past, enabling the production of AOZ with purity levels reaching 99%, which is critical for meeting the rigorous specifications demanded by global regulatory agencies.
Mechanistic Insights into Boc-Protection and Cyclization Strategy
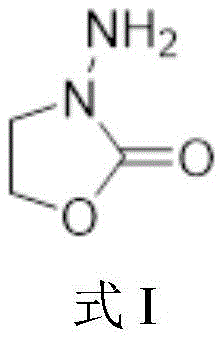
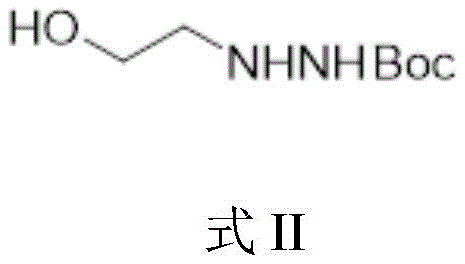
The core innovation of this synthetic route lies in its meticulous control over the reactivity of the hydrazine functional group. The mechanism begins with the conversion of ethylene glycol to 2-bromoethanol using phosphorus tribromide, a standard halogenation that sets the stage for nucleophilic substitution. Subsequently, the introduction of tert-butyloxycarbonyl-hydrazine in the presence of a base facilitates a clean SN2 substitution, yielding the stable Boc-protected intermediate shown in Formula II. This protection step is crucial as it masks the nucleophilicity of one nitrogen atom, directing the subsequent cyclization reaction with diethyl carbonate specifically to form the desired oxazolone ring without polymerization. The cyclization proceeds via a nucleophilic attack of the unprotected nitrogen on the carbonyl carbon of the carbonate, followed by intramolecular ring closure and elimination of ethanol, driven by the thermodynamic stability of the five-membered heterocyclic ring.
Furthermore, the final deprotection step utilizes mild acidic conditions to remove the Boc group, regenerating the free amine without degrading the sensitive oxazolone core. This selectivity is vital for maintaining the structural integrity of the metabolite, ensuring that the final product accurately mimics the biological residue found in animal tissues. The entire mechanistic pathway is designed to minimize the generation of hazardous waste and maximize atom economy, aligning with modern green chemistry principles. For R&D teams, understanding these mechanistic nuances is key to troubleshooting potential scale-up issues and optimizing reaction parameters for commercial scale-up of complex pharmaceutical intermediates. The robustness of this chemistry ensures consistent batch-to-batch reproducibility, a non-negotiable requirement for suppliers serving the analytical standards market.
How to Synthesize 3-Amino-2-Oxazolone Efficiently
The practical implementation of this synthesis involves a straightforward four-step protocol that balances reaction efficiency with operational safety. The process initiates with the bromination of ethylene glycol, followed by the coupling with protected hydrazine, cyclization with diethyl carbonate, and final acid hydrolysis. Each step has been optimized to utilize common solvents like ethanol and ethyl acetate, reducing the need for specialized equipment or exotic reagents. The detailed standardized synthesis steps below outline the specific molar ratios, temperatures, and workup procedures required to achieve the high yields reported in the patent data, providing a clear roadmap for laboratory and pilot plant execution.
- React ethylene glycol with phosphorus tribromide (PBr3) under reflux to generate 2-bromoethanol.
- Couple 2-bromoethanol with tert-butyloxycarbonyl-hydrazine in the presence of a base to form Boc-protected 2-hydrazinoethanol.
- Cyclize the protected hydrazine with diethyl carbonate using an alkali catalyst to obtain Boc-protected 3-amino-2-oxazolone.
- Perform acid-mediated deprotection using hydrochloric acid to yield the final high-purity 3-amino-2-oxazolone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling strategic benefits that extend beyond simple technical feasibility. The reliance on bulk commodity chemicals significantly de-risks the supply chain, as these materials are widely available from multiple global vendors, preventing bottlenecks associated with specialty reagents. The simplified purification process reduces the consumption of silica gel and solvents during chromatography, leading to a leaner manufacturing footprint and lower waste disposal costs. Moreover, the high purity achieved directly from the synthesis minimizes the need for extensive recrystallization or reprocessing, accelerating the time-to-market for finished reference standards. This efficiency translates into a more resilient supply chain capable of responding rapidly to fluctuating demand in the food safety and veterinary monitoring sectors.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of inexpensive starting materials like ethylene glycol drastically lower the raw material cost base. The high yield at each step minimizes material loss, ensuring that the overall process economics are highly favorable compared to legacy methods. Additionally, the reduced need for complex purification infrastructure lowers capital expenditure requirements for production facilities.
- Enhanced Supply Chain Reliability: By utilizing a synthetic pathway based on stable, shelf-stable intermediates, the risk of raw material degradation during storage and transport is significantly mitigated. The robustness of the Boc-protected intermediates allows for potential stockpiling, providing a buffer against market volatility. This stability ensures a continuous and predictable supply of the final API intermediate, crucial for maintaining uninterrupted testing operations for regulatory bodies.
- Scalability and Environmental Compliance: The reaction conditions, ranging from room temperature to moderate heating, are easily transferable from laboratory glassware to industrial reactors without significant engineering modifications. The use of common organic solvents simplifies solvent recovery and recycling processes, aiding in compliance with increasingly strict environmental regulations. This scalability ensures that production can be ramped up to meet surging demand for veterinary drug residue testing without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3-amino-2-oxazolone. These insights are derived directly from the patent specifications and industry best practices, aimed at clarifying the value proposition of this advanced synthetic method for stakeholders across the value chain.
Q: Why is high-purity 3-Amino-2-Oxazolone (AOZ) critical for regulatory compliance?
A: AOZ is the primary protein-bound metabolite of furazolidone, a banned nitrofuran antibiotic. Regulatory bodies require highly pure AOZ standards to accurately detect residue levels in food products, ensuring compliance with strict zero-tolerance policies regarding veterinary drug abuse.
Q: How does this novel synthetic route improve upon conventional methods?
A: Traditional methods often suffer from low overall yields and complex purification challenges. This patented 4-step route utilizes a Boc-protection strategy that stabilizes reactive intermediates, significantly improving isolation efficiency and achieving consistent high purity (up to 99%) suitable for analytical reference standards.
Q: Is this manufacturing process scalable for industrial production?
A: Yes, the process relies on commodity chemicals like ethylene glycol and diethyl carbonate and operates under mild conditions (room temperature to 70°C). The absence of exotic catalysts and the use of standard solvents like ethanol and ethyl acetate make it highly amenable to scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-2-Oxazolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity metabolites in ensuring global food safety and regulatory compliance. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-amino-2-oxazolone meets the exacting standards required for analytical reference materials. Our commitment to quality assurance means that you can rely on us for a stable supply of this essential intermediate, supporting your mission to detect and prevent veterinary drug residues effectively.
We invite you to collaborate with us to optimize your sourcing strategy for this critical compound. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain resilience and reduce your overall procurement costs.
