Scalable Synthesis of 4,5-Dihydro-Pyrazolo[3,4-c]Pyridin-2-Ones for Commercial API Production
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex heterocyclic scaffolds, particularly those serving as critical intermediates for cardiovascular therapeutics. Patent CN101065379A discloses a novel and highly efficient process for the preparation of 4,5-dihydro-pyrazolo[3,4-c]pyridin-2-ones, which are potent Factor Xa inhibitors. This technology represents a significant advancement over prior art by streamlining the construction of the fused ring system through a strategic 1,3-dipolar cycloaddition followed by a sequence of reduction and cyclization steps. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates. The method described eliminates several inefficiencies associated with traditional syntheses, offering a clearer path from raw materials to the active drug substance. By leveraging specific catalytic systems and dehydration protocols, this process ensures consistent quality and improved yield profiles, making it an attractive option for commercial-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fused pyrazolo-pyridone systems has been plagued by multi-step sequences that suffer from low overall yields and the accumulation of difficult-to-remove impurities. Conventional routes often rely on harsh conditions that compromise the integrity of sensitive functional groups, necessitating extensive purification protocols that drive up production costs and extend lead times. Many traditional methods utilize stoichiometric amounts of hazardous reagents or require cryogenic temperatures that are energy-intensive and challenging to maintain on an industrial scale. Furthermore, the lack of regioselectivity in older cycloaddition strategies frequently results in isomeric mixtures, complicating downstream processing and reducing the final purity of the API intermediate. These bottlenecks create significant supply chain vulnerabilities, as any deviation in reaction parameters can lead to batch failures. For procurement managers, these inefficiencies translate into higher unit costs and unpredictable delivery schedules, hindering the ability to meet market demand for life-saving anticoagulant medications.
The Novel Approach
In contrast, the methodology outlined in CN101065379A introduces a streamlined approach that addresses these historical challenges through a carefully orchestrated sequence of reactions. The core innovation lies in the use of a 1,3-dipolar cycloaddition between a specifically substituted hydrazone and a dipolarophile, which rapidly constructs the bicyclic framework with high fidelity. This step is conducted in the presence of substituted amine bases, such as triethylamine or DBU, in aprotic solvents like toluene, allowing for moderate temperatures that are easily manageable in standard glass-lined reactors. Following the formation of the core, the process employs catalytic hydrogenation to reduce the nitro functionality, avoiding the use of toxic metal reductants. The subsequent cyclization to form the lactam ring is facilitated by chemical dehydrating agents, ensuring high conversion rates without the need for extreme thermal conditions. This integrated strategy minimizes the number of isolation steps, thereby reducing material loss and solvent consumption. 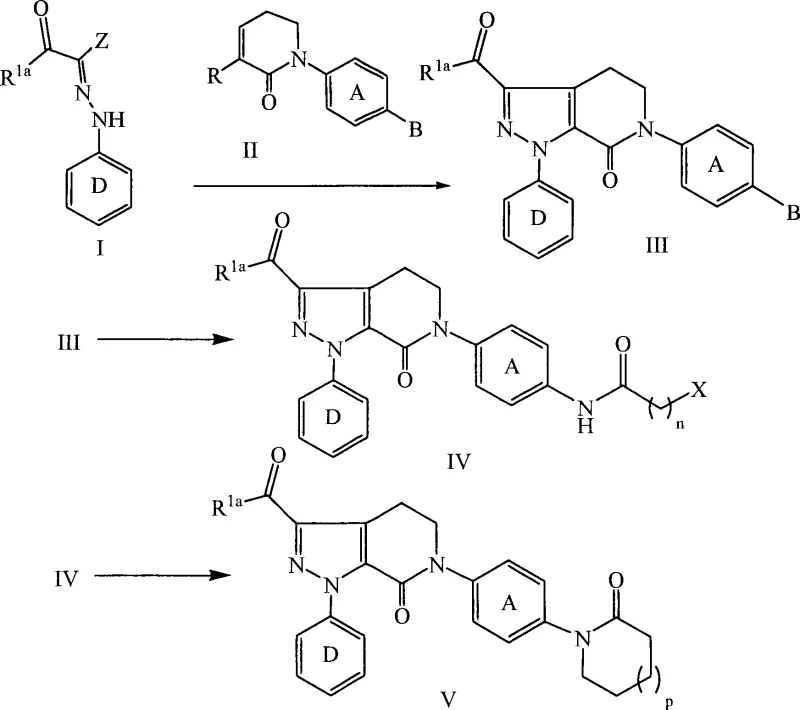
Mechanistic Insights into 1,3-Dipolar Cycloaddition and Cyclization
The success of this synthetic route hinges on the precise control of the 1,3-dipolar cycloaddition reaction, which forms the foundational 4,5-dihydro-pyrazolo[3,4-c]pyridin-2-one skeleton. Mechanistically, the hydrazone acts as the 1,3-dipole, reacting with the electron-deficient dipolarophile to close the pyrazole ring. The choice of base is critical here; non-nucleophilic tertiary amines facilitate the generation of the reactive dipole species without interfering with the electrophilic centers of the dipolarophile. Solvent selection also plays a pivotal role, with toluene providing an optimal balance of solubility and boiling point to drive the reaction to completion while allowing for easy removal post-reaction. The subsequent transformation involves the reduction of the nitro group on the phenyl ring to an aniline derivative. This is typically achieved using palladium catalysts supported on carbon or alumina under hydrogen pressure. The choice of support, such as Pd/Al2O3, can influence the filtration characteristics and metal leaching profiles, which are crucial for meeting stringent heavy metal specifications in pharmaceutical products. 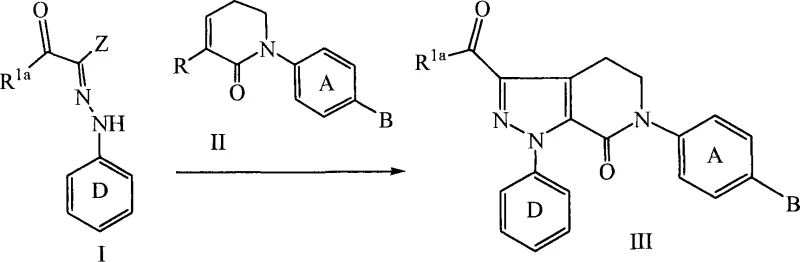
Following reduction, the newly formed amine undergoes acylation with an alkyl acid halide to introduce the side chain necessary for the final ring closure. The most critical step in terms of impurity control is the intramolecular cyclization to form the lactam ring. This transformation requires the removal of water to drive the equilibrium towards the product. The patent specifies the use of strong acids like trifluoroacetic acid (TFA) in combination with orthoformates, such as triethyl orthoformate. This combination acts as a chemical dehydrating agent, scavenging water generated during the amidation and cyclization processes. The use of alkoxide bases, such as sodium ethoxide, in the final stages ensures the completion of the ring closure and neutralizes acidic byproducts. This meticulous control over reaction conditions minimizes the formation of open-chain byproducts and ensures a high-purity profile for the final intermediate, which is essential for downstream API synthesis.
How to Synthesize 4,5-Dihydro-Pyrazolo[3,4-c]Pyridin-2-Ones Efficiently
Implementing this synthesis on a commercial scale requires adherence to standardized operating procedures that prioritize safety and reproducibility. The process begins with the preparation of the hydrazone starting material, followed by the cycloaddition step which sets the stereochemistry and core structure. Operators must monitor reaction temperatures closely, particularly during the addition of bases and acid chlorides, to prevent exotherms. The hydrogenation step requires specialized equipment capable of handling pressurized hydrogen safely, along with rigorous protocols for catalyst handling and disposal. After the cyclization, the product is typically isolated via crystallization or filtration, with washing steps designed to remove residual solvents and salts. Detailed standard operating procedures for each unit operation are essential to ensure consistency across batches.
- Perform 1,3-dipolar cycloaddition between hydrazone and dipolarophile using amine bases in aprotic solvents.
- Reduce the nitro group to an amine using catalytic hydrogenation with Pd/C or Pd/Al2O3.
- Execute intramolecular cyclization using chemical dehydrating agents like TFA and orthoformates to form the lactam ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages that directly impact the bottom line and supply chain resilience. The reduction in the number of synthetic steps compared to conventional methods translates to lower operational expenditures and reduced capital investment in equipment. By minimizing isolation steps, the process significantly cuts down on solvent usage and waste generation, aligning with modern environmental compliance standards and reducing disposal costs. The use of commercially available reagents and catalysts ensures that raw material sourcing is stable and not subject to the volatility of specialty chemical markets. Furthermore, the robustness of the reaction conditions allows for flexibility in manufacturing locations, enabling companies to diversify their supply base and mitigate geopolitical risks. The high purity of the intermediate reduces the burden on downstream purification, accelerating the overall timeline from synthesis to final drug product release.
- Cost Reduction in Manufacturing: The streamlined nature of this process eliminates the need for expensive transition metal catalysts in the cyclization step, relying instead on organic dehydrating agents that are cost-effective and easy to handle. The ability to use crude reaction mixtures in subsequent steps without extensive purification significantly reduces material loss and processing time. This efficiency leads to substantial cost savings in terms of raw material consumption and utility usage, making the final intermediate more price-competitive in the global market. Additionally, the high yield of the cycloaddition step maximizes the throughput of the manufacturing facility, allowing for better asset utilization.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as toluene, triethylamine, and palladium on carbon ensures that the supply chain is not dependent on single-source suppliers for exotic reagents. This diversity in sourcing options enhances the reliability of supply, reducing the risk of production stoppages due to material shortages. The scalability of the process, demonstrated by examples ranging from gram to kilogram scales, indicates that it can be readily adapted to multi-ton production volumes without significant re-engineering. This scalability provides procurement managers with the confidence to secure long-term contracts and plan inventory levels more accurately.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing solvents that can be recovered and recycled, thereby minimizing the environmental footprint. The avoidance of stoichiometric heavy metal reductants reduces the load of toxic waste, simplifying compliance with increasingly stringent environmental regulations. The robust nature of the chemistry allows for safe scale-up in standard pharmaceutical manufacturing equipment, reducing the need for specialized containment facilities. This ease of scale-up ensures that production can be ramped up quickly to meet surges in demand without compromising on safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of these critical pharmaceutical intermediates. Understanding these aspects helps stakeholders make informed decisions about integrating this technology into their production pipelines. The answers are derived from the detailed experimental data and process descriptions provided in the patent literature.
Q: What is the key reaction mechanism for forming the pyrazolo-pyridone core?
A: The core structure is formed via a 1,3-dipolar cycloaddition between a hydrazone derivative and a functionalized dipolarophile, typically catalyzed by substituted amine bases in solvents like toluene.
Q: How is the nitro group reduced in this process?
A: The nitro group is reduced to an amino group using catalytic hydrogenation with palladium catalysts such as Pd/C or Pd/Al2O3 in aprotic solvents like THF or NMP.
Q: What dehydrating agents are used for the final cyclization?
A: The cyclization to form the lactam ring utilizes strong acids like trifluoroacetic acid (TFA) combined with orthoformates such as triethyl orthoformate to remove water effectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,5-Dihydro-Pyrazolo[3,4-c]Pyridin-2-One Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to bring this sophisticated synthetic route to life on a commercial scale. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We understand the critical importance of stringent purity specifications in the pharmaceutical industry and operate rigorous QC labs to verify every batch against the highest standards. Our commitment to quality assurance means that every intermediate we supply is accompanied by comprehensive analytical data, giving you confidence in the performance of your final drug product.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your project goals. Let us be your partner in delivering high-quality chemical solutions that drive your business forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
