Advanced Synthesis of Halogen-Substituted Benzenedimethanol for Global Pharmaceutical and Agrochemical Supply Chains
Advanced Synthesis of Halogen-Substituted Benzenedimethanol for Global Pharmaceutical and Agrochemical Supply Chains
The global demand for high-purity fluorinated and halogenated building blocks continues to surge, driven by the relentless innovation in crop protection and medicinal chemistry sectors. Patent CN101370759B introduces a transformative methodology for the preparation of halogen-substituted benzenedimethanols, specifically addressing the critical bottlenecks associated with traditional reduction pathways. This technology leverages a precise borohydride reduction strategy followed by a thermally controlled hydrochloric acid workup, achieving yields that were previously unattainable with conventional acidic hydrolysis methods. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and economically viable manufacturing processes for complex aromatic diols. The ability to produce these intermediates with minimal byproduct formation directly impacts the cost of goods sold (COGS) for downstream active pharmaceutical ingredients (APIs) and agrochemical actives.
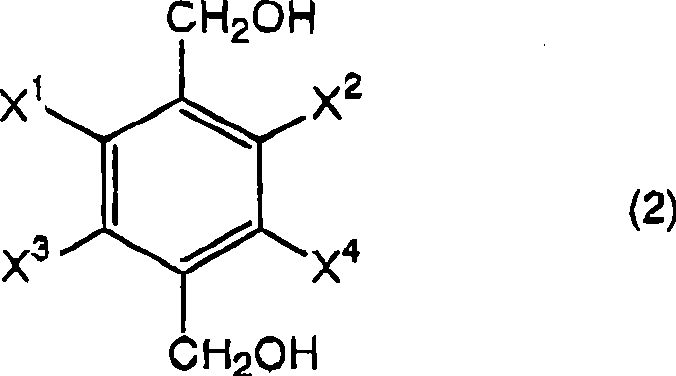
As a reliable agrochemical intermediate supplier and partner in fine chemical synthesis, understanding the nuances of this patent is essential for securing long-term supply continuity. The core innovation lies not merely in the choice of reducing agent, but in the rigorous thermal management during the quenching phase, which prevents the stalling of the reaction at the mono-alcohol stage. This technical insight report dissects the mechanistic advantages and commercial implications of adopting this route for large-scale production. By integrating these findings into our manufacturing roadmap, we can offer clients a robust alternative to legacy processes that often suffer from inconsistent quality and excessive waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in US6759558, typically rely on a multi-step sequence involving reduction followed by treatment with alkylating agents, sulfuric acid, or sulfonic acids. These conventional approaches are fraught with significant operational hazards and efficiency losses that burden the supply chain. The use of strong mineral acids like sulfuric acid necessitates specialized corrosion-resistant equipment and generates substantial quantities of acidic wastewater, complicating environmental compliance and increasing disposal costs. Furthermore, the reliance on alkylating agents introduces toxicity concerns and requires stringent safety protocols to protect personnel, thereby inflating the operational expenditure of the manufacturing facility. From a chemical perspective, these harsh conditions often lead to non-selective reactions, resulting in complex impurity profiles that are difficult and expensive to remove during purification.
The Novel Approach
In stark contrast, the novel approach detailed in CN101370759B utilizes a streamlined protocol where halogen-substituted terephthalic acid is reacted directly with a borohydride compound in an organic solvent. 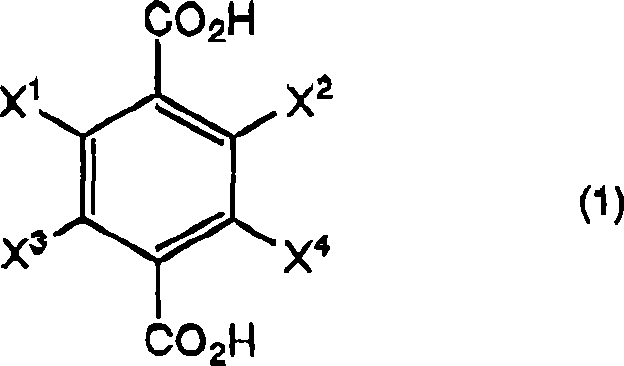 The critical differentiator is the subsequent contact of the reaction mixture with hydrogen chloride at a specifically controlled temperature range of 40 to 70°C. This method elegantly bypasses the need for hazardous alkylating reagents and aggressive sulfuric acid treatments, significantly simplifying the process flow. By optimizing the acid workup conditions, the process ensures the complete conversion of intermediate borate esters into the desired diol, thereby maximizing atom economy. This shift not only enhances the safety profile of the operation but also drastically reduces the environmental footprint, aligning perfectly with modern green chemistry principles demanded by top-tier multinational corporations.
The critical differentiator is the subsequent contact of the reaction mixture with hydrogen chloride at a specifically controlled temperature range of 40 to 70°C. This method elegantly bypasses the need for hazardous alkylating reagents and aggressive sulfuric acid treatments, significantly simplifying the process flow. By optimizing the acid workup conditions, the process ensures the complete conversion of intermediate borate esters into the desired diol, thereby maximizing atom economy. This shift not only enhances the safety profile of the operation but also drastically reduces the environmental footprint, aligning perfectly with modern green chemistry principles demanded by top-tier multinational corporations.
Mechanistic Insights into Borohydride Reduction and Acidolytic Workup
The chemical transformation hinges on the nucleophilic attack of the borohydride anion on the carbonyl carbons of the terephthalic acid derivative, forming a tetrahedral intermediate that eventually collapses to the alcohol oxidation state. However, the true sophistication of this patent lies in the management of the resulting borate ester species. In standard reductions, simply adding water or mild acid at ambient temperatures often leaves a fraction of the material trapped as stable borate complexes or partially reduced mono-alcohols. The patent data explicitly demonstrates that maintaining the system between 40°C and 70°C during the introduction of hydrogen chloride provides the necessary activation energy to cleave these robust boron-oxygen bonds efficiently. This thermal window is narrow enough to prevent degradation of the sensitive halogenated aromatic ring yet sufficient to drive the hydrolysis to completion, ensuring that the final product stream is dominated by the target benzenedimethanol rather than partially reduced contaminants.
Impurity control is paramount for pharmaceutical applications, and this mechanism offers a distinct advantage in minimizing the formation of 4-carboxy-2,3,5,6-tetrafluorobenzyl alcohol, a stubborn byproduct observed in comparative examples performed at lower temperatures. When the acid contact is conducted at 25 to 30°C, as shown in the patent's comparative data, the reaction stalls, leaving nearly 40% of the starting material unreacted and generating significant amounts of the mono-alcohol impurity. By adhering to the elevated temperature protocol, the kinetic barrier for the second reduction step is overcome, ensuring a uniform conversion across the entire batch. This mechanistic precision translates directly to a cleaner crude product, reducing the load on downstream purification units like crystallization or chromatography columns, which is a critical factor for scaling up production without compromising quality specifications.
How to Synthesize Halogen-Substituted Benzenedimethanol Efficiently
Implementing this synthesis route requires careful attention to solvent selection and addition rates to manage the exothermic nature of the borohydride reaction. The patent recommends ether solvents, particularly dimethoxyethane, due to their ability to solubilize both the inorganic borohydride salts and the organic acid substrate effectively. The process begins with the suspension of the reducing agent, followed by the controlled addition of the halogenated terephthalic acid to maintain thermal stability. Once the reduction is complete, the transition to the acid workup phase must be executed with precision, ensuring the temperature remains within the optimal 40 to 70°C window to guarantee high yield. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational parameters.
- Suspend sodium borohydride in an ether solvent such as dimethoxyethane and heat the mixture to approximately 50°C to activate the reducing agent.
- Slowly add the halogen-substituted terephthalic acid substrate to the reaction mixture while maintaining the temperature between 50°C and 60°C for several hours to ensure complete reduction.
- Quench the reaction by adding concentrated hydrochloric acid while strictly maintaining the temperature between 40°C and 70°C to maximize yield and minimize byproduct formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound strategic benefits that extend beyond simple yield improvements. The elimination of sulfuric acid and alkylating agents from the process map removes a major source of supply chain volatility, as these commodities are often subject to strict regulatory controls and price fluctuations. By switching to a borohydride and hydrochloric acid system, manufacturers can secure a more stable and predictable raw material base, reducing the risk of production stoppages due to reagent shortages. Furthermore, the simplified waste stream, which no longer contains heavy sulfate loads or toxic alkylating residues, significantly lowers the cost and complexity of effluent treatment, contributing to substantial overall cost savings in pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic increase in reaction yield, which directly lowers the consumption of expensive halogenated starting materials. By avoiding the formation of hard-to-separate byproducts, the need for extensive recycling loops or destructive purification steps is minimized, leading to a more efficient use of reactor time and utilities. Additionally, the removal of corrosive sulfuric acid reduces the maintenance frequency of reactor vessels and piping, extending the lifespan of capital equipment and lowering long-term depreciation costs. These cumulative efficiencies result in a leaner production model that can withstand market pressure without sacrificing margin.
- Enhanced Supply Chain Reliability: Utilizing widely available reagents like sodium borohydride and hydrochloric acid mitigates the risk associated with sourcing specialized or hazardous chemicals that may face transportation restrictions. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling suppliers to respond more agilely to fluctuating demand from downstream API manufacturers. This reliability is crucial for maintaining just-in-time inventory levels and ensuring that critical drug development timelines are not compromised by raw material delays. Consequently, partners can rely on a consistent flow of high-quality intermediates to support their own commercial launches.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the severe exotherms and gas evolution issues often associated with sulfuric acid quenches on a large scale. The moderate temperature requirements and the use of common organic solvents facilitate a smooth transition from pilot plant to commercial production without the need for exotic engineering controls. From an environmental standpoint, the reduction in hazardous waste generation aligns with increasingly stringent global regulations, ensuring that the manufacturing site remains compliant and avoids potential fines or shutdowns. This sustainability profile enhances the brand value of the supply chain, appealing to end-clients who prioritize green chemistry in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, derived directly from the experimental data and claims within the patent documentation. Understanding these specifics is vital for process engineers aiming to replicate the high yields reported in the intellectual property. The answers provided reflect the optimal conditions identified by the inventors to ensure successful technology transfer and commercial viability.
Q: Why is the temperature control critical during the hydrochloric acid workup step?
A: Maintaining the temperature between 40°C and 70°C during the acid contact phase is essential to drive the decomposition of intermediate borate esters. Lower temperatures, such as 25°C, result in incomplete conversion and significant accumulation of mono-alcohol byproducts, drastically reducing the overall yield.
Q: What solvents are preferred for this borohydride reduction process?
A: Ether solvents are highly preferred for this transformation, with dimethoxyethane (DME) demonstrating superior performance in terms of solubility and reaction kinetics. Aromatic hydrocarbons like toluene can be used as co-solvents during the workup phase to facilitate phase separation.
Q: How does this method compare to previous alkylation-based routes?
A: This method eliminates the need for hazardous alkylating agents and strong mineral acids like sulfuric acid used in prior art. By utilizing a direct borohydride reduction followed by a controlled hydrochloric acid quench, the process achieves significantly higher purity and simplifies the downstream purification workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogen-Substituted Benzenedimethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate thermal balances required for this borohydride reduction are maintained even at the largest volumes. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch of halogen-substituted benzenedimethanol meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to technical precision ensures that the impurity profiles remain consistent, providing our clients with the confidence needed to file regulatory dossiers without delay.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this optimized route for your specific supply chain. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals. Let us be your partner in delivering high-performance intermediates that drive innovation and efficiency in your final products.
