Advanced Chemical Synthesis of Hydroxytyrosol: Scalable Routes for Global Pharmaceutical Supply Chains
Introduction to Patent CN101891595A and Technological Breakthroughs
The global demand for high-purity hydroxytyrosol, a potent polyphenolic antioxidant widely utilized in pharmaceutical and nutraceutical formulations, has necessitated the development of robust and economically viable synthetic pathways beyond traditional extraction methods. Patent CN101891595A presents a significant advancement in this domain by detailing a novel preparation method that optimizes both yield and cost-efficiency through a strategic four-step organic synthesis. This intellectual property outlines a process that begins with the readily available 3,4-dihydroxybenzaldehyde, circumventing the prohibitive costs associated with prior art starting materials such as 3,4-dihydroxyphenylacetic acid. By integrating benzyl protection strategies with efficient condensation and reduction reactions, the disclosed method achieves a commendable overall yield ranging from 50% to 60%, establishing a new benchmark for industrial feasibility. For R&D directors and procurement specialists, this patent represents a critical opportunity to secure a stable supply of this high-value active ingredient, decoupling production from the volatility of agricultural harvests and ensuring consistent quality specifications required for regulatory compliance in sensitive therapeutic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the sourcing of hydroxytyrosol has been plagued by significant economic and logistical challenges inherent to both natural extraction and earlier chemical synthesis routes. Traditional extraction from olive mill wastewater or plant tissues, while perceived as natural, suffers from extremely low recovery rates, complex purification requirements to remove impurities, and severe dependency on seasonal crop yields, leading to unpredictable supply chain disruptions. Furthermore, existing chemical synthesis methods often relied on expensive precursors like 3,4-dihydroxyphenylacetic acid or tyrosol, which drastically inflated the cost of goods sold (COGS) and rendered large-scale manufacturing economically unfeasible for mass-market applications. These conventional pathways frequently involved harsh reaction conditions or multiple protection-deprotection cycles that accumulated waste and reduced the final throughput, creating bottlenecks for manufacturers aiming to meet the surging global demand for antioxidant therapeutics. The reliance on such costly starting materials not only strained procurement budgets but also limited the ability of suppliers to offer competitive pricing structures necessary for market penetration in the highly price-sensitive dietary supplement sector.
The Novel Approach
In stark contrast to these legacy limitations, the methodology described in patent CN101891595A introduces a streamlined synthetic architecture that prioritizes raw material accessibility and reaction efficiency. By selecting 3,4-dihydroxybenzaldehyde as the foundational building block, the process leverages a commodity chemical that is significantly more affordable and abundantly available in the global fine chemical market compared to its acid or alcohol counterparts. The innovation lies in the clever application of benzyl groups to protect the reactive phenolic hydroxyls early in the sequence, allowing subsequent transformations to proceed with high selectivity and minimal side reactions. This approach effectively mitigates the formation of difficult-to-remove impurities, thereby simplifying downstream purification and enhancing the overall purity profile of the final API intermediate. The strategic design of this route ensures that each step contributes maximally to the molecular complexity required, eliminating unnecessary operations and resulting in a cumulative yield that supports commercial viability without compromising on the stringent quality standards demanded by pharmaceutical regulators.
Mechanistic Insights into Benzyl-Protection and Catalytic Deprotection Strategy
The core of this synthetic success relies on a sophisticated interplay of protection chemistry and catalytic hydrogenation, designed to navigate the reactivity of the catechol moiety. The initial step involves the nucleophilic substitution of the hydroxyl protons on 3,4-dihydroxybenzaldehyde with benzyl groups using benzyl bromide in the presence of a base like potassium carbonate. This transformation is critical as it masks the highly oxidative-prone catechol system, preventing polymerization or degradation during the subsequent carbon-carbon bond-forming steps. Following protection, the aldehyde undergoes a condensation reaction with N-methylaniline acetonitrile, facilitated by a strong base such as sodium hydride, to form an acrylonitrile intermediate which is subsequently hydrolyzed under acidic conditions to yield the protected phenylacetic acid derivative. This sequence effectively extends the carbon chain by two atoms, constructing the ethyl backbone essential for the hydroxytyrosol structure while maintaining the integrity of the aromatic ring system throughout the rigorous acidic and basic environments.
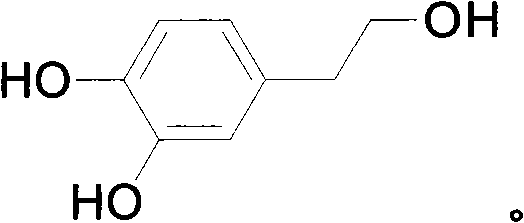
The final stages of the mechanism focus on the precise reduction of the carboxylic acid functionality and the orthogonal removal of the protecting groups. The reduction of the 3,4-dibenzyloxyphenylacetic acid to the corresponding alcohol is achieved using potent hydride reducing agents such as lithium borohydride or lithium aluminum hydride, which selectively target the carbonyl group without affecting the ether linkages. The culmination of the synthesis is the catalytic hydrogenation step employing palladium on carbon (Pd/C), which serves a dual purpose of cleaving the robust benzyl ether bonds to regenerate the free phenolic hydroxyls while simultaneously saturating any potential olefinic impurities. This mechanistic elegance ensures that the final product, hydroxytyrosol, is obtained with a high degree of chemical purity, as the catalytic deprotection is exceptionally clean and generates toluene as the only major byproduct, which is easily removed during workup. For technical teams, understanding this mechanism highlights the process's robustness against impurity carryover, a key factor in validating the method for GMP manufacturing environments.
How to Synthesize Hydroxytyrosol Efficiently
Implementing this synthesis at an industrial scale requires strict adherence to the optimized reaction parameters defined in the patent to ensure maximum yield and safety. The process is designed to be operationally straightforward, utilizing common solvents like acetone, THF, and toluene, which facilitates easy solvent recovery and recycling in a production setting. Detailed standard operating procedures (SOPs) would dictate specific molar ratios, such as the 1:2 to 1:4 ratio of aldehyde to base in the protection step, and precise temperature controls during the exothermic reduction phases. To assist our technical partners in evaluating the feasibility of this route for their specific production lines, we have outlined the critical operational milestones below.
- Protect the hydroxyl groups of 3,4-dihydroxybenzaldehyde using benzyl bromide to form 3,4-dibenzyloxybenzaldehyde.
- Condense the protected aldehyde with N-methylaniline acetonitrile followed by acidic hydrolysis to yield 3,4-dibenzyloxyphenylacetic acid.
- Reduce the carboxylic acid intermediate to the corresponding alcohol using lithium borohydride or similar reducing agents.
- Perform catalytic hydrogenation using Pd/C to remove benzyl protecting groups and finalize the hydroxytyrosol structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthetic route offers profound advantages in terms of cost stability and supply chain resilience. The primary driver of cost reduction is the substitution of expensive niche starting materials with 3,4-dihydroxybenzaldehyde, a bulk chemical with a mature and competitive global supply market. This shift fundamentally alters the cost structure of the final product, allowing for significant margin improvements or more aggressive pricing strategies without sacrificing quality. Furthermore, the elimination of complex extraction equipment and the reliance on standard batch reactor chemistry means that capital expenditure (CAPEX) for setting up production lines is minimized, as existing multipurpose facilities can be readily adapted for this synthesis. The simplified purification workflow, resulting from the high selectivity of the benzyl protection strategy, also translates to reduced consumption of chromatography media and solvents, further driving down the variable costs associated with manufacturing.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the use of commodity-grade reagents and the avoidance of precious metal catalysts in the early stages, reserving the expensive palladium catalyst only for the final deprotection step where it can be efficiently recovered and recycled. By streamlining the synthesis to four distinct steps with high individual yields, the process minimizes material loss at each stage, ensuring that the theoretical yield is closely approached in practice. This efficiency directly correlates to a lower cost per kilogram of the active ingredient, providing a substantial competitive edge in markets where price sensitivity is a key determinant of supplier selection. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower overall utility cost profile for the manufacturing facility.
- Enhanced Supply Chain Reliability: Diversifying the source of hydroxytyrosol from agriculture-dependent extraction to chemical synthesis mitigates the risks associated with crop failures, climate change impacts on olive groves, and seasonal availability fluctuations. Chemical synthesis offers the distinct advantage of on-demand production capability, allowing inventory levels to be adjusted dynamically in response to market demand without the long lead times inherent to harvesting and processing biomass. The reliance on stable, shelf-stable chemical intermediates ensures that production can continue uninterrupted even during off-seasons, guaranteeing a continuous supply to downstream pharmaceutical and nutraceutical clients. This reliability is crucial for long-term supply agreements where consistency of supply is often valued higher than marginal price differences.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as filtration, distillation, and crystallization that are well-understood and easily expanded from pilot to commercial scale. The use of Pd/C hydrogenation, while requiring safety protocols for handling hydrogen gas, is a standard industry practice with established engineering controls, ensuring that scale-up does not introduce unforeseen safety hazards. From an environmental standpoint, the generation of waste is minimized through the high atom economy of the condensation steps and the recyclability of solvents, aligning with increasingly stringent global environmental regulations. The absence of heavy metal contaminants in the final product, ensured by the filtration of the Pd/C catalyst, simplifies the regulatory filing process and reduces the burden of extensive heavy metal testing, accelerating time-to-market for new formulations.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding the implementation of this technology, we have compiled a set of answers based on the specific data points and advantages outlined in the patent documentation. These responses are designed to clarify the operational realities of the synthesis and its implications for quality assurance and regulatory compliance. Understanding these nuances is essential for stakeholders evaluating the transition from extraction-based sourcing to synthetic manufacturing.
Q: What are the primary advantages of this synthetic route over natural extraction?
A: Unlike natural extraction from olive leaves which suffers from seasonal variability and low yields, this chemical synthesis offers consistent batch-to-batch reproducibility, higher overall yields of 50-60%, and independence from agricultural supply chains.
Q: How does the choice of starting material impact production costs?
A: By utilizing 3,4-dihydroxybenzaldehyde instead of the more expensive 3,4-dihydroxyphenylacetic acid or tyrosol, the process significantly lowers raw material input costs while maintaining a streamlined reaction pathway suitable for industrial scaling.
Q: Is the catalytic deprotection step scalable for commercial manufacturing?
A: Yes, the final step utilizes palladium on carbon (Pd/C) catalytic hydrogenation, a standard and highly scalable unit operation in the fine chemical industry that ensures clean removal of protecting groups without generating excessive hazardous waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxytyrosol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route detailed in patent CN101891595A for securing a sustainable future for hydroxytyrosol supply. As a premier CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with the necessary hydrogenation reactors and purification systems to execute this benzyl-protection strategy with precision, adhering to stringent purity specifications and rigorous QC labs protocols that exceed international pharmacopoeia standards. We are committed to delivering high-purity hydroxytyrosol that meets the exacting requirements of the global pharmaceutical and nutraceutical industries, leveraging our technical expertise to optimize yield and minimize impurities at every stage of the manufacturing process.
We invite forward-thinking procurement leaders and R&D directors to collaborate with us to leverage this advanced synthesis technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data from our recent batches and comprehensive route feasibility assessments. Let us help you secure a reliable, cost-effective, and high-quality supply of hydroxytyrosol that empowers your product development and strengthens your market position.
