Advanced One-Pot Synthesis of Acetyl Syringate-2-Chloroethanol Ester for Commercial Scale
Advanced One-Pot Synthesis of Acetyl Syringate-2-Chloroethanol Ester for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for complex intermediates that balance efficiency with purity. A significant breakthrough in this domain is detailed in patent CN107573240B, which outlines a novel method for synthesizing acetyl syringate-2-chloroethanol ester. This compound, a derivative of syringic acid, holds immense potential as a versatile building block for various bioactive molecules and functional materials. The structural complexity of the target molecule, featuring both methoxy groups and a reactive chloroethyl ester moiety, traditionally poses challenges in selective functionalization. However, the disclosed technology offers a streamlined solution that addresses these synthetic hurdles effectively.
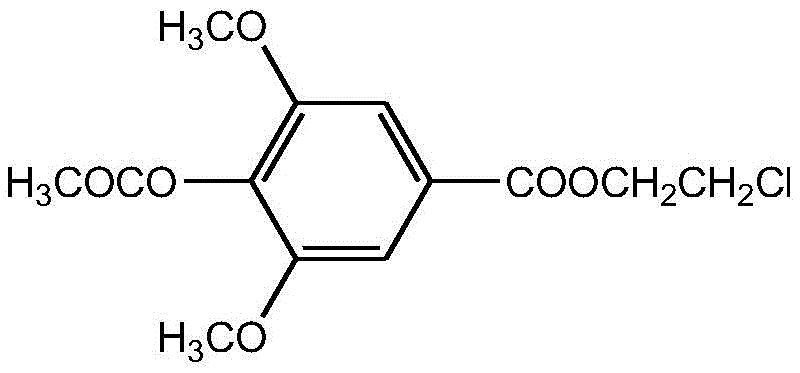
This innovative approach leverages a one-pot strategy that combines acetylation and esterification steps seamlessly. By avoiding the isolation of unstable or difficult-to-handle intermediates, the process not only simplifies the operational workflow but also enhances the overall material throughput. For R&D directors and process chemists, this represents a shift towards more atom-economical and time-efficient methodologies. The ability to generate high-purity products directly from crude reaction mixtures reduces the burden on downstream purification units, a critical factor when considering the economic viability of producing high-value pharmaceutical intermediates on a commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for functionalized benzoic acid derivatives often suffer from inefficiencies related to multi-step protection and deprotection strategies. In conventional scenarios, synthesizing a molecule with both an acetylated phenol and a chloroethyl ester would typically require separate reaction vessels and distinct purification stages for each functional group transformation. This fragmentation leads to increased solvent consumption, higher energy costs due to repeated heating and cooling cycles, and significant loss of material during transfer and isolation steps. Furthermore, the use of specialized reagents for selective esterification can drive up raw material costs and introduce hazardous waste streams that complicate environmental compliance.
Another critical drawback of older methodologies is the sensitivity of reaction conditions. Many classical esterification protocols require harsh acidic conditions or elevated temperatures that can degrade sensitive functional groups, such as the methoxy substituents found on the syringic acid backbone. This degradation results in the formation of complex impurity profiles that are difficult to remove, ultimately compromising the quality of the final active pharmaceutical ingredient (API) precursor. For procurement managers, these inefficiencies translate into volatile supply chains and unpredictable pricing, as yield fluctuations in early synthesis stages cascade through the entire production schedule, causing delays and resource wastage.
The Novel Approach
The methodology described in the patent data introduces a paradigm shift by utilizing a tandem reaction system where syringic acid, 1,2-dichloroethane, acetic anhydride, and triethylamine are combined in a single reactor. This one-pot design allows for the simultaneous acetylation of the phenolic hydroxyl group and the esterification of the carboxylic acid moiety without the need to isolate the intermediate species. The reaction conditions are remarkably mild, operating effectively within a temperature range of 40-75°C, which minimizes thermal stress on the molecular framework. This gentle approach preserves the integrity of the aromatic ring and prevents the decomposition of the chloroethyl chain, ensuring a cleaner reaction profile.
From a process engineering perspective, the elimination of intermediate separation steps drastically reduces the cycle time and labor intensity associated with the manufacturing process. The use of triethylamine serves a dual purpose as both a catalyst promoter and an acid scavenger, neutralizing the hydrochloric acid byproduct generated during the substitution reaction. This internal neutralization simplifies the workup procedure, allowing for straightforward washing and recrystallization to achieve purities exceeding 97%. For supply chain heads, this reliability means consistent batch-to-batch quality and the ability to scale production volumes without the bottleneck of complex purification infrastructure, thereby securing a steady flow of critical intermediates for downstream drug synthesis.
Mechanistic Insights into One-Pot Dual Functionalization
The core of this synthetic innovation lies in the synergistic interaction between the reactants under basic conditions. The reaction initiates with the activation of syringic acid by triethylamine, forming a carboxylate salt that is highly nucleophilic. Simultaneously, acetic anhydride reacts with the phenolic hydroxyl group to form the acetyl ester. The presence of 1,2-dichloroethane is crucial; it acts not merely as a solvent but as the source of the chloroethyl group. The carboxylate anion attacks one of the carbon atoms in the 1,2-dichloroethane molecule, displacing a chloride ion in an SN2-type substitution mechanism. This concerted pathway ensures that both functionalizations occur in a coordinated manner, driven by the thermodynamic stability of the resulting ester bonds.
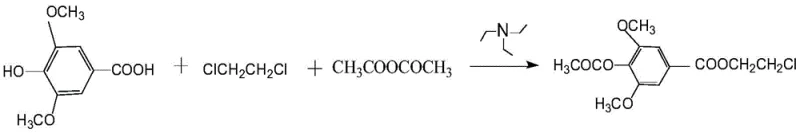
Controlling impurities in such a multi-component system is paramount for pharmaceutical applications. The patent specifies a rigorous purification protocol involving sequential washing with water, dilute sodium hydroxide, and dilute hydrochloric acid. This sequence is designed to remove unreacted syringic acid, excess acetic anhydride, and triethylamine salts effectively. The alkaline wash removes acidic impurities, while the acid wash neutralizes any residual amine base. Finally, recrystallization from petroleum ether exploits the solubility differences between the target ester and any remaining organic byproducts. This meticulous workup ensures that the final product meets stringent purity specifications, typically ranging from 97.3% to 98.5%, which is essential for preventing toxicological issues in subsequent drug development stages.
How to Synthesize Acetyl Syringate-2-Chloroethanol Ester Efficiently
Implementing this synthesis route requires precise control over stoichiometry and thermal parameters to maximize yield and minimize side reactions. The process begins by establishing an inert nitrogen atmosphere to prevent oxidation of the sensitive phenolic components. The reactants are mixed in specific mass-to-volume ratios, such as 3.6-4.2 g of syringic acid to 25-35 ml of 1,2-dichloroethane, ensuring optimal concentration for the nucleophilic attack. The addition of triethylamine is timed carefully after the initial heating phase to control the exotherm and maintain the reaction trajectory. Detailed standardized operating procedures for this synthesis are outlined below to guide technical teams in replicating these high-efficiency results.
- Mix syringic acid, 1,2-dichloroethane, and acetic anhydride under nitrogen protection and heat to 40-75°C with stirring.
- Add triethylamine to the mixture, maintain temperature for 6-15 hours, then raise temperature to reflux for another 6-15 hours.
- Purify the reflux liquid by washing with water, NaOH, and HCl solutions, drying with anhydrous sodium sulfate, and recrystallizing from petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations managing the procurement of specialty chemicals, the transition to this one-pot synthesis method offers substantial strategic benefits. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures. By consolidating two distinct chemical transformations into a single vessel, facilities can lower their utility consumption, specifically regarding heating and agitation energy. Furthermore, the reduction in unit operations means less equipment occupancy time, allowing for higher throughput within existing infrastructure. This efficiency gain is critical for maintaining competitive pricing in the global market for pharmaceutical intermediates, where margin pressures are increasingly intense.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps removes the need for extensive drying and filtration equipment, leading to significant capital expenditure savings. Additionally, the high atom economy of using 1,2-dichloroethane as both reactant and solvent reduces the volume of raw materials required per kilogram of product. The simplified purification process also lowers the consumption of auxiliary solvents and reagents used in workup, contributing to a leaner cost structure. These cumulative savings allow suppliers to offer more competitive pricing without compromising on quality standards, providing a clear financial advantage for long-term supply contracts.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent production schedules, mitigating the risk of delays caused by complex purification bottlenecks. Since the reagents involved, such as acetic anhydride and triethylamine, are commodity chemicals with stable global availability, the supply chain is less vulnerable to raw material shortages. The mild reaction conditions also reduce the likelihood of equipment failure or safety incidents, further stabilizing the production timeline. For supply chain heads, this predictability translates into improved inventory management and the ability to meet Just-In-Time delivery requirements for downstream pharmaceutical manufacturers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is straightforward due to the absence of hazardous high-pressure or high-temperature steps. The温和 nature of the reaction minimizes the generation of hazardous waste, aligning with modern green chemistry principles and regulatory expectations. The efficient use of solvents and the ability to recover and recycle 1,2-dichloroethane further reduce the environmental footprint of the manufacturing process. This compliance with environmental standards not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational corporations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its adoption for commercial production. The following questions address common concerns regarding yield optimization, purity control, and scalability. These answers are derived directly from the experimental data and process descriptions provided in the patent literature, ensuring that the information is grounded in verified scientific results. This transparency helps build confidence among technical buyers and quality assurance teams who are responsible for validating new supply sources.
Q: What are the key advantages of the one-pot synthesis method for this ester?
A: The one-pot method eliminates the need for intermediate isolation, significantly reducing energy consumption and operational complexity while maintaining high purity levels above 97%.
Q: How does this process ensure high yield compared to traditional methods?
A: By utilizing 1,2-dichloroethane as both a reactant and solvent medium alongside acetic anhydride, the reaction drives dual functionalization efficiently, achieving yields consistently around 84-86%.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the mild reaction conditions (40-75°C) and simple workup procedure involving standard washing and recrystallization make it highly suitable for large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acetyl Syringate-2-Chloroethanol Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development pipelines. Our team of expert chemists has extensively analyzed advanced synthetic routes like the one described in CN107573240B to ensure we can deliver products that meet the most rigorous industry standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of acetyl syringate-2-chloroethanol ester performs reliably in your downstream processes.
We invite you to collaborate with us to optimize your supply chain for this valuable intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. By partnering with us, you gain access to deep technical expertise and a commitment to quality that few competitors can match. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term business goals.
