Advanced Green Synthesis of Beclomethasone Dipropionate Intermediates for Commercial Scale-up
Advanced Green Synthesis of Beclomethasone Dipropionate Intermediates for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with environmental sustainability, particularly for potent glucocorticoids like Beclomethasone dipropionate. Patent CN111944002A introduces a transformative preparation method that redefines the efficiency of producing this critical anti-inflammatory agent. By utilizing compound DB11 as a strategic starting material, the disclosed technology orchestrates a seamless sequence of macrocyclization and ring-opening reactions within a unified system. This approach effectively bypasses the cumbersome isolation steps characteristic of legacy processes, thereby minimizing solvent consumption and waste generation. The structural integrity of the final molecule, characterized by its specific chlorination and esterification patterns, is meticulously preserved through controlled reaction conditions. As a reliable API intermediate supplier, understanding such technological leaps is vital for maintaining competitive advantage in the global market. The following analysis dissects the chemical ingenuity and commercial viability of this patented route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Beclomethasone dipropionate has been plagued by operational inefficiencies that inflate both cost and environmental footprint. The traditional route typically initiates with DB11 but proceeds through a fragmented series of discrete unit operations. After the initial chlorination at the 9th position to form the first intermediate, the process mandates a complete drying step before proceeding to macrocyclization. This intermediate is then subjected to ring-opening in an ethanol-aluminum trichloride system, followed by yet another drying phase prior to propionylation in DMF. Such a disjointed workflow not only extends the production timeline significantly but also necessitates the handling and disposal of multiple solvent systems, leading to substantial wastewater volumes. Furthermore, the cumulative yield of this conventional pathway often hovers around 105%, indicating significant material loss during the repeated isolation and purification stages. These inherent bottlenecks render the traditional method less attractive for modern, high-volume manufacturing where lean principles are paramount.
The Novel Approach
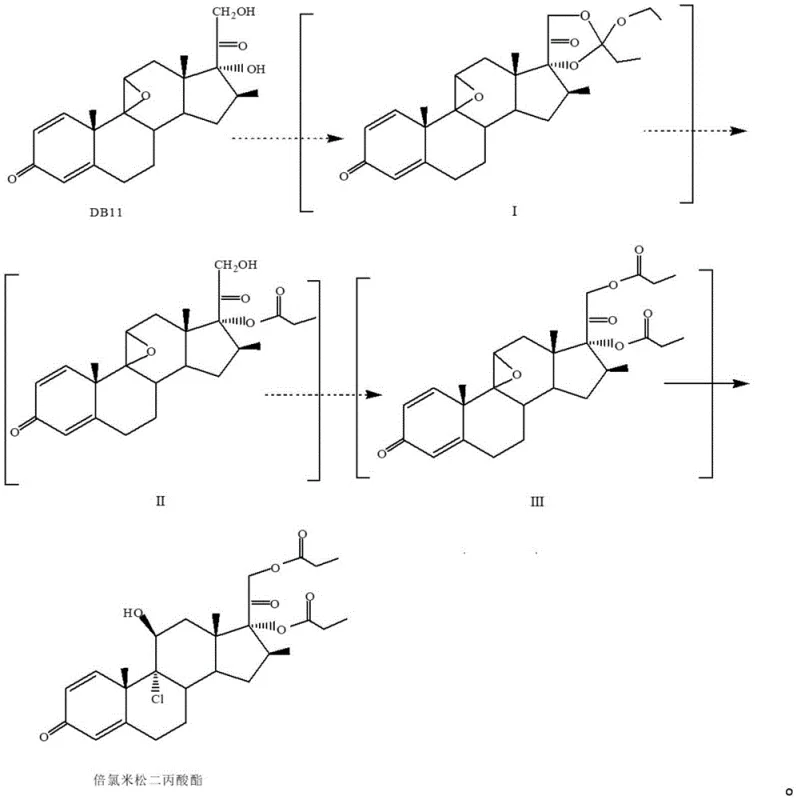
In stark contrast, the methodology outlined in CN111944002A presents a streamlined architecture that consolidates critical transformation steps. The innovation lies in the strategic adjustment of the structural modification sequence, allowing macrocyclization and ring-opening to occur in a coordinated manner without intermediate isolation. By reacting DB11 with orthopropionate and p-toluenesulfonic acid, the system generates a protected intermediate which is subsequently treated with aluminum trichloride solution directly. This telescoping of reactions eliminates the energy-intensive drying phases between steps. The process further optimizes solvent usage by concentrating the initial organic solvent and introducing a chloroalkane for dissolution before separation. This cohesive strategy not only accelerates the production period but also markedly enhances the overall yield, with reported figures exceeding 115%. The ability to achieve such efficiency while maintaining a clean production profile underscores the superiority of this novel approach over established practices.
Mechanistic Insights into Orthoester Protection and Lewis Acid Catalysis
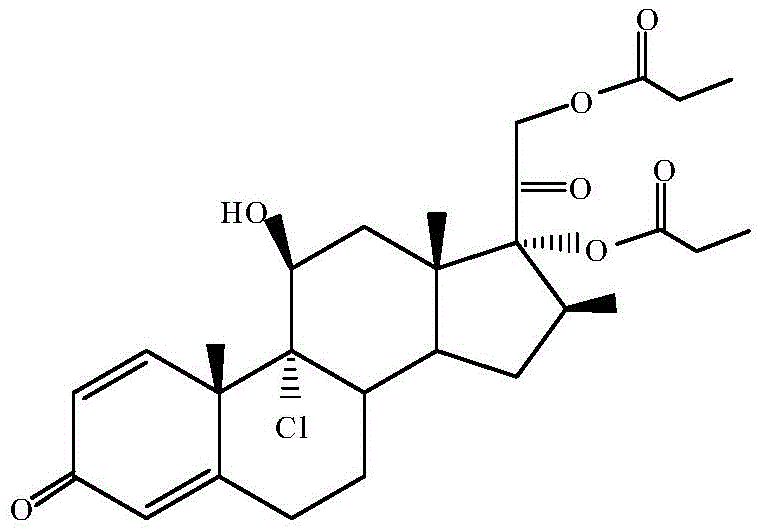
The chemical elegance of this synthesis relies heavily on the precise manipulation of protecting groups and Lewis acid catalysis to direct regioselectivity. The initial reaction with orthopropionate serves to protect the 17,21-dihydroxy functionality, forming a cyclic orthoester intermediate (Formula I). This protection is crucial as it prevents unwanted side reactions at these hydroxyl sites during the subsequent harsh chlorination conditions. The introduction of aluminum trichloride acts as a potent Lewis acid, facilitating the ring-opening of the epoxide at the 9,11-position while simultaneously introducing the chlorine atom at the 9-alpha position. The mechanistic pathway ensures that the stereochemistry is strictly controlled, yielding the desired 9-chloro-11-beta-hydroxy configuration essential for biological activity. Following this, the propionylation step utilizes catalysts such as DMAP or triethylamine to selectively esterify the remaining hydroxyl groups. The final acidic treatment with hydrochloric acid cleaves the orthoester protecting group, regenerating the 17,21-dihydroxy moieties and completing the synthesis of the target dipropionate. This sequence demonstrates a sophisticated understanding of steroid chemistry, ensuring that each functional group is modified at the optimal stage to minimize impurity formation.
Impurity control is a cornerstone of this patented process, directly addressing the rigorous demands of regulatory bodies for pharmaceutical intermediates. The high purity levels, reported at greater than 99.5% by HPLC, are achieved through the minimization of side reactions enabled by the mild conditions of the orthoester formation and the specificity of the Lewis acid catalysis. By avoiding the thermal stress associated with repeated drying and solvent switching, the degradation of the sensitive steroid backbone is significantly mitigated. The process parameters, such as maintaining temperatures between 0°C and 25°C during key steps, further suppress the formation of by-products. Additionally, the recrystallization step using ethyl acetate and activated carbon serves as a final polishing operation, removing trace colored impurities and residual solvents. This robust control over the impurity profile, with any single impurity kept below 0.10%, ensures that the resulting API intermediate is suitable for direct use in final drug formulation without extensive additional purification.
How to Synthesize Beclomethasone Dipropionate Efficiently
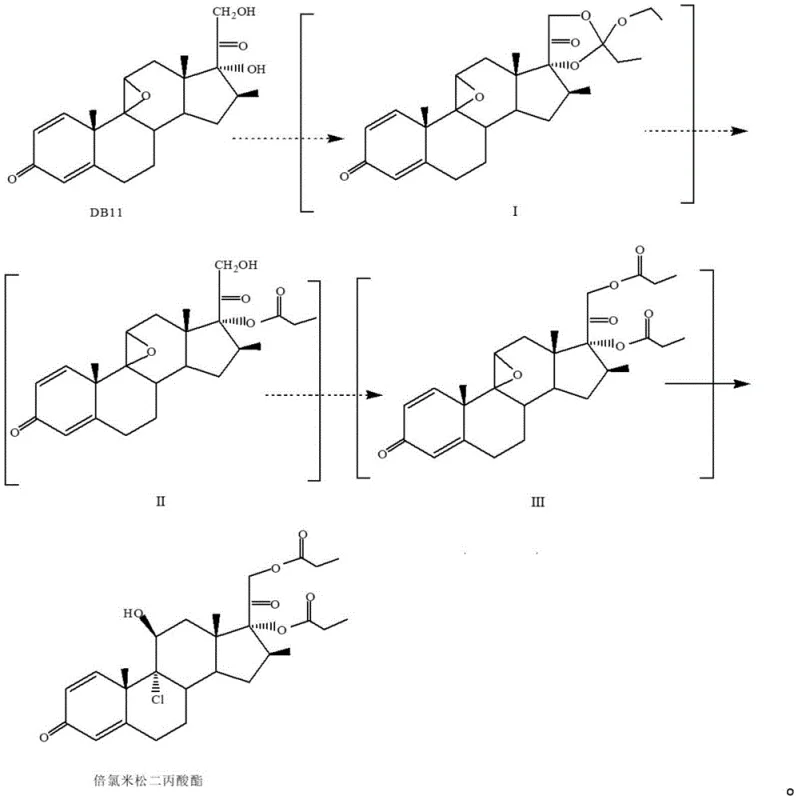
The practical implementation of this synthesis route requires careful attention to solvent selection and reaction monitoring to replicate the high yields described in the patent. The process begins with the dissolution of DB11 in a solvent like tetrahydrofuran or DMF, followed by the addition of triethyl orthopropionate and a catalytic amount of p-toluenesulfonic acid. Once the formation of Intermediate I is confirmed via TLC, the system is cooled, and an aqueous aluminum trichloride solution is introduced dropwise to effect the ring-opening chlorination. After concentration and solvent exchange to a chloroalkane, the organic layer is separated and subjected to propionylation using propionic anhydride or propionyl chloride in the presence of a base catalyst. The final deprotection is achieved by treating the propionylated intermediate with hydrochloric acid at low temperatures. For a comprehensive breakdown of the specific operational parameters and safety considerations, the detailed standardized synthesis steps are provided in the guide below.
- React compound DB11 with orthopropionate and p-toluenesulfonic acid in a first organic solvent to form Intermediate I.
- Add aluminum trichloride solution to Intermediate I for ring-opening chlorination, followed by solvent exchange to obtain Intermediate II.
- Propionylate Intermediate II using a catalyst and propionylation reagent to generate Intermediate III.
- Treat Intermediate III with hydrochloric acid for final deprotection and ring opening to yield crude Beclomethasone dipropionate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible benefits that extend beyond mere chemical yield. The consolidation of reaction steps translates directly into a reduction of operational overhead, as fewer unit operations mean less equipment occupancy time and lower labor costs. The elimination of intermediate drying steps is particularly significant, as it removes a major bottleneck in batch processing and reduces the energy load on facility utilities. Furthermore, the reduction in wastewater generation aligns with increasingly strict environmental regulations, potentially lowering waste disposal fees and simplifying compliance reporting. These factors collectively contribute to a more resilient and cost-efficient supply chain for high-purity API intermediates.
- Cost Reduction in Manufacturing: The telescoped nature of the reaction sequence drastically simplifies the manufacturing workflow by removing the need for isolating and drying intermediates. This reduction in processing steps leads to substantial cost savings in terms of energy consumption, solvent usage, and labor hours. By avoiding the purchase and handling of multiple distinct solvent systems required in traditional routes, the overall material cost is significantly optimized. Additionally, the improved yield means that less raw material is required to produce the same amount of final product, further enhancing the economic viability of the process.
- Enhanced Supply Chain Reliability: The use of readily available starting materials like DB11 and common industrial solvents ensures a stable supply base that is not subject to the volatility of exotic reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent choices, provides flexibility in sourcing and reduces the risk of production stoppages due to material shortages. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor configurations and avoiding hazardous reagents that would complicate scale-up. The significant reduction in wastewater volume simplifies effluent treatment requirements, making it easier to comply with environmental standards across different geographic regions. This environmental friendliness not only reduces regulatory risk but also enhances the corporate sustainability profile of the manufacturing entity, a key consideration for modern supply chain partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does the new synthesis route improve upon traditional methods for Beclomethasone dipropionate?
A: The novel route telescopes the macrocyclization and ring-opening reactions into a single system, eliminating intermediate isolation and drying steps. This significantly reduces wastewater generation and shortens the production cycle compared to the traditional multi-solvent approach.
Q: What is the achieved purity level of the final product using this method?
A: Analytical data indicates that the refined product achieves an HPLC content of greater than 99.5%, with any single impurity controlled to less than 0.10%, meeting stringent pharmaceutical standards.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common industrial solvents like tetrahydrofuran and dichloromethane and avoids complex transition metal catalysts, making it highly scalable and cost-effective for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beclomethasone Dipropionate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern pharmaceutical landscape. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN111944002A can be successfully translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Beclomethasone dipropionate intermediate adheres to the highest quality standards required for global markets.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your operation. We encourage you to contact us for specific COA data and route feasibility assessments to ensure that this technology meets your precise manufacturing requirements and drives value for your organization.
