Advanced Two-Step Synthesis of Lithium Fluorosulfonyl Difluorophosphorimide for High-Performance Battery Electrolytes
The landscape of lithium-ion battery technology is constantly evolving, driven by the relentless demand for higher energy density and longer cycle life. A pivotal breakthrough in this domain is detailed in patent CN109422252B, which introduces a novel preparation method for lithium fluorosulfonyl difluorophosphorimide, a critical component for next-generation electrolytes. This innovation addresses long-standing challenges in the synthesis of complex lithium salts, specifically targeting the issues of low yield, complex purification, and the use of hazardous reagents found in traditional methods. By streamlining the synthetic pathway into a concise two-step process, this technology not only enhances the chemical integrity of the final product but also offers a robust framework for industrial scalability. For R&D directors and procurement specialists alike, understanding this shift from legacy chemistry to this optimized route is essential for securing a competitive edge in the rapidly expanding energy storage market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated phosphorus-sulfur imide salts has been plagued by inefficiencies that hinder large-scale commercial adoption. Traditional routes, such as those disclosed in earlier patents like CN102617414B, often rely on the use of phosphorus oxychloride (POCl3), a reagent known for its high reactivity and significant safety hazards. These conventional methodologies typically involve multiple reaction steps, each introducing potential points of failure and yield loss. The cumulative effect of these multi-step processes results in a lower overall yield, often struggling to reach economic viability for mass production. Furthermore, the intermediates generated in these older pathways are frequently difficult to purify, requiring extensive and costly downstream processing to remove trace impurities that can degrade battery performance. The introduction of moisture during these complex sequences is another critical vulnerability, as water can hydrolyze sensitive intermediates, leading to product degradation and inconsistent quality.
The Novel Approach
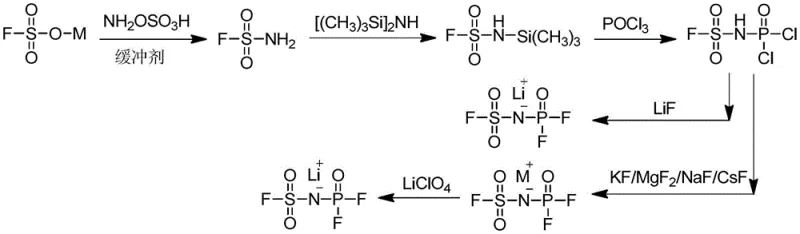
In stark contrast, the methodology outlined in CN109422252B represents a paradigm shift towards efficiency and safety. This novel approach cleverly bypasses the need for aggressive chlorinating agents like POCl3 by utilizing hexamethyldisiloxane and sulfamoyl chloride as primary building blocks. The entire synthesis is condensed into merely two distinct reaction stages, drastically reducing the operational complexity and the time required for production. By avoiding the introduction of water throughout the entire process, the method effectively eliminates a major source of side reactions and product decomposition. This streamlined workflow allows for simple purification of intermediates, which directly translates to a substantial increase in the final product yield, reported to exceed 85%. The ability to achieve such high yields with fewer steps not only lowers the cost of goods sold but also minimizes waste generation, aligning perfectly with modern green chemistry principles and environmental compliance standards.
Mechanistic Insights into the Two-Step Fluorination Process
The core of this technological advancement lies in its elegant reaction mechanism, which leverages the nucleophilic properties of silylated intermediates to construct the P-N-S backbone efficiently. In the first stage, sulfamoyl chloride reacts with hexamethyldisiloxane in an aprotic polar solvent under reflux conditions. This initial step generates a reactive silylated species that is primed for subsequent phosphorylation. The addition of an alkali metal hexafluorophosphate then facilitates the formation of the sulfonyl difluorophosphoryl imine alkali metal salt. This transformation is critical as it establishes the fundamental structural motif required for the final lithium salt without generating excessive halogenated byproducts. The reaction conditions are meticulously controlled, typically maintaining temperatures between 50°C and 100°C, ensuring that the reaction proceeds smoothly without thermal degradation of the sensitive fluorine-containing groups.
The second stage involves a strategic fluorination step where the alkali metal salt intermediate is treated with a fluorine-containing lithium salt, such as lithium fluoride (LiF). This metathesis reaction effectively swaps the alkali metal cation for lithium, finalizing the structure of the lithium fluorosulfonyl difluorophosphorimide. The choice of LiF is particularly advantageous due to its stability and cost-effectiveness compared to other fluorinating agents. The reaction proceeds under inert atmosphere protection, typically using nitrogen or argon, to prevent any atmospheric moisture from compromising the product integrity. Following the reaction, the separation process is remarkably straightforward; insoluble matter is removed by filtration, and the solvent is evaporated. The crude product can then be recrystallized from ethyl acetate to achieve the exceptional purity levels of over 99.9% cited in the patent data.
How to Synthesize Lithium Fluorosulfonyl Difluorophosphorimide Efficiently
Implementing this synthesis route requires precise control over reaction parameters and solvent selection to maximize yield and purity. The process begins with the careful selection of aprotic polar solvents such as acetonitrile or dimethyl carbonate, which provide the necessary solubility for the reactants while remaining inert under the reaction conditions. Operators must ensure that the molar ratios of sulfamoyl chloride to hexamethyldisiloxane are maintained within the optimal range of 1:(3-4) to drive the silylation to completion. The subsequent addition of the hexafluorophosphate salt must be performed dropwise to manage the exotherm and ensure uniform mixing. Detailed standard operating procedures regarding temperature ramping, reflux times, and vacuum drying protocols are essential for reproducibility. For a comprehensive guide on the specific equipment setup and step-by-step execution, please refer to the standardized synthesis protocol below.
- React sulfamoyl chloride with hexamethyldisiloxane in an aprotic polar solvent under reflux to generate the silylated intermediate.
- Add alkali metal hexafluorophosphate to the reaction mixture and continue refluxing to form the alkali metal sulfonyl difluorophosphoryl imine salt.
- Dissolve the intermediate salt and react with a fluorine-containing lithium salt (such as LiF) to finalize the lithium fluorosulfonyl difluorophosphorimide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers compelling strategic advantages that extend beyond mere technical specifications. The primary value proposition lies in the significant simplification of the manufacturing process, which directly correlates to reduced operational expenditures and enhanced supply reliability. By eliminating the need for hazardous and tightly regulated reagents like POCl3, facilities can reduce their safety compliance burdens and insurance costs. Furthermore, the reduction in reaction steps means that production throughput can be increased without necessitating additional reactor volume, effectively debottlenecking existing production lines. This efficiency gain allows suppliers to respond more agilely to fluctuating market demands for battery electrolyte additives, ensuring a steady flow of materials to cell manufacturers.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the substitution of expensive and difficult-to-handle reagents with more commodity-grade chemicals. The elimination of complex purification stages for intermediates reduces the consumption of solvents and energy, leading to substantial cost savings in utility and waste disposal. Additionally, the high yield of over 85% means that less raw material is wasted per kilogram of final product, optimizing the atom economy of the process. These factors combine to create a much lower cost base for the final lithium salt, allowing for more competitive pricing strategies in the electronic chemical manufacturing sector without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials such as sulfamoyl chloride and hexamethyldisiloxane, which are less prone to geopolitical supply shocks compared to specialized fluorinating agents. The robustness of the reaction against minor variations in conditions ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification products. Moreover, the simplified workflow reduces the dependency on highly specialized labor for complex multi-step operations, making it easier to scale up production capacity across different manufacturing sites. This reliability is crucial for maintaining the continuity of battery production lines which operate on tight just-in-time schedules.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns well with increasingly stringent global regulations regarding chemical manufacturing. The avoidance of chlorinated byproducts and the reduction in solvent usage significantly lower the environmental footprint of the production facility. The mild reaction conditions reduce the energy intensity of the process, contributing to lower carbon emissions per unit of product. Scalability is inherently supported by the simplicity of the two-step design, which can be easily translated from laboratory glassware to large-scale industrial reactors with minimal engineering modifications. This ease of scale-up ensures that suppliers can rapidly expand capacity to meet the surging demand for high-performance electric vehicle batteries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when adopting this methodology. Understanding these nuances is vital for making informed decisions about integrating this advanced lithium salt into your electrolyte formulations.
Q: What are the purity levels achievable with this new synthesis method?
A: The patented process described in CN109422252B achieves product purity exceeding 99.9%, significantly reducing impurity profiles compared to conventional multi-step routes.
Q: How does this method improve supply chain stability for electrolyte additives?
A: By replacing highly reactive and hazardous reagents like POCl3 with more stable alternatives and reducing the step count to just two major reactions, the method simplifies purification and enhances production feasibility.
Q: Does this lithium salt improve battery cycle life?
A: Yes, experimental data indicates that batteries utilizing this electrolyte additive demonstrate significantly improved capacity retention rates over 200 cycles compared to standard formulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lithium Fluorosulfonyl Difluorophosphorimide Supplier
As the global demand for high-performance energy storage solutions accelerates, the need for reliable sources of advanced electrolyte additives has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in organic fluorine chemistry to deliver superior battery materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric requirements of major cell manufacturers. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of lithium fluorosulfonyl difluorophosphorimide meets the exacting standards required for premium electric vehicle applications.
We invite you to explore how our optimized synthesis capabilities can enhance your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our technology can serve as a cornerstone for your next-generation battery projects.
