Advanced Synthesis of Soluble Polyimides for Next-Gen Microelectronics
The rapid evolution of the microelectronics and optoelectronics industries has created an insatiable demand for advanced polymer materials that can withstand rigorous processing conditions while maintaining superior electrical and optical properties. Traditional polyimides, such as the commercially ubiquitous Kapton, have long been valued for their thermal stability but are often hindered by poor solubility, deep coloration, and high dielectric constants, which limit their utility in next-generation flexible displays and high-frequency communication devices. Addressing these critical limitations, recent technological advancements disclosed in patent CN115448898A introduce a novel class of aromatic diamine monomers containing phenolphthalein Cardo structures alongside alkyl substituents. This innovation represents a significant leap forward for any reliable electronic chemical supplier aiming to support the fabrication of high-performance, soluble, and transparent polyimide films. By strategically integrating rigid yet twisted molecular architectures, this technology enables the production of polymers that balance thermal robustness with enhanced processability, directly addressing the pain points of R&D teams struggling with the trade-off between stability and solubility in high-end material design.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-performance polyimides has relied heavily on rigid, planar aromatic monomers that facilitate strong intermolecular interactions and high crystallinity. While this structural rigidity imparts exceptional thermal stability and mechanical strength, it inevitably leads to severe processing challenges, primarily due to the formation of intense Charge Transfer Complexes (CTC) between electron-donor diamine units and electron-acceptor dianhydride units. These CTC interactions result in polymers that are deeply colored, often ranging from yellow to dark brown, which renders them unsuitable for optical applications requiring high transparency. Furthermore, the strong chain packing and lack of free volume in conventional polyimides make them insoluble in common organic solvents and infusible, necessitating a two-step synthesis route involving the formation of a polyamic acid precursor followed by thermal imidization. This traditional approach not only complicates the manufacturing workflow but also introduces issues related to solvent retention and film defects, creating significant bottlenecks for cost reduction in display materials manufacturing where yield and uniformity are paramount.
The Novel Approach
The innovative methodology outlined in the patent data circumvents these traditional drawbacks by employing a meticulously designed diamine monomer, specifically 5,5'-diisopropyl-4,4'-diaminophenoxy-2,2'-dimethylphenolphthalein. This molecule incorporates a bulky phenolphthalein Cardo group, which acts as a steric hindrance to disrupt the parallel alignment of polymer chains, thereby effectively suppressing the formation of charge transfer complexes. Simultaneously, the introduction of isopropyl and methyl alkyl groups further increases the fractional free volume within the polymer matrix, enhancing chain mobility and solubility without sacrificing the inherent thermal stability provided by the aromatic backbone. This structural engineering allows for a direct one-step solution polycondensation process, eliminating the need for unstable polyamic acid intermediates and enabling the direct formation of high molecular weight polyimides. For procurement specialists, this translates to a streamlined production protocol that utilizes readily available starting materials like thymolphthalein, offering a pathway toward more efficient and scalable commercial scale-up of complex optoelectronic materials.
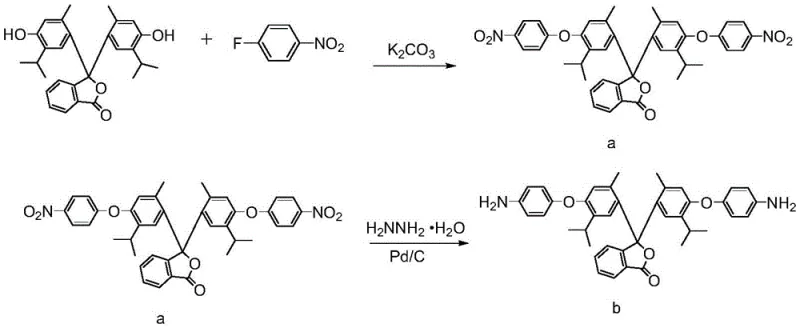
Mechanistic Insights into Aromatic Nucleophilic Substitution and Polycondensation
The synthesis of the key diamine monomer relies on a robust aromatic nucleophilic substitution reaction, where the phenolic hydroxyl groups of thymolphthalein act as nucleophiles attacking the electron-deficient aromatic ring of p-fluoronitrobenzene. This reaction is facilitated by a base catalyst, typically potassium carbonate, which deprotonates the phenolic hydroxyls to generate highly reactive phenoxide anions. The reaction proceeds under elevated temperatures, typically between 140°C and 160°C, ensuring complete conversion to the dinitro intermediate. The presence of the nitro group is crucial as it activates the fluorine atom for displacement, while the subsequent reduction step utilizes hydrazine hydrate in the presence of a palladium on carbon (Pd/C) catalyst. This catalytic hydrogenation selectively reduces the nitro groups to primary amines without affecting the sensitive ether linkages or the Cardo lactone ring, demonstrating high chemoselectivity. Understanding this mechanism is vital for R&D directors focused on high-purity polyimide precursors, as controlling the stoichiometry and reaction temperature is essential to minimize side reactions and ensure the structural integrity of the final monomer.
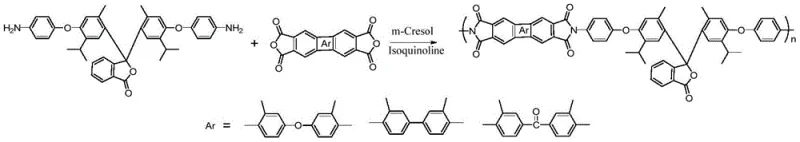
Following the monomer synthesis, the polymerization mechanism involves a direct polycondensation between the novel diamine and various aromatic dianhydrides, such as diphenyl ether tetra-acid dianhydride or benzophenone tetra-acid dianhydride. This reaction is catalyzed by isoquinoline in a high-boiling solvent like m-cresol or N-methylpyrrolidone. The isoquinoline catalyst plays a pivotal role in facilitating the dehydration and cyclization steps required to form the imide rings directly from the salt intermediate, bypassing the isolation of polyamic acid. The resulting polymer chains possess a unique combination of rigid aromatic segments and bulky Cardo structures, which prevents dense packing and maintains solubility even at high molecular weights. This mechanistic advantage ensures that the final polyimide films exhibit low dielectric constants and high optical transparency, making them ideal candidates for reducing lead time for high-purity aromatic diamines in the supply chain of advanced electronic components.
How to Synthesize 5,5'-Diisopropyl-4,4'-diaminophenoxy-2,2'-dimethylphenolphthalein Efficiently
The preparation of this specialized diamine monomer follows a precise two-stage protocol designed to maximize yield and purity while minimizing operational complexity. The process begins with the careful selection of high-purity thymolphthalein and p-fluoronitrobenzene, which are reacted under inert atmosphere to prevent oxidation of the phenolic species. Following the isolation of the dinitro intermediate, the reduction step requires strict control over the catalyst loading and reflux conditions to ensure complete conversion to the amine. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and temperature profiles optimized for industrial reproducibility, are provided in the guide below.
- Conduct aromatic nucleophilic substitution between thymolphthalein and p-fluoronitrobenzene using potassium carbonate catalyst at elevated temperatures.
- Isolate the intermediate dinitro compound through precipitation, filtration, and recrystallization to ensure high purity.
- Perform catalytic reduction of the dinitro intermediate using hydrazine hydrate and Pd/C catalyst to yield the final aromatic diamine monomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this novel monomer technology offers profound advantages that extend beyond mere technical performance, directly impacting the bottom line and supply chain resilience. The reliance on thymolphthalein, a commodity chemical widely used in pH indicators, as the primary starting material ensures a stable and cost-effective raw material base, mitigating the risks associated with sourcing exotic or proprietary intermediates. Furthermore, the ability to perform direct one-step polycondensation eliminates the energy-intensive thermal imidization cycles and the handling of hazardous polyamic acid solutions, leading to significant reductions in processing time and utility consumption. For supply chain heads, this simplification of the manufacturing workflow translates to enhanced throughput and reduced inventory holding times, fostering a more agile response to market demands.
- Cost Reduction in Manufacturing: The elimination of the two-step polyamic acid route significantly lowers operational expenditures by removing the need for separate imidization ovens and reducing solvent exchange steps. Additionally, the high solubility of the resulting polyimide allows for processing at lower solid contents or with cheaper solvent systems, further driving down the cost per kilogram of the final resin. The use of inexpensive catalysts like potassium carbonate and isoquinoline, rather than precious metal complexes for polymerization, ensures that the variable costs remain low and predictable, facilitating better budget forecasting for large-scale production runs.
- Enhanced Supply Chain Reliability: By utilizing a synthetic route based on widely available bulk chemicals, the risk of supply disruption is markedly decreased compared to processes relying on niche fluorinated monomers. The robustness of the synthesis, characterized by high yields and straightforward purification via precipitation and recrystallization, ensures consistent batch-to-bquality, which is critical for maintaining long-term contracts with electronics manufacturers. This reliability allows procurement managers to secure long-term supply agreements with confidence, knowing that the production capacity can be scaled without encountering bottlenecks related to raw material scarcity.
- Scalability and Environmental Compliance: The process is inherently scalable, as the reaction conditions (temperatures between 100°C and 200°C) are compatible with standard stainless steel reactors found in most fine chemical facilities. Moreover, the improved solubility of the polymer reduces the generation of insoluble waste and facilitates easier recycling of process solvents, aligning with increasingly stringent environmental regulations. The absence of volatile byproducts during the direct imidization step further simplifies废气 treatment requirements, making this technology a sustainable choice for modern chemical manufacturing facilities aiming to reduce their environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phenolphthalein-based polyimide technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the material's capabilities and limitations for potential adopters.
Q: How does the Cardo structure improve polyimide solubility?
A: The bulky, non-coplanar Cardo structure derived from phenolphthalein disrupts the regular packing of polymer chains, increasing free volume and reducing charge transfer complex (CTC) formation, which significantly enhances solubility and optical transparency.
Q: What are the thermal stability characteristics of these new polyimides?
A: Despite the introduction of solubilizing groups, the rigid aromatic backbone maintains high thermal stability, with glass transition temperatures typically exceeding 260°C and 10% weight loss temperatures above 400°C in nitrogen atmospheres.
Q: Can these monomers be used for colorless polyimide films?
A: Yes, the structural design specifically targets the reduction of intermolecular charge transfer interactions, resulting in films with high optical transparency and cutoff wavelengths suitable for optoelectronic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,5'-Diisopropyl-4,4'-diaminophenoxy-2,2'-dimethylphenolphthalein Supplier
As the global demand for high-performance electronic materials continues to surge, partnering with an experienced CDMO capable of navigating complex synthetic pathways is essential for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial application is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, meeting the exacting standards required for microelectronics and optoelectronic applications. We understand that consistency is key in the semiconductor supply chain, and our dedicated process engineering team is committed to optimizing every step of the synthesis to deliver superior quality monomers.
We invite you to collaborate with us to leverage this cutting-edge technology for your next-generation product lines. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and processing constraints. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical synthesis can accelerate your time-to-market while optimizing your overall production costs.
