Advanced Solvent-Based Synthesis of Nitro Phthalonitrile for Commercial Scale Production
Advanced Solvent-Based Synthesis of Nitro Phthalonitrile for Commercial Scale Production
The landscape of high-performance organic materials is constantly evolving, driven by the demand for superior purity and scalable manufacturing processes. A pivotal advancement in this domain is detailed in patent CN1132812C, which introduces a robust methodology for synthesizing 3- or 4-nitro phthalonitrile. These compounds serve as critical precursors for substituted phthalocyanines, which are indispensable in high-tech sectors ranging from laser printing photoconductors to optical recording media for VCD and DVD technologies. The traditional reliance on solid-phase baking methods has long plagued the industry with issues regarding harsh reaction conditions and difficult post-processing. This new technical disclosure offers a transformative solution by adopting a solvent-based approach that not only mitigates these operational risks but also significantly enhances the final product quality. For R&D directors and procurement specialists alike, understanding this shift is crucial for securing a reliable supply chain of high-purity electronic chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
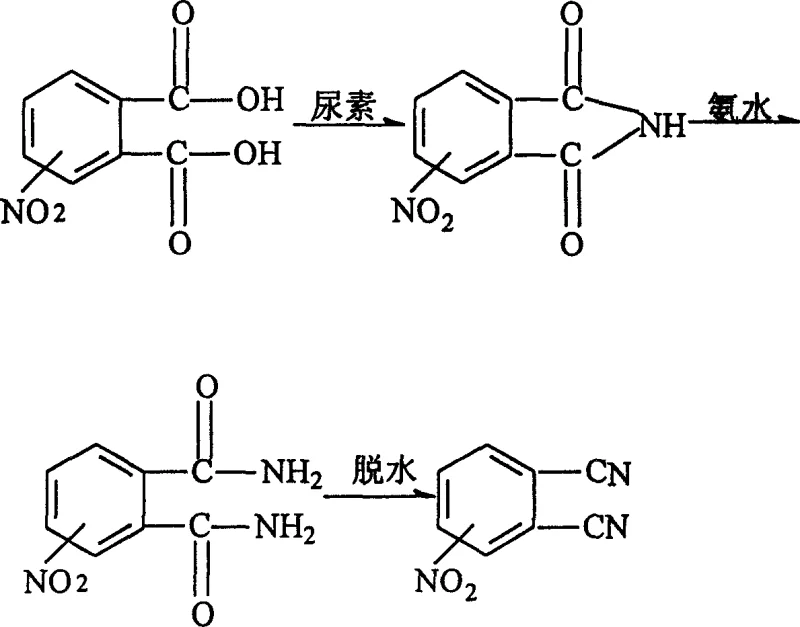
Historically, the synthesis of nitro-substituted phthalonitriles has been hindered by the severe constraints of solid-state chemistry. Prior art methods, such as those described in earlier literature, necessitated baking 3-nitrophthalic acid with ammonium salts at temperatures reaching up to 290°C. This extreme thermal requirement poses significant challenges for industrial suitability, as maintaining such high temperatures uniformly across large batches is energetically inefficient and technically demanding. Furthermore, the post-treatment of the reaction mass in a molten state is notoriously difficult, often leading to substantial product loss during isolation. The resulting crude material frequently exhibits a undesirable yellowish-brown coloration, indicative of thermal degradation and impurity formation, which directly compromises the quality of the target compound. Additionally, alternative liquid-phase methods relying on tetrahydrofuran (THF) introduce severe safety liabilities due to the solvent's low boiling point, high flammability, and toxicity, creating unacceptable risks for large-scale manufacturing environments.
The Novel Approach
In stark contrast to these legacy techniques, the innovative process outlined in the patent data leverages a sophisticated solvent strategy to overcome thermal and safety barriers. By utilizing high-boiling organic solvents such as o-dichlorobenzene or nitrobenzene, the reaction temperature is effectively moderated to a range of 160-250°C, which is far more conducive to controlled industrial operations. A key breakthrough in this methodology is the exploitation of solubility differentials; the desired nitrophthalimide intermediate is soluble in the refluxing solvent but becomes virtually insoluble upon cooling. This physical property allows for the product to be isolated simply by filtration, effectively integrating a recrystallization step into the synthesis itself. Consequently, this approach not only simplifies the workflow but also ensures a final product purity of approximately 99.6%, eliminating the need for complex downstream purification. Moreover, the substitution of hazardous organic solvents with aqueous ammonia in the subsequent amidation step drastically reduces environmental pollution and operational danger.
Mechanistic Insights into Solvent-Mediated Cyclization and Dehydration
The core of this synthetic route lies in the precise manipulation of reaction equilibria through solvent selection. The initial cyclization involves the reaction of nitrophthalic acid with urea, where the molar ratio is carefully optimized between 1:1 and 1:10 to drive the formation of the imide ring. The choice of solvent is paramount; o-dichlorobenzene acts not merely as a heat transfer medium but as a selective crystallization agent. As the reaction proceeds at 200-220°C, the system remains homogeneous, ensuring efficient molecular collision and conversion. However, upon cooling to room temperature, the solubility of the nitrophthalimide drops precipitously, causing it to precipitate out of the solution while impurities and by-products remain dissolved. This mechanism inherently purifies the intermediate, preventing the carryover of colored contaminants that typically plague solid-phase reactions. The subsequent amidation with strong aqua ammonia proceeds under mild conditions (25-60°C), avoiding the harsh conditions that could lead to side reactions or decomposition of the sensitive nitro group.
The final transformation involves the dehydration of the diamide to the dinitrile, a step that requires rigorous control to prevent hydrolysis or polymerization. Utilizing thionyl chloride in a polar aprotic solvent like DMF at low temperatures (0-25°C) ensures a clean conversion. The mechanistic pathway here involves the activation of the amide carbonyls by thionyl chloride, followed by elimination to form the nitrile triple bonds. The low-temperature protocol is critical for maintaining the integrity of the nitro substituent and preventing the formation of chlorinated by-products. This multi-stage control over reaction parameters—from high-temperature cyclization to cryogenic dehydration—demonstrates a deep understanding of physical organic chemistry, allowing for the production of high-purity intermediates essential for advanced optoelectronic applications. For a reliable electronic chemical supplier, mastering these nuances is the difference between a commodity chemical and a high-value specialty material.
How to Synthesize 3- or 4-Nitro Phthalonitrile Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process is designed to be linear and scalable, moving from the initial condensation to the final dehydration without the need for intermediate chromatographic purification. Operators must focus on the thermal profiles and solvent ratios, as these are the primary drivers of yield and purity. The following guide outlines the critical operational phases derived from the technical disclosure, providing a framework for laboratory validation and pilot plant trials. Detailed standard operating procedures should be developed based on these foundational steps to maximize efficiency.
- React 3- or 4-nitrophthalic acid with urea in a high-boiling solvent like o-dichlorobenzene at 160-250°C to form the imide.
- Perform amidation by reacting the resulting nitrophthalimide with concentrated aqueous ammonia at 25-60°C.
- Execute dehydration of the diamide using thionyl chloride in DMF at low temperatures (0-25°C) to yield the final nitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from solid-phase baking to this solvent-based methodology represents a significant opportunity for cost reduction in display material manufacturing. The elimination of extreme temperature requirements reduces energy consumption and extends the lifespan of reactor vessels, leading to lower capital expenditure over time. Furthermore, the inherent purification achieved during the cooling and filtration steps removes the need for expensive and time-consuming recrystallization or column chromatography processes. This streamlining of the production workflow translates directly into faster turnaround times and reduced labor costs. By avoiding the use of volatile and hazardous solvents like THF, facilities can also lower their insurance premiums and compliance costs associated with handling dangerous goods, creating a more resilient and economically viable supply chain.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by integrating purification into the reaction workup, thereby eliminating separate refining stages that consume additional solvents and energy. The high yield of the cyclization step, reported at approximately 93%, minimizes raw material waste, ensuring that the expensive nitrophthalic acid starting material is utilized with maximum efficiency. Additionally, the use of recoverable high-boiling solvents allows for recycling loops that further decrease the variable cost per kilogram of the final product. These factors combined create a highly competitive cost structure for producing high-purity optoelectronic material intermediates.
- Enhanced Supply Chain Reliability: Adopting a method that avoids the logistical and safety challenges of handling large quantities of flammable THF significantly de-risks the manufacturing operation. The use of aqueous ammonia and stable high-boiling solvents simplifies storage requirements and reduces the likelihood of production stoppages due to safety incidents or regulatory inspections. This stability ensures a consistent flow of goods, which is critical for downstream manufacturers of laser printers and optical media who rely on just-in-time delivery models. The robustness of the process against minor fluctuations in reaction conditions also contributes to batch-to-batch consistency, a key metric for supply chain reliability.
- Scalability and Environmental Compliance: The moderate reaction temperatures and the absence of noxious gas evolution (common in solid-phase baking) make this process exceptionally easy to scale from pilot to commercial production. The waste stream is easier to manage, as the primary by-products are soluble in the reaction medium and can be separated cleanly, reducing the burden on wastewater treatment facilities. This alignment with green chemistry principles not only satisfies increasingly stringent environmental regulations but also enhances the corporate sustainability profile of the manufacturer. Such environmental compliance is becoming a decisive factor for multinational corporations when selecting partners for their specialty chemical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of nitro phthalonitriles, based on the specific advantages and mechanisms detailed in the patent literature. Understanding these aspects helps stakeholders evaluate the feasibility of adopting this technology for their specific application needs. The answers provided reflect the consensus on best practices for handling these intermediates safely and efficiently.
Q: How does the new solvent method improve product purity compared to solid-phase baking?
A: The solvent method utilizes the solubility difference where the product is insoluble in cold high-boiling solvents, effectively acting as an in-situ recrystallization step that yields purity up to 99.6%, whereas solid-phase baking often results in yellowish-brown impurities.
Q: What are the safety advantages of replacing THF with aqueous ammonia in the amidation step?
A: Replacing tetrahydrofuran (THF), which is highly flammable, toxic, and volatile, with aqueous ammonia significantly reduces operational hazards, eliminates the risk of solvent explosions, and simplifies waste treatment protocols.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates at moderate temperatures (160-250°C) compared to the harsh 290°C required for solid-phase baking, making it far more manageable for industrial reactors and reducing energy consumption and equipment stress.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Nitro Phthalonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis methods requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel solvent-based process can be fully realized at an industrial level. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 3- or 4-nitro phthalonitrile meets the exacting standards required for high-tech electronic applications. Our infrastructure is designed to handle complex chemistries safely, providing our clients with a secure and consistent source of critical intermediates.
We invite you to collaborate with us to optimize your supply chain for next-generation optical and electronic materials. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing processes can enhance your product quality while reducing overall procurement costs. Let us be your strategic partner in navigating the complexities of fine chemical sourcing.
