Scaling Green Electrochemical Synthesis for High-Purity Agrochemical Intermediates
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical intermediates, specifically focusing on sustainability and cost-efficiency. Patent CN115874202A introduces a groundbreaking methodology for the preparation of sulfonyl compounds through the electrolytic oxidation of sulfur-based compounds, with a specific embodiment detailing the synthesis of 4-methylsulfonyl-2,3-dimethylbromobenzene. This compound serves as a pivotal building block in the manufacturing of Topramezone, a widely used HPPD inhibitor herbicide. The traditional reliance on stoichiometric chemical oxidants is being challenged by this electrochemical approach, which leverages electricity as a clean reagent to drive the transformation. By integrating surfactant technology into the electrolytic cell, the process overcomes the inherent solubility challenges of hydrophobic organic substrates in aqueous media. 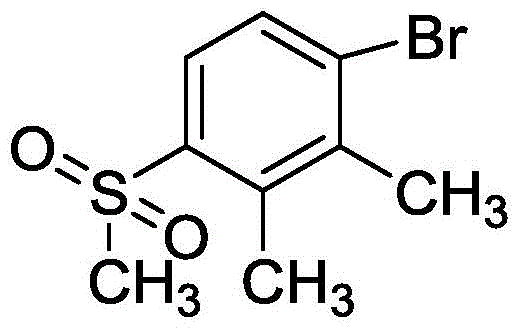 This structural advancement allows for a transition away from volatile organic solvents towards a greener, water-based system, marking a significant shift in how fine chemical manufacturers approach large-scale oxidation reactions.
This structural advancement allows for a transition away from volatile organic solvents towards a greener, water-based system, marking a significant shift in how fine chemical manufacturers approach large-scale oxidation reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of thioethers to sulfones has relied heavily on chemical oxidants such as hydrogen peroxide coupled with metal catalysts or sodium hypochlorite systems. While effective on a small scale, these methods present substantial hurdles for industrial application. The hydrogen peroxide pathway often suffers from low atom economy and requires the use of transition metal catalysts that are difficult to recover completely, leading to potential heavy metal contamination in the final product and significant environmental burden during wastewater treatment. Alternatively, the use of sodium hypochlorite, while inexpensive, introduces stability issues; the available chlorine content degrades rapidly upon storage, leading to inconsistent reaction outcomes. Furthermore, chemical oxidation with hypochlorite generates vast quantities of inorganic salt waste, creating a complex and costly downstream purification and disposal challenge that negatively impacts the overall process economics and environmental compliance status of the manufacturing facility.
The Novel Approach
The innovative process described in the patent data circumvents these traditional bottlenecks by utilizing an electrochemical cell where the oxidant is generated in-situ. Instead of adding external oxidizing agents, the system electrolytically oxidizes chloride ions present in the solution to generate active chlorine species directly at the anode surface. This "on-demand" generation ensures high utilization efficiency of the oxidizing equivalents and eliminates the storage and handling risks associated with bulk oxidants. Crucially, the addition of water-soluble surfactants, specifically quaternary ammonium salts, enables the reaction to proceed in a predominantly aqueous phase. This eliminates the need for large volumes of organic solvents and expensive supporting electrolytes like perchlorates, which are hazardous and costly. The result is a streamlined, continuous-flow compatible process that offers superior control over reaction selectivity and minimizes the formation of chlorinated by-products often seen in batch chemical oxidations.
Mechanistic Insights into Surfactant-Assisted Electrolytic Oxidation
The core mechanism of this transformation relies on the synergistic interaction between electrochemical parameters and colloidal chemistry. In the absence of a surfactant, the hydrophobic 4-methylthio-2,3-dimethylbromobenzene forms a separate oil phase, resulting in minimal contact with the aqueous electrolyte and consequently negligible conversion rates, as evidenced by comparative data showing yields dropping to near zero without emulsification. The introduction of cationic surfactants like dodecyltrimethylammonium chloride creates a micro-emulsion or micellar environment where the organic substrate is solubilized within the hydrophobic cores of the micelles dispersed in the water. This dramatically increases the interfacial area available for the electrochemical reaction. At the anode, typically composed of a dimensionally stable material like titanium coated with ruthenium and iridium oxides, chloride ions are oxidized to form active chlorine species such as hypochlorous acid or chlorine gas dissolved in the micro-environment. These reactive species then diffuse to the solubilized thioether, effecting a two-electron oxidation to the sulfone state with high specificity.
Controlling impurities in this electrochemical system is achieved through precise modulation of current density and temperature. High current densities can lead to the over-generation of active chlorine, which may attack the aromatic ring or the methyl groups, leading to chlorinated by-products. The patent data indicates that maintaining a current density around 200A/m² and a temperature near 20°C optimizes the balance between oxidation rate and selectivity. Furthermore, the choice of electrode material plays a vital role; plate-shaped RuO2/IrO2 composite electrodes demonstrate superior performance compared to mesh electrodes, likely due to reduced adsorption of intermediates on the electrode surface which prevents further unwanted side reactions. The surfactant itself also contributes to purity; cationic surfactants are preferred over anionic ones because they are less likely to migrate to the anode and undergo oxidative decomposition themselves, thereby maintaining the stability of the electrolytic medium throughout the prolonged reaction cycle required for complete conversion.
How to Synthesize 4-Methylsulfonyl-2,3-dimethylbromobenzene Efficiently
The synthesis protocol outlined in the patent provides a robust framework for reproducing this high-yielding transformation in a pilot or production setting. The procedure begins with the preparation of the electrolyte solution, where a specific concentration of chloride salt and a carefully selected quaternary ammonium surfactant are dissolved in water. The hydrophobic starting material is then introduced to this aqueous phase under vigorous stirring to ensure a homogeneous dispersion before the application of electrical current. The reaction is conducted in an undivided cell equipped with optimized electrode plates, maintaining strict control over the thermal profile to prevent the degradation of the active oxidizing species.
- Prepare the electrolytic system by dissolving a quaternary ammonium salt surfactant (e.g., dodecyltrimethylammonium chloride) and a chloride salt catalyst in water to form an aqueous phase.
- Disperse the hydrophobic starting material, 4-methylthio-2,3-dimethylbromobenzene, into the aqueous electrolyte solution under vigorous stirring to ensure effective emulsification.
- Perform electrolysis using a Ti-based RuO2/IrO2 composite anode and a stainless steel cathode at a controlled current density of approximately 200A/m² and temperature of 20°C until the desired conversion is achieved.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this surfactant-assisted electrochemical route offers compelling strategic advantages beyond mere technical feasibility. The most significant impact is observed in the reduction of raw material complexity and cost. By replacing expensive and hazardous supporting electrolytes like tetrabutylammonium hexafluorophosphate or perchlorates with common industrial salts like sodium chloride, the direct material cost is drastically lowered. Additionally, the elimination of transition metal catalysts removes the need for sophisticated and costly metal scavenging steps during downstream processing, which simplifies the purification workflow and reduces the consumption of silica gel or specialized resins. This simplification translates directly into lower operational expenditures and a reduced carbon footprint, aligning with the increasingly stringent ESG (Environmental, Social, and Governance) criteria demanded by global agrochemical conglomerates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of high-cost reagents with commodity chemicals. Traditional methods often require stoichiometric amounts of oxidants and expensive catalysts, whereas this electrochemical method uses electricity and cheap chloride salts as the primary drivers of the reaction. The removal of organic solvents from the main reaction phase further reduces costs associated with solvent recovery, distillation, and loss. Moreover, the avoidance of heavy metal catalysts eliminates the regulatory and financial burden associated with metal residue testing and removal, ensuring a cleaner product stream that requires less intensive purification, thereby enhancing the overall profit margin for the manufactured intermediate.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on readily available commodities like sodium chloride and water enhances resilience against market volatility. Unlike specialized oxidants or rare metal catalysts which may face supply disruptions or price spikes, the key inputs for this electrochemical process are abundant and globally sourced. The simplified reagent profile also reduces the logistical complexity of storing hazardous chemicals on-site, lowering insurance premiums and safety compliance costs. This stability ensures consistent production schedules and reliable delivery timelines for downstream customers, mitigating the risk of production stoppages due to raw material shortages.
- Scalability and Environmental Compliance: The aqueous nature of this process inherently supports easier scale-up compared to solvent-intensive organic syntheses. Water acts as an excellent heat sink, facilitating temperature control in large reactors and reducing the risk of thermal runaway. Environmentally, the process generates significantly less hazardous waste; the absence of heavy metals and the minimization of organic solvent usage simplify wastewater treatment protocols. The chloride ions can theoretically be recycled within the system, moving towards a closed-loop manufacturing model. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also serves as a powerful marketing asset when engaging with sustainability-focused clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrolytic oxidation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational parameters and benefits for potential adopters. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term value proposition of this synthetic route.
Q: Why is a surfactant critical in this electrochemical oxidation process?
A: The starting material, 4-methylthio-2,3-dimethylbromobenzene, is an oily substance insoluble in water. The surfactant acts as an emulsifier, dispersing the organic phase into the aqueous electrolyte, which drastically increases the interfacial area for reaction and prevents the need for large volumes of organic solvents.
Q: What are the advantages of using chloride salts over perchlorates in this system?
A: Chloride salts like sodium chloride are significantly cheaper and safer than perchlorates, which are expensive and pose safety hazards due to their explosive nature under heat or impact. Furthermore, chloride ions are electrochemically oxidized in-situ to generate the active oxidizing species, simplifying the reagent supply chain.
Q: How does this method improve environmental compliance compared to traditional oxidation?
A: Traditional methods often utilize hydrogen peroxide with metal catalysts, leading to heavy metal contamination in wastewater, or sodium hypochlorite, which generates large amounts of waste salt. This electrochemical method operates in a water-based system with recyclable chloride ions and avoids heavy metals, significantly reducing the environmental footprint and waste treatment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methylsulfonyl-2,3-dimethylbromobenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-value agrochemical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN115874202A are successfully translated into robust industrial operations. Our facilities are equipped with state-of-the-art electrochemical reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of 4-methylsulfonyl-2,3-dimethylbromobenzene meets the exacting standards required for herbicide synthesis. We are committed to delivering not just a product, but a reliable supply solution that integrates seamlessly into your manufacturing value chain.
We invite you to collaborate with us to explore how this green synthesis route can optimize your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating the tangible economic benefits of switching to this electrochemical method. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective source for your critical intermediates.
