Advanced Manufacturing of Halogen-Substituted Benzene Dimethanol for Global Pharma Supply Chains
The global demand for high-purity fluorinated and chlorinated aromatic building blocks continues to surge, driven by the relentless innovation in pharmaceutical and agrochemical sectors. A pivotal advancement in this domain is detailed in patent CN102341367B, which discloses a robust and highly selective method for producing halogen-substituted benzene dimethanol. This technology addresses long-standing challenges in the synthesis of polyhalogenated diols, which serve as critical precursors for bioactive molecules. Unlike traditional routes that often struggle with selectivity issues or require hazardous reagents, this novel approach leverages a strategic N-protection mechanism followed by mild reduction. For R&D directors and procurement specialists alike, understanding the nuances of this pathway is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering complex structures with consistent quality. The patent outlines a transformative two-step sequence that converts readily available halogen-substituted terephthalamides into valuable diols, offering a compelling alternative to legacy manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogen-substituted benzene dimethanol has relied heavily on the direct reduction of halogen-substituted terephthalic acids or their corresponding esters. As referenced in prior art such as JP-A-2007-224017, these conventional pathways typically necessitate the use of powerful reducing agents like lithium aluminum hydride (LiAlH4) or high-pressure hydrogenation. These methods present significant operational drawbacks, including extreme sensitivity to moisture, the generation of large volumes of aluminum waste, and the risk of dehalogenation side reactions which compromise the integrity of the fluorine or chlorine substituents. Furthermore, the handling of pyrophoric reagents imposes stringent safety protocols and increases capital expenditure for specialized reactor infrastructure. From a supply chain perspective, the reliance on such hazardous chemistry often leads to extended lead times due to rigorous safety audits and limited vendor availability, creating bottlenecks for manufacturers seeking cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The methodology described in CN102341367B introduces a paradigm shift by utilizing an amide-based starting material rather than an acid or ester. The core innovation lies in the conversion of the primary amide groups into N,N-di(alkoxycarbonyl) derivatives prior to reduction. This activation strategy fundamentally alters the electronic properties of the carbonyl group, rendering it susceptible to reduction by much milder hydride sources. By employing dialkyl dicarbonates, such as di-tert-butyl dicarbonate, in the presence of a nucleophilic catalyst like 4-dimethylaminopyridine (DMAP), the process achieves high conversion rates under relatively benign thermal conditions. This approach not only mitigates the safety risks associated with pyrophoric reagents but also enhances the overall atom economy of the synthesis. The ability to utilize sodium borohydride, a commodity chemical, instead of specialized reducing agents represents a significant leap forward in process sustainability and economic viability for the commercial scale-up of complex polymer additives and fine chemicals.
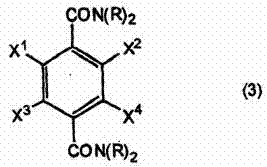
This first transformation is critical as it sets the stage for the subsequent reduction. The reaction proceeds efficiently in solvents such as acetonitrile or toluene, with temperatures ranging from 30°C to 100°C, ensuring that the sensitive halogen substituents remain intact. The resulting intermediate, a protected diamide, exhibits improved solubility and stability, facilitating easier handling and purification before the final reduction step. This strategic decoupling of activation and reduction allows for precise control over impurity profiles, a key concern for regulatory compliance in API synthesis.
Mechanistic Insights into DMAP-Catalyzed N-Protection and Borohydride Reduction
The mechanistic elegance of this process lies in the dual activation provided by the N-protecting groups. In the first step, the nucleophilic attack of the amide nitrogen on the dialkyl dicarbonate is accelerated by the DMAP catalyst. DMAP acts as a potent acyl transfer agent, forming a reactive N-acylpyridinium intermediate that is rapidly attacked by the amide nitrogen. This results in the formation of the N,N-di(alkoxycarbonyl) moiety, effectively transforming the electron-rich amide nitrogen into an electron-withdrawing center. This electronic withdrawal significantly increases the electrophilicity of the adjacent carbonyl carbon, making it far more receptive to nucleophilic attack by hydride ions in the subsequent step. Without this protection, the resonance stabilization of the primary amide would render the carbonyl carbon inert to mild reducing agents like sodium borohydride.
In the second stage, the reduction mechanism involves the delivery of hydride ions from the borohydride species to the activated carbonyl carbon. The presence of a monohydric alcohol, such as methanol or ethanol, plays a crucial role in this phase. The alcohol serves not only as a co-solvent to enhance the solubility of the borohydride salt but also participates in the protonation steps necessary to collapse the tetrahedral intermediate into the final alcohol product. The patent specifies that the reaction is optimally conducted at temperatures between 20°C and 70°C, preventing thermal degradation of the product. This controlled environment ensures that the halogen atoms, particularly fluorine, are not displaced, preserving the structural fidelity required for downstream biological activity. The meticulous balance of reagent stoichiometry, typically using 1 to 3 moles of borohydride per mole of substrate, further minimizes the formation of over-reduced byproducts or unreacted starting materials.
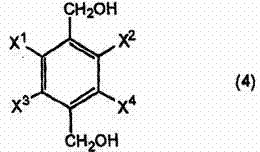
Furthermore, the choice of sodium borohydride as the reducing agent offers distinct advantages over lithium aluminum hydride in terms of workup and waste disposal. The quenching of the reaction can be safely performed with dilute aqueous acid, generating harmless borate salts that are easily separated from the organic product layer. This simplifies the downstream processing and reduces the environmental footprint of the manufacturing process. For quality control teams, this translates to a cleaner crude product with fewer heavy metal contaminants, streamlining the purification process and ensuring that the final high-purity OLED material or pharmaceutical intermediate meets stringent specifications.
How to Synthesize Halogen-Substituted Benzene Dimethanol Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The process begins with the preparation of the protected intermediate, where the ratio of dialkyl dicarbonate to the starting amide is critical. Following the isolation or direct use of the intermediate, the reduction step must be managed to control exotherms and ensure complete conversion. The detailed standardized synthesis steps, including specific solvent ratios, addition rates, and workup procedures, are outlined in the guide below.
- React halogen-substituted terephthalamide with dialkyl dicarbonate in the presence of DMAP catalyst to form the protected diamide intermediate.
- Reduce the protected diamide intermediate using an alkali metal borohydride (e.g., sodium borohydride) in a solvent system containing a monohydric alcohol.
- Quench the reaction with aqueous acid, separate the organic layer, and purify the final halogen-substituted benzene dimethanol via crystallization or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits beyond mere chemical efficiency. The shift away from hazardous, high-energy processes towards a modular, mild-temperature workflow directly impacts the total cost of ownership. By eliminating the need for specialized high-pressure reactors or cryogenic cooling systems required for traditional reductions, manufacturers can utilize existing general-purpose infrastructure, thereby accelerating time-to-market for new products. This flexibility is crucial for maintaining agility in a volatile global market where demand for specialized intermediates can fluctuate rapidly.
- Cost Reduction in Manufacturing: The substitution of expensive and dangerous reducing agents like lithium aluminum hydride with commodity-grade sodium borohydride results in significant raw material cost savings. Additionally, the simplified workup procedure reduces the consumption of quenching agents and minimizes waste disposal costs associated with heavy metal sludge. The high selectivity of the reaction also means less material is lost to side products, improving the overall mass balance and reducing the cost per kilogram of the final active ingredient. These factors collectively contribute to a more lean and economically resilient manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials, such as halogen-substituted terephthalamides and di-tert-butyl dicarbonate, mitigates the risk of supply disruptions. Unlike specialized reagents that may have single-source suppliers, the inputs for this process are produced by multiple global chemical manufacturers, ensuring a stable and competitive supply base. Furthermore, the robustness of the reaction conditions allows for production in diverse geographic locations, enabling a distributed manufacturing strategy that enhances supply chain resilience against regional instabilities or logistics bottlenecks.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram-scale laboratory experiments to multi-kilogram pilot runs without loss of efficiency. The use of common organic solvents like acetonitrile and THF facilitates solvent recovery and recycling, aligning with modern green chemistry principles. The absence of heavy metal catalysts simplifies regulatory compliance and reduces the burden of environmental reporting. This eco-friendly profile is increasingly becoming a prerequisite for partnerships with major multinational corporations committed to sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: Why is the N,N-di(alkoxycarbonyl) protection step necessary?
A: Direct reduction of primary amides to alcohols is chemically challenging and often requires harsh reagents like lithium aluminum hydride. Protecting the amide nitrogen with alkoxycarbonyl groups activates the carbonyl carbon, allowing for efficient reduction using milder and safer reagents like sodium borohydride.
Q: What are the advantages of this method over direct ester reduction?
A: This method avoids the use of expensive and hazardous reducing agents typically required for esters or acids. It utilizes readily available terephthalamides and operates under milder thermal conditions, significantly improving process safety and reducing equipment corrosion risks associated with strong acids or bases.
Q: Can this process be scaled for industrial production?
A: Yes, the process uses common industrial solvents like acetonitrile, toluene, and THF, and operates at moderate temperatures (30-100°C). The reagents, such as di-tert-butyl dicarbonate and sodium borohydride, are commercially available in bulk quantities, making the route highly scalable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogen-Substituted Benzene Dimethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in driving innovation across the pharmaceutical and agrochemical industries. Our team of expert chemists has extensively evaluated the pathway described in CN102341367B and possesses the technical capability to execute this route with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of halogen-substituted benzene dimethanol meets the highest industry standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can optimize your supply chain and accelerate your product development timeline.
