Advanced Pyrroline Chiral Bisphosphine Ligands: Scalable Synthesis and Catalytic Applications
Advanced Pyrroline Chiral Bisphosphine Ligands: Scalable Synthesis and Catalytic Applications
The landscape of asymmetric catalysis is continually evolving, driven by the demand for more efficient and selective chiral ligands. A significant breakthrough in this domain is documented in Chinese patent CN110041365B, which discloses a novel class of pyrroline chiral bisphosphine ligands and their preparation methods. These compounds, characterized by a rigid pyrroline backbone bearing two phosphine oxide groups, represent a substantial advancement in ligand design. The patent details a streamlined synthetic route that leverages a copper-catalyzed [3+2] cycloaddition reaction, offering a direct path to optically pure structures without the need for tedious resolution processes. For R&D directors and procurement specialists in the fine chemical sector, this technology presents a compelling opportunity to access high-performance catalytic tools that can drive down costs and improve the sustainability of pharmaceutical intermediate manufacturing.
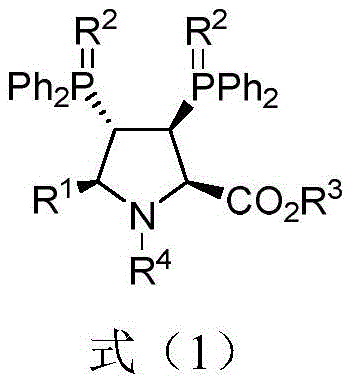
The structural versatility of these ligands is defined by variable substituents at the R1, R2, R3, and R4 positions, allowing for fine-tuning of steric and electronic properties to match specific catalytic requirements. The core innovation lies not just in the molecule itself but in the efficiency of its construction. By utilizing a modular approach where diverse imine esters and phosphine oxide alkenes can be coupled, the methodology supports the rapid generation of ligand libraries. This adaptability is crucial for process chemists who need to screen multiple catalysts to optimize asymmetric transformations. Furthermore, the patent highlights the dual utility of these compounds; they are not merely static ligands but active participants in subsequent catalytic cycles, such as the gold-catalyzed synthesis of complex heterocycles, thereby expanding their value proposition across multiple synthetic workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral phosphine ligands has been fraught with challenges that hinder large-scale adoption. Conventional routes often rely on the resolution of racemic mixtures, a process that inherently caps the maximum theoretical yield at 50% and generates significant chemical waste. Alternatively, many established methods require chiral pool starting materials that are expensive, scarce, or limited in structural diversity. The multi-step sequences typically associated with building the phosphine-carbon framework often involve harsh reaction conditions, sensitive intermediates, and difficult purification steps. These factors contribute to high production costs and extended lead times, creating bottlenecks for supply chain managers who require reliable volumes of high-purity catalysts. Moreover, the lack of modularity in older synthetic strategies makes it difficult to rapidly iterate on ligand structure to improve enantioselectivity for new substrates.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110041365B introduces a highly efficient [3+2] cycloaddition strategy that circumvents these historical hurdles. By reacting a bis-phosphine oxide alkene with an imine ester in the presence of a monovalent copper catalyst and a chiral phosphoramidite ligand, the process constructs the pyrroline ring and installs the chiral centers in a single operational step. This convergent synthesis allows for yields ranging from 62% to 98%, far exceeding the limits of resolution-based methods. The reaction proceeds under mild temperatures, typically between -30°C and 30°C, which reduces energy consumption and enhances safety profiles. The ability to directly access both enantiomers by simply switching the chirality of the auxiliary ligand adds another layer of operational flexibility. This novel approach transforms the production of chiral bisphosphine ligands from a bottleneck into a streamlined, scalable process suitable for modern industrial demands.
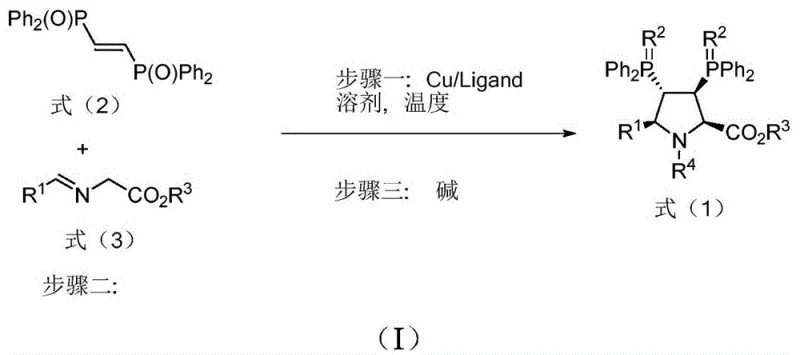
Mechanistic Insights into Copper-Catalyzed Asymmetric Cycloaddition
The success of this synthesis hinges on the precise orchestration of the copper-catalyzed [3+2] cycloaddition mechanism. The reaction initiates with the formation of an active copper species coordinated by the chiral phosphoramidite ligand. This chiral environment dictates the facial selectivity during the addition of the dipolarophile to the azomethine ylide intermediate generated in situ. The rigidity of the resulting pyrroline ring is critical; it locks the relative stereochemistry of the substituents, ensuring high diastereoselectivity. The phosphine oxide groups, while initially serving as stabilizing elements during the cyclization, can be subsequently reduced or modified to yield the active phosphine ligands required for transition metal catalysis. Understanding this mechanistic pathway is vital for R&D teams aiming to replicate or modify the process, as subtle changes in solvent polarity or counter-ions can influence the stability of the metallacycle intermediates.
Beyond their synthesis, these ligands exhibit remarkable performance when applied to other transformations, specifically the intermolecular asymmetric cycloaddition of enynones and nitrones. In this application, the ligand complexes with gold salts to form a highly active cationic gold species. This complex activates the alkyne moiety of the enynone towards nucleophilic attack by the nitrone. The bulky phosphine groups on the pyrroline scaffold create a well-defined chiral pocket that shields one face of the reactive intermediate, forcing the cyclization to proceed with high enantioselectivity. The patent reports the successful synthesis of oxazinofuran derivatives with yields up to 73% and enantiomeric excess values of 77%, demonstrating the ligand's efficacy in constructing complex heterocyclic frameworks found in bioactive molecules.
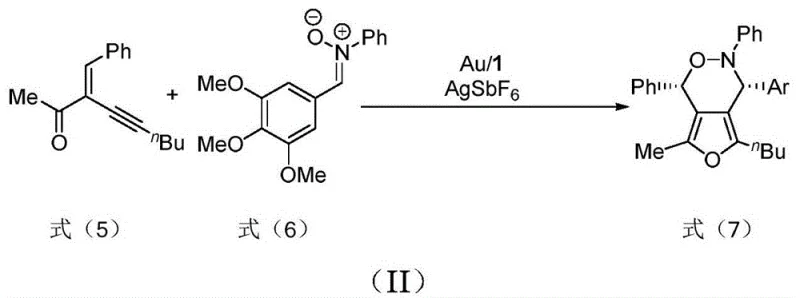
How to Synthesize Pyrroline Chiral Bisphosphine Ligands Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized conditions detailed in the patent to ensure reproducibility and high optical purity. The process is designed to be robust, utilizing commercially available copper salts and standard organic solvents, which facilitates easy technology transfer. Operators must maintain an inert atmosphere throughout the reaction to prevent oxidation of the sensitive copper catalyst and the phosphine species. The sequential addition of reagents and the precise control of temperature during the cycloaddition phase are critical parameters that directly impact the enantiomeric excess of the final product. For a comprehensive guide on the specific molar ratios, solvent choices, and workup procedures, please refer to the standardized protocol below.
- Prepare the catalytic system by mixing a monovalent copper salt (e.g., Cu(CH3CN)4PF6) and a chiral phosphoramidite ligand in a dry solvent like chloroform or ether under inert atmosphere.
- Add the bis-phosphine oxide alkene substrate and the imine ester reactant to the reactor at controlled temperatures between -30°C and 30°C to initiate the [3+2] cycloaddition.
- Treat the reaction mixture with a base such as Cs2CO3 or Et3N to complete the cyclization and isolate the chiral bisphosphine ligand with high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel ligand synthesis technology offers tangible strategic benefits that extend beyond simple chemical performance. The shift from resolution-based methods to direct asymmetric synthesis fundamentally alters the cost structure of producing chiral catalysts. By eliminating the 50% yield loss inherent in racemic resolution, the process effectively doubles the material throughput from the same amount of starting feedstock. This efficiency gain translates directly into substantial cost savings in raw material procurement and waste disposal. Furthermore, the use of mild reaction conditions reduces the energy load on manufacturing facilities, contributing to lower operational expenditures and a smaller carbon footprint, which is increasingly important for meeting corporate sustainability goals.
- Cost Reduction in Manufacturing: The high-yielding nature of the [3+2] cycloaddition significantly lowers the cost per gram of the final ligand. Since the reaction does not require expensive chiral pool starting materials but rather builds chirality catalytically, the input costs are drastically reduced. Additionally, the high selectivity minimizes the formation of byproducts, simplifying the purification process and reducing the consumption of silica gel and solvents during chromatography. This streamlined downstream processing further enhances the overall economic viability of the method for large-scale production.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as phosphine oxide alkenes and imine esters, are structurally simple and can be sourced from multiple suppliers or synthesized via established routes. This reduces the risk of supply disruptions associated with niche, single-source chiral reagents. The robustness of the reaction conditions also means that the process is less susceptible to variations in utility quality or minor operational deviations, ensuring consistent batch-to-batch quality and reliable delivery schedules for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The protocol avoids the use of highly toxic reagents or extreme pressures, making it safer and easier to scale from kilogram to tonne quantities. The simplified workup procedure, which often involves basic aqueous washes and crystallization rather than complex distillations, generates less hazardous waste. This aligns well with stringent environmental regulations and reduces the burden on waste treatment facilities, facilitating smoother regulatory approvals for commercial manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of these pyrroline chiral bisphosphine ligands. The answers are derived directly from the experimental data and scope defined in the underlying patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this technology into their existing synthetic pipelines.
Q: What are the primary advantages of this new pyrroline ligand synthesis method?
A: The method described in patent CN110041365B offers a direct, one-pot [3+2] cycloaddition route that avoids complex resolution steps. It utilizes readily available starting materials and achieves high yields (62%-98%) with excellent enantioselectivity (up to 99% ee), significantly simplifying the production of high-purity chiral ligands.
Q: Can these ligands be used for reactions other than their own synthesis?
A: Yes, beyond serving as valuable intermediates, these pyrroline chiral bisphosphine ligands function effectively as catalysts themselves. Specifically, when complexed with gold salts, they catalyze the intermolecular asymmetric cycloaddition of enynones and nitrones to synthesize oxazinofuran compounds with good reactivity and stereoselectivity.
Q: Is the synthesis process scalable for industrial manufacturing?
A: The process operates under mild conditions (-30°C to 30°C) using common organic solvents and stable copper catalysts. The robustness of the reaction conditions and the high purity of the crude products suggest strong potential for commercial scale-up, reducing the need for extensive downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrroline Chiral Bisphosphine Ligand Supplier
The development of advanced catalytic technologies like those described in CN110041365B underscores the critical need for partners who can bridge the gap between academic innovation and industrial reality. NINGBO INNO PHARMCHEM stands at the forefront of this transition, offering specialized expertise in the custom synthesis and scale-up of complex chiral ligands and pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory results can be translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art chiral HPLC and NMR instrumentation to guarantee the quality of every batch we deliver.
We invite R&D directors and procurement leaders to collaborate with us to evaluate the potential of these pyrroline ligands in your specific applications. Whether you require custom modifications to the ligand structure or a full feasibility study for process integration, our technical team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments tailored to your project needs. Let us help you secure a competitive advantage through superior catalytic performance and supply chain efficiency.
